Difference between revisions of "Vitamin C" - New World Encyclopedia
| Line 46: | Line 46: | ||
}} | }} | ||
| − | '''Vitamin C''' or ''' | + | '''Vitamin C''' (or '''ascorbic acid''') is an [[essential nutrient]] for [[simian|higher primates]], and a small number of other species. The presence of ascorbate is required for a range of essential [[metabolism|metabolic reactions]] in all animals and in plants and is [[biosynthesis|made internally]] by almost all organisms, (humans being one notable exception). It is widely known as the [[vitamin]] that prevents [[scurvy]] in humans |
| − | + | An example of a water-soluble vitamin, an organic molecule needed in small amounts in the diets of some higher animals | |
| + | |||
| + | These molecules serve nearly the same roles in all forms of life, but higher animals have lost the capacity to synthesize them. (hence, referred to as a vitamin - must acquire it from their diets | ||
| + | |||
| + | In living organisms, ascorbate is an [[antioxidant]], as it protects the body against [[oxidative stress]], and is a [[cofactor]] in several vital [[enzyme|enzymatic]] reactions. | ||
As a nutrient, its uses and the daily requirement are matters of on-going debate. As a [[food additive]], vitamin C is used as an [[antioxidant]] [[preservative]] and an [[acidity regulator]]. | As a nutrient, its uses and the daily requirement are matters of on-going debate. As a [[food additive]], vitamin C is used as an [[antioxidant]] [[preservative]] and an [[acidity regulator]]. | ||
| − | + | The name is derived from ''a-'' (anti-) and ''scorbuticus'' ([[scurvy]]) as a shortage of this molecule may lead to scurvy. | |
| + | |||
| + | ==The structure and properties of ascorbic acid== | ||
| + | '''Ascorbic acid''' is a weak [[organic chemistry|organic]] acid that appears as a white, crystalline compound. Structurally, ascorbic acid is related to the six-carbon [[sugar]] [[glucose]], from which most animals are able to derive the molecule in a four-step process. Like glucose, ascorbic acid is [[soluble]] in water. | ||
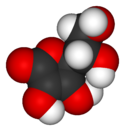
| − | + | [[Image:Ascorbic-acid-3D-vdW.png|thumb|125px|left|A three-dimensional model of an ascorbic acid [[molecule]]. Black represents [[carbon]]; red, [[oxygen]]; and white, [[hydrogen]].]] | |
| − | + | The ionized form of ascorbic acid is known as ''ascorbate''. The ascorbate [[ion]] represents what is called the ''pharmacophore'' of vitamin C; that is, the structural feature (or set of features) that are responsible for the molecule’s biological activity (Gund, 1977). It is the presence of the ascorbate ion that contributes to Vitamin C’s role as a strong reducing agent (''antioxidant''). | |
| − | ''' | ||
| − | [[ | + | Ascorbate occurs in two forms, both of which are [[Chirality (chemistry)|mirror images]] of the same molecular structure. Vitamin C is specifically the [[Enantiomer|<small>L</small>-enantiomer]] of ascorbate; the opposite [[Enantiomer|<small>D</small>-enantiomer]] has no physiological significance. <small>L</small>-ascorbate naturally occurs either attached to a [[hydrogen ion]], forming ''ascorbic acid'', or joined to a [[metal|metal ion]], forming a [[mineral ascorbate]]. |
| − | + | When <small>L</small>-ascorbate carries out its [[Redox|reducing]] function, it is converted to its [[Redox|oxidized]] form, [[Dehydroascorbic acid|<small>L</small>-dehydroascorbate, which can then be converted back to the active <small>L</small>-ascorbate form by specialized [[enzyme]]s and the [[peptide]] [[glutathione]] in the body. | |
| − | |||
== Biological functions == | == Biological functions == | ||
| − | + | ===Vitamin C is a cofactor in the synthesis of collagen=== | |
| − | + | In humans, Vitamin C acts as an [[electron donor]] for eight different enzymes in the synthesis of important biochemicals (Levine, et al., 2000). Three of these enzymes participate in the synthesis of [[collagen]], the major protein of connective tissues, such as skin, bone, cartilage, and tendon (Prockop, et al., 1995; Peterofsky, 1991; Kivirikko and Myllyla, 1985). These reactions add [[Hydroxide|hydroxyl groups]] to the amino acids [[proline]] or [[lysine]] in the collagen molecule, yielding hydroproline, an amino acid that gives allows the collagen molecule to assume its triple helix structure by forming intrastrand hydrogen bonds. Collagen synthesized in the absence of ascorbate has a lower melting temperature than that of the normal protein, making the molecule less stable. The abnormal fibers formed by insufficiently hydroxylated collagen contribute to the skin lesions and blood vessel fragility found in scurvy. Thus, vitamin C is essential to the development and maintenance of [[scar tissue]], [[blood vessel]]s, and cartilage (McGee, date). | |
| − | Three participate in [[collagen]] | ||
| − | + | ===Vitamin C is a beneficial antioxidant=== | |
| + | Along with vitamins A and E, and a and a group of related compounds called coenzyme Q, vitamin C may also act as a general (i.e., non-enzyme-specific) antioxidant. Antioxidants work by making themselves available for energetically favorable oxidation. [[Free radical]]s, which may be produced by the body or generated by environmental conditions, such as exposure to ultraviolet light and tobacco smoke, contain an unpaired electron and thus are highly reactive. They may, for example, oxidize (take electrons from) the lipid molecules that make up cell membranes and other vital tissues, altering their function. Antioxidants like ascorbate react readily with these free radicals before they can react with other compounds in the body. Radicals oxidize ascorbate first to monodehydroascorbate and then dehydroascorbate. The radicals are reduced to water while the oxidized forms of ascorbate are relatively stable and unreactive, and do not cause cellular damage. | ||
== Biosynthesis == | == Biosynthesis == | ||
| − | [[Image:Goat.jpg|thumb| | + | [[Image:Goat.jpg|thumb|225px|Goats, like almost all animals, synthesize their own vitamin C.]] |
| − | The vast majority of animals and plants are able to | + | The vast majority of animals and plants are able to convert glucose into vitamin C through a sequence of four enzyme-driven steps. The glucose needed to produce ascorbate in the [[liver]] of [[mammals]] and [[perching birds]] is extracted from [[glycogen]] (the storage form of glucose) (Bánhegyi and Mándl, 2001). In [[reptiles]] and [[birds]], the biosynthesis of ascorbate is carried out in the [[kidney]]s. |
| − | Among the animals that have lost the ability to | + | Among the animals that have lost the ability to synthesize vitamin C are [[simian]]s, [[guinea pig]]s, the [[red-vented bulbul]],and [[Megabat|fruit-eating bats]] (Expert Group on Vitamins and Minerals, 2003). Thus, along with other members of the ape family, humans have no capability to manufacture vitamin C. The cause of this phenomenon is that the final enzyme in the synthesic process, [[L-gulonolactone oxidase|<small>L</small>-gulonolactone oxidase]], cannot be produced due to a defective gene for the enzyme (Harris, 1996). This genetic [[mutation]], which occurred in the course of evolution, has not led to the extinction of these species because vitamin C is prevalent in their food sources, with many of their natural diets consisting largely of [[fruit]]. |
| − | + | It has been noted that the loss of the ability to synthesize ascorbate strikingly parallels the evolutionary loss of the ability to break down [[uric acid]]. Uric acid and ascorbate are both strong reducing agents. This observation has led to the suggestion that in higher primates, uric acid may have taken over some of the functions of ascorbate (Proctor, 1970). | |
| − | It has been noted that the loss of the ability to synthesize ascorbate strikingly parallels the evolutionary loss of the ability to break down [[uric acid]]. Uric acid and ascorbate are both strong | ||
| − | An adult [[goat]], a typical example of a vitamin C-producing animal, will manufacture more than 13,000 mg of vitamin C per day in normal health and as much as 100,000 mg daily when faced with life-threatening disease, trauma or stress (Stone, 1978). | + | An adult [[goat]], a typical example of a vitamin C-producing animal, will manufacture more than 13,000 mg of vitamin C per day in normal health and as much as 100,000 mg daily when faced with life-threatening disease, trauma or stress (Stone, 1978). Trauma and injury have also been demonstrated to use up large quantities of vitamin C in humans (Long, et al., year). |
| − | + | ==Vitamin-C deficiency is linked to scurvy== | |
| − | |||
| − | == Vitamin-C deficiency is linked to scurvy== | ||
[[Image:James lind.jpg|left|thumb|150px|[[James Lind]], a British Royal Navy surgeon who, in 1747, noted that a quality in certain fruits prevented scurvy.]] | [[Image:James lind.jpg|left|thumb|150px|[[James Lind]], a British Royal Navy surgeon who, in 1747, noted that a quality in certain fruits prevented scurvy.]] | ||
| Line 98: | Line 100: | ||
[[Image:GyorgyiNIH.jpg|thumb|150px|left|Albert Szent-Györgyi, pictured here in 1948, was awarded the 1937 [[Nobel Prize in Physiology or Medicine|Nobel Prize in Medicine]] for the discovery of vitamin C.]] | [[Image:GyorgyiNIH.jpg|thumb|150px|left|Albert Szent-Györgyi, pictured here in 1948, was awarded the 1937 [[Nobel Prize in Physiology or Medicine|Nobel Prize in Medicine]] for the discovery of vitamin C.]] | ||
| − | The name "antiscorbutic" was used in the eighteenth and nineteenth centuries as a general term for those foods known to prevent scurvy, even though the underlying compound responsible for its effectiveness was not yet understood. Between 1928 | + | The name "antiscorbutic" was used in the eighteenth and nineteenth centuries as a general term for those foods known to prevent scurvy, even though the underlying compound responsible for its effectiveness was not yet understood. Between 1928 and 1933, the [[Hungary|Hungarian]] research team of [[Joseph L Svirbely]] and [[Albert Szent-Györgyi]] and, independently, the [[United States|American]] [[Charles Glen King]], first isolated ascorbic acid. |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
== Natural and artificial dietary sources == | == Natural and artificial dietary sources == | ||
| − | [[Image:Rosa canina hips.jpg|right|thumb|[[Rose hips]] are a particularly rich source of vitamin C]] | + | [[Image:Rosa canina hips.jpg|right|thumb|[[Rose hips]] are a particularly rich source of vitamin C.]] |
| − | The richest natural sources are fruits and vegetables | + | The richest natural sources of vitamin C are fruits and vegetables; of the former, the [[camu camu]] fruit and the [[Kakadu plum]] contain the highest concentration of the vitamin. It is also present in some cuts of meat, especially liver, but not in significant quantities. |
| − | + | Since the stability of ascorbic acid decreases with increases in temperature and pH, the nutritional value of foods containing the vitamin may be lost or dimnished in high-temperature processes such as [[pressure cooking]], roasting, frying and [[grilling]]. Longer cooking times also add to this effect, as will copper food vessels, which catalyze the molecule's decomposition by oxidation. | |
| − | Another cause of vitamin C | + | Another cause of vitamin C loss from food is a process called [[leaching]], during which the water-soluble vitamin dissolves into the cooking water. However, vitamin C does not leach from all vegetables at the same rate; research shows that [[broccoli]] seems to retain more of the nutrient than other greens (Combs, 2001). Research has also shown that fresh-cut fruit does not lose significant nutrients when stored in the refrigerator for a few days (Hitt, 2006). |
=== Plant sources === | === Plant sources === | ||
| − | While plants are generally a good source of vitamin C, the amount | + | While plants are generally a good source of vitamin C, the amount depends on the precise variety of the plant, the soil condition, the climate in which it grew, the length of time since it was picked, the storage conditions, and the method of preparation (Danish Veterinary and Food Administration, 2007). |
| + | |||
| + | The following table shows the relative abundance of vitamin C in different raw plant sources (Nutrient Database, date). The amount is given in milligrams per 100 grams of fruit or vegetable and is a rounded average from multiple authoritative sources (Nutrient Data Laboratory, 2007; Healthy Eating Club, 2001; Natural Food Hub, 2001): | ||
| − | |||
<div style="float:left; padding: .5em;"> | <div style="float:left; padding: .5em;"> | ||
{| class="wikitable" border="1" cellpadding="1" | {| class="wikitable" border="1" cellpadding="1" | ||
| Line 279: | Line 259: | ||
=== Vitamin C supplements === | === Vitamin C supplements === | ||
| − | [[Image:RedoxonVitaminC.jpg|thumb|right|Vitamin C is widely available | + | [[Image:RedoxonVitaminC.jpg|thumb|right|Vitamin C is widely available as a tablet and powder. The [[Redoxon]] brand, launched in 1934 by [[Hoffmann-La Roche]], was the first mass-produced synthetic vitamin C.]] |
| − | Vitamin C is the most widely taken | + | Vitamin C is the most widely taken [[nutritional supplement]] (Diet Channel, 2007). It is available in many forms including caplets, tablets, capsules, drink-mix packets, and as part of multi-vitamin formulas. In supplements, vitamin C most often comes in the form of various [[mineral ascorbates]], as they are easier to absorb, more easily tolerated, and provide a source of several [[dietary mineral]]s. Absorption of the vitamin is slowed by large quantities of sugar in the intestines or bloodstream. |
| + | |||
| + | Ascorbic acid was the first vitamin to be artificially produced. Between 1933 and 1934, the British chemists Sir [[Walter Norman Haworth]] and Sir [[Edmund Hirst]] and, independently, the Polish chemist [[Tadeus Reichstein]], succeeded in synthesizing the vitamin. Only Haworth was awarded the 1937 [[Nobel Prize in Chemistry]] for this work, but the process for vitamin C retained Reichstein's name. In 1934 [[Hoffmann–La Roche]] became the first pharmaceutical company to mass-produce synthetic vitamin C, under the brand name of [[Redoxon]]. | ||
| + | |||
| + | Vitamin C is produced from [[glucose]] by two main routes. The [[Tadeusz Reichstein|Reichstein process]], developed in the 1930s, uses a single pre-fermentation followed by a purely chemical route. The modern two-step [[Fermentation|fermentation process]], originally developed in [[China]] in the 1960s, uses additional fermentation to replace part of the later chemical stages. Both processes yield approximately 60% vitamin C from the glucose feed (Competition Commission, 2001). | ||
| + | |||
| + | Research is underway at the [[Scottish Crop Research Institute]] to create a strain of yeast that can synthesise vitamin C in a single fermentation step from [[galactose]], a technology expected to reduce manufacturing costs considerably. | ||
| + | |||
| + | == Daily requirements == | ||
| + | The North American [[Dietary Reference Intake]] recommends 90 [[orders of magnitude (mass)|milligram]]s per day for ran adult male, and and no more than 2 grams per day (2000 milligrams per day) (us rda). Other related species sharing the same inability to produce vitamin C and requiring exogenous vitamin C consume 20 to 80 times this reference intake. There is continuing debate within the scientific community over the best dose schedule (the amount and frequency of intake) of vitamin C for maintaining optimal health in humans (newswire, 2004). It is generally agreed that a balanced diet without supplementation contains enough vitamin C to prevent ''[[acute]]'' scurvy in an average healthy adult, while those who are pregnant, smoke tobacco, or are under stress require slightly more (us rda). | ||
| + | |||
| + | High doses (thousands of milligrams) may result in [[diarrhea]], which is harmless if the dose is reduced immediately. | ||
| + | |||
| + | Recommendations for vitamin C intake have been set by various national agencies: | ||
| + | *40 milligrams per day — the United Kingdom's [[Food Standards Agency]] | ||
| + | *45 milligrams per day — the [[World Health Organization]] (who, 2004) | ||
| + | *60-95 milligrams per day — United States' [[United States National Academy of Sciences|National Academy of Sciences]] | ||
| + | |||
| + | ==Vitamin C as macronutrient== | ||
| + | Most [[simian]]s consume the vitamin in amounts 10 to 20 times higher than that recommended by governments for humans (Milton, 1999). It is thought that the human Vitamin C requirement is far lower than that of Vitamin C-synthesizing mammals due to increased Vitamin-C recycling efficiency (Linster and van Schaftingen, 2006). | ||
| + | |||
| + | There is a strong advocacy movement for large doses of vitamin C, promoting a great deal of added benefits. Many pro-vitamin C organizations promote usage levels well beyond the current [[Dietary Reference Intake]]. The movement is led by scientists and doctors such as [[Robert Cathcart]], [[Ewan Cameron (Vitamin C)|Ewan Cameron]], [[Steve Hickey]], [[Irwin Stone]] and the twice [[Nobel Prize]] laureate [[Linus Pauling]] and the more controversial [[Matthias Rath]]. There is an extensive and growing scientific literature critical of governmental agency dose recommendations (Forman, 1981). The [[biological halflife]] for vitamin C is fairly short, about 30 minutes in blood plasma, a fact which high dose advocates say that mainstream researchers have failed to take into account (Sardi, 2004). | ||
| + | |||
| + | Stone and Pauling calculated, based on the diet of our primate cousins (similar to what our [[common descent]]s are likely to have consumed when the gene mutated), that the optimum daily requirement of vitamin C is around 2300 milligrams for a human requiring 2500 [[calorie|kcal]] a day (Stone, 1972; Pauling, date; Milton, 2003). The established RDA has been criticized by Pauling to be one that will prevent [[acute (medical)|acute]] [[scurvy]], and is not necessarily the dosage for optimal health. | ||
| + | |||
| + | Since its discovery vitamin C has been considered by some enthusiastic proponents a "[[panacea (medicine)|universal panacea]]", although this led to suspicions by others of it being over-hyped. Other proponents of high dose vitamin C consider that if it is given "in the right form, with the proper technique, in frequent enough doses, in high enough doses, along with certain additional agents and for a long enough period of time, it can prevent and, in many cases, cure, a wide range of common and/or lethal diseases, notably the [[common cold]] and [[heart disease]] (Levy, 2002; Rath, date; Pauling, date). Some proponents issued controversial statements involving it being a cure for [[AIDS]],<ref>{{cite web |url=http://allafrica.com/stories/200605220885.html |title=Nigeria: Vitamin C Can Suppress HIV/Aids Virus |accessdate=2006-06-16 |date=[[2006-05-22]] |author= |publisher=allAfrica.com }}</ref> [[H5N1|bird flu]], and [[SARS]].<ref>{{cite journal |author=Hemilä H |title=Vitamin C and SARS coronavirus |journal=J Antimicrob Chemother |volume=52 |issue=6 |pages=1049-50 |year=2003 |pmid=14613951 |url=http://jac.oxfordjournals.org/cgi/content/full/52/6/1049}}</ref><ref>{{cite news | url = http://www.guardian.co.uk/aids/story/0,7369,1483821,00.html |title=Discredited doctor's 'cure' for Aids ignites life-and-death struggle in South Africa | accessdate=2007-02-21 |date=[[2005-05-14]] |author= Boseley, Sarah | publisher = [[The Guardian]] }}</ref><ref>{{cite web |url=http://www4.dr-rath-foundation.org/THE_FOUNDATION/openletter_20060407.htm |title=Open letter from Dr. Matthias Rath MD to German Chancellor Merkel |accessdate=2007-02-21 |date=2005 |author=Rath, Matthias |publisher=Dr. Rath Health Foundation }}</ref> | ||
| + | |||
| + | Probably the most controversial issue, the putative role of ascorbate in the management of AIDS, is still unresolved, more than 16 years after the landmark study published in the prestigious [[Proceedings of National Academy of Sciences]] (USA) showing that non toxic doses of ascorbate suppress [[HIV]] replication ''in vitro''.<ref>{{cite journal |author=Harakeh S, Jariwalla R, Pauling L |title=Suppression of human immunodeficiency virus replication by ascorbate in chronically and acutely infected cells |journal=Proc Natl Acad Sci U S A |volume=87 |issue=18 |pages=7245-9 |year=1990 |pmid=1698293}}</ref> Other studies expanded on those results, but still, no large scale trials have yet been conducted.<ref>{{cite journal |author=Harakeh S, Jariwalla R |title=Comparative study of the anti-HIV activities of ascorbate and thiol-containing reducing agents in chronically HIV-infected cells |journal=Am J Clin Nutr |volume=54 |issue=6 Suppl |pages=1231S-1235S |year=1991 |pmid=1720598}}</ref><ref>{{cite journal |author=Harakeh S, Jariwalla R |title=NF-kappa B-independent suppression of HIV expression by ascorbic acid |journal=AIDS Res Hum Retroviruses |volume=13 |issue=3 |pages=235-9 |year=1997 |pmid=9115810}}</ref><ref>{{cite journal |author=Harakeh S, Jariwalla R |title=Ascorbate effect on cytokine stimulation of HIV production |journal=Nutrition |volume=11 |issue=5 Suppl |pages=684-7 |year= |pmid=8748252}}</ref> | ||
| + | |||
| + | A 1986 study indicates that vitamin C may be important in regulation of endogenous cholesterol synthesis.<ref>{{cite journal |author=Harwood H, Greene Y, Stacpoole P |title=Inhibition of human leukocyte 3-hydroxy-3-methylglutaryl coenzyme A reductase activity by ascorbic acid. An effect mediated by the free radical monodehydroascorbate |journal=J Biol Chem |volume=261 |issue=16 |pages=7127-35 |year=1986 |pmid=3711081}}</ref> | ||
| + | |||
| + | In January 2007 the US [[Food and Drug Administration]] approved a new trial of intravenous vitamin C as a cancer treatment for "patients who have exhausted all other conventional treatment options." Additional studies over several years would be needed to demonstrate whether it is effective.<ref>{{cite web |url=http://www.physorg.com/news87833644.html |title=FDA OKs vitamin C trial for cancer |accessdate=2007-04-06 |last= |first= |authorlink= |coauthors= |date=January 12, 2007 |year= |month= |format= |work= |publisher=[[Physorg.com]] |pages= |language= |archiveurl= |archivedate= |quote=Federal approval of a clinical trial on intravenous vitamin C as a cancer treatment lends credence to alternative cancer care, U.S. researchers said.}}</ref> | ||
| − | |||
| − | |||
==References== | ==References== | ||
| − | * | + | *Atkins, P. and L. Jones. 2005. ''Chemical Principles'', 3rd ed. New York: W.H. Freeman. |
| − | * | + | *Bánhegyi, G. and J. Mándl. 2001. The hepatic glycogenoreticular system. ''Pathol Oncol Res'' 7(2):107-10. PMID 11458272 |
| − | * | + | *Combs, G.F. 2001. ''The Vitamins, Fundamental Aspects in Nutrition and Health'', 2nd ed. San Diego, CA: Academic Press. |
| − | + | *Competition Commission. 2001. [cite web |url=http://www.competition-commission.org.uk/rep_pub/reports/2001/fulltext/456a4.2.pdf The production of vitamin C.] Retrieved February 20, 2007. | |
| + | *Danish Veterinary and Food Administration. 2007. [http://www.uk.foedevarestyrelsen.dk/Nutrition/Vitamin_mineral_content_is_stable/forside.htm The vitamin and mineral content is stable.] Retrieved March 7, 2007. | ||
| + | *The Diet Channel. 2007. [http://www.thedietchannel.com/Vitamin-C.htm Vitamin C: General Info.] Retrieved June 30, 2007. | ||
*Expert Group on Vitamins and Minerals. 2003. [http://www.food.gov.uk/multimedia/pdfs/evm_c.pdf Vitamin C – Risk Assessment.] UK Food Standards Agency. Retrieved February 19, 2007. | *Expert Group on Vitamins and Minerals. 2003. [http://www.food.gov.uk/multimedia/pdfs/evm_c.pdf Vitamin C – Risk Assessment.] UK Food Standards Agency. Retrieved February 19, 2007. | ||
| + | *Forman, R. 1981. [http://www.seanet.com/~alexs/ascorbate/198x/forman-r-med_hypotheses-1981-v7-n8-p1009.htm Medical Resistance to Innovation.] ''Medical Hypotheses'' 7(8):1009-17. Retrieved February 23, 2007 | ||
*Harris, J.R. 1996. ''Ascorbic Acid: Subcellular Biochemistry''. New York: Springer. ISBN 0-306-45148-4 | *Harris, J.R. 1996. ''Ascorbic Acid: Subcellular Biochemistry''. New York: Springer. ISBN 0-306-45148-4 | ||
| − | * | + | *Healthy Eating Club. 2001. [http://www.healthyeatingclub.com/info/books-phds/books/foodfacts/html/data/data4i.html Vitamin C Food Data Chart.] Retrieved March 7, 2007. |
| − | * | + | *Hediger, M.A. 2002. [http://www.nature.com/nm/journal/v8/n5/full/nm0502-445.html New view at C.] ''Nature Medicine'' 8:445-6. |
| − | * | + | *Hitt, Miranda. 2006. [http://www.webmd.com/content/article/123/115022.htm Fresh-Cut Fruit May Keep Its Vitamins.] Retrieved February 25, 2007. |
| + | *Institute of Medicine of the National Academies. 2001. [http://www.iom.edu/Object.File/Master/7/296/webtablevitamins.pdf US Recommended Dietary Allowance (RDA).] Retrieved February 19, 2007. | ||
*Kivirikko, K.I. and R. Myllyla. 1985. Post-translational processing of procollagens. ''Ann NY Acad Sci'' 460:187–201. | *Kivirikko, K.I. and R. Myllyla. 1985. Post-translational processing of procollagens. ''Ann NY Acad Sci'' 460:187–201. | ||
| − | * | + | *Levy, T.E. 2002. ''Curing the Incurable: Vitamin C, Infectious Diseases, and Toxins''. Livon Books. ISBN 1-4010-6963-0 |
| − | |||
| − | |||
| − | |||
| − | |||
*Linster, C. and E. Van Schaftingen. 2006. [http://www.blackwell-synergy.com/doi/full/10.1111/j.1742-4658.2006.05607.x?cookieSet=1 Vitamin C: Biosynthesis, recycling and degradation in mammals.] Retrieved April 30, 2007. | *Linster, C. and E. Van Schaftingen. 2006. [http://www.blackwell-synergy.com/doi/full/10.1111/j.1742-4658.2006.05607.x?cookieSet=1 Vitamin C: Biosynthesis, recycling and degradation in mammals.] Retrieved April 30, 2007. | ||
*Long, C. et al. 2003. Ascorbic acid dynamics in the seriously ill and injured. ''Journal of Surgical Research'' 109(2):144–8. | *Long, C. et al. 2003. Ascorbic acid dynamics in the seriously ill and injured. ''Journal of Surgical Research'' 109(2):144–8. | ||
*Martini, E. 2002. [http://www.ncbi.nlm.nih.gov/sites/entrez?cmd=Retrieve&db=PubMed&list_uids=12422875&dopt=Abstract Jacques Cartier witnesses a treatment for scurvy.] ''Vesalius'' 8(1):2-6. Retrieved February 25, 2007. | *Martini, E. 2002. [http://www.ncbi.nlm.nih.gov/sites/entrez?cmd=Retrieve&db=PubMed&list_uids=12422875&dopt=Abstract Jacques Cartier witnesses a treatment for scurvy.] ''Vesalius'' 8(1):2-6. Retrieved February 25, 2007. | ||
| + | *McGee, W. 2007. [http://www.nlm.nih.gov/medlineplus/ency/article/002404.htm Ascorbic acid.] Medical Encyclopedia. Retrieved June 30, 2007. | ||
| + | *Meister, A. 1994. Glutathione-ascorbic acid antioxidant system in animals. ''J Biol Chem'' 269(213): 9397-400. PMID 8144521 | ||
| + | *Milton, K. 2003. [http://nature.berkeley.edu/miltonlab/pdfs/kmilton_micronutrient.pdf Micronutrient intakes of wild primates: are humans different?] ''Comp Biochem Physiol'' 136(1):47-59. PMID 14527629 | ||
| + | *Milton, K. 1999. Nutritional characteristics of wild primate foods: do the diets of our closest living relatives have lessons for us? ''Nutrition'' 15(6):488-98. | ||
| + | *The Natural Food Hub. 2001. [http://www.naturalhub.com/natural_food_guide_fruit_vitamin_c.htm Natural food-Fruit Vitamin C Content.] March 7, 2007. | ||
| + | *Nutrient Data Laboratory of the US Agricultural Research Service. 2007. [http://www.nal.usda.gov/fnic/foodcomp/search/ National Nutrient Database.] Retrieved March 7, 2007. | ||
| + | *Padayatty, S., Katz, A., Wang, Y., Eck, P., Kwon, O., Lee, J., Chen, S., Corpe, C., Dutta, A., Dutta, S., and M. Levine. 2003. Vitamin C as an antioxidant: evaluation of its role in disease prevention. ''J Am Coll Nutr'' 22(1):18-35. | ||
| + | *Pauling, L. 1970. Evolution and the need for ascorbic acid. ''Proc Natl Acad Sci'' 67(4):1643-8. | ||
| + | *Peterkofsky, B. 1991. Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. ''Am J Clin Nutr'' 54:1135S–40S. | ||
| + | *Prockop, D.J. and K.I. Kivirikko. 1995. Collagens: molecular biology, diseases, and potentials for therapy. ''Annu Rev Biochem'' 64:403–34. | ||
| + | *Proctor, P. 1970. Similar functions of uric acid and ascorbate in man? ''Nature'' 228(5274): 868. | ||
*Sardi, B. 2004. [http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=109&STORY=/www/story/07-06-2004/0002204911 Linus Pauling Vindicated; Researchers Claim RDA For Vitamin C is Flawed.] Knowledge of Health. Retrieved February 20, 2007. | *Sardi, B. 2004. [http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=109&STORY=/www/story/07-06-2004/0002204911 Linus Pauling Vindicated; Researchers Claim RDA For Vitamin C is Flawed.] Knowledge of Health. Retrieved February 20, 2007. | ||
| − | * | + | *Svirbelf, J.L. and A. Szent-Gyorgyi. 1932. [http://profiles.nlm.nih.gov/WG/B/B/G/W/_/wgbbgw.pdf The Chemical Nature Of Vitamin C]. The National Library of Medicine. Accessed June 30, 2007. |
| − | |||
| − | |||
*Sardi, B. 2004. [http://www.newmediaexplorer.org/chris/2004/07/09/the_vitamin_c_fanatics_were_right_all_along.htm The Vitamin C Fanatics Were Right All Along.] Knowledge of Health. Retrieved February 22, 2007. | *Sardi, B. 2004. [http://www.newmediaexplorer.org/chris/2004/07/09/the_vitamin_c_fanatics_were_right_all_along.htm The Vitamin C Fanatics Were Right All Along.] Knowledge of Health. Retrieved February 22, 2007. | ||
| − | * | + | *Stipanuk, M.H. 2000. "Biochemical and Physiological Aspects of Human Nutrition." Philadelphia: Saunders. |
| + | *Stone, I. 1979. [http://www.seanet.com/~alexs/ascorbate/197x/stone-i-orthomol_psych-1979-v8-n2-p58.htm Eight Decades of Scurvy. The Case History of a Misleading Dietary Hypothesis.] ''Orthomolecular Psychiatry'' 8(2):58-62. Retrieved April 4, 2007. | ||
*Stone, I. 1972. ''The Healing Factor: Vitamin C Against Disease''. New York:Grosset and Dunlap. ISBN 0-448-11693-6 | *Stone, I. 1972. ''The Healing Factor: Vitamin C Against Disease''. New York:Grosset and Dunlap. ISBN 0-448-11693-6 | ||
| − | * | + | *Stryer, L. 1995. ''Biochemistry'', 4th edition. New York: W.H. Freeman. ISBN 0716720094 |
| − | + | *World Health Organization. 2004. [http://whqlibdoc.who.int/publications/2004/9241546123_chap7.pdf ''Vitamin and mineral requirements in human nutrition'', 2nd ed.] Retrieved February 20, 2007. | |
| − | * | + | . |
| − | |||
== Further reading == | == Further reading == | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
*{{cite book |last=Pauling |first=Linus |authorlink=Linus Pauling |coauthors= |title=Vitamin C and the Common Cold |year=1970 |publisher=W. H. Freeman & Company |location= |isbn=071670160X}} | *{{cite book |last=Pauling |first=Linus |authorlink=Linus Pauling |coauthors= |title=Vitamin C and the Common Cold |year=1970 |publisher=W. H. Freeman & Company |location= |isbn=071670160X}} | ||
*{{cite book |last=Pauling |first=Linus |authorlink=Linus Pauling |coauthors= |title=Vitamin C, the Common Cold, and the Flu'' |year=1976 |publisher=W H Freeman & Co |location= |isbn=0716703610}} | *{{cite book |last=Pauling |first=Linus |authorlink=Linus Pauling |coauthors= |title=Vitamin C, the Common Cold, and the Flu'' |year=1976 |publisher=W H Freeman & Co |location= |isbn=0716703610}} | ||
Revision as of 19:56, 1 July 2007
| |
| |
Vitamin C
| |
| Systematic name | |
| IUPAC name 2-oxo-L-threo-hexono-1,4- lactone-2,3-enediol or (R)-3,4-dihydroxy-5-((S)- 1,2-dihydroxyethyl)furan-2(5H)-one | |
| Identifiers | |
| CAS number | 50-81-7 |
| ATC code | A11G |
| PubChem | 644104 |
| Chemical data | |
| Formula | C6H8O6 |
| Mol. weight | 176.13 grams per mol |
| Synonyms | L-ascorbate |
| Physical data | |
| Melt. point | 190°C (374°F) |
| Pharmacokinetic data | |
| Bioavailability | rapid & complete |
| Protein binding | negligible |
| Metabolism | ? |
| Half life | 30 minutes |
| Excretion | renal |
| Therapeutic considerations | |
| Pregnancy cat. | A |
| Legal status | general public availability |
| Routes | oral |
Vitamin C (or ascorbic acid) is an essential nutrient for higher primates, and a small number of other species. The presence of ascorbate is required for a range of essential metabolic reactions in all animals and in plants and is made internally by almost all organisms, (humans being one notable exception). It is widely known as the vitamin that prevents scurvy in humans
An example of a water-soluble vitamin, an organic molecule needed in small amounts in the diets of some higher animals
These molecules serve nearly the same roles in all forms of life, but higher animals have lost the capacity to synthesize them. (hence, referred to as a vitamin - must acquire it from their diets
In living organisms, ascorbate is an antioxidant, as it protects the body against oxidative stress, and is a cofactor in several vital enzymatic reactions.
As a nutrient, its uses and the daily requirement are matters of on-going debate. As a food additive, vitamin C is used as an antioxidant preservative and an acidity regulator.
The name is derived from a- (anti-) and scorbuticus (scurvy) as a shortage of this molecule may lead to scurvy.
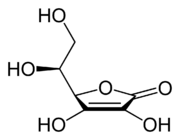
The structure and properties of ascorbic acid
Ascorbic acid is a weak organic acid that appears as a white, crystalline compound. Structurally, ascorbic acid is related to the six-carbon sugar glucose, from which most animals are able to derive the molecule in a four-step process. Like glucose, ascorbic acid is soluble in water.
The ionized form of ascorbic acid is known as ascorbate. The ascorbate ion represents what is called the pharmacophore of vitamin C; that is, the structural feature (or set of features) that are responsible for the molecule’s biological activity (Gund, 1977). It is the presence of the ascorbate ion that contributes to Vitamin C’s role as a strong reducing agent (antioxidant).
Ascorbate occurs in two forms, both of which are mirror images of the same molecular structure. Vitamin C is specifically the L-enantiomer of ascorbate; the opposite D-enantiomer has no physiological significance. L-ascorbate naturally occurs either attached to a hydrogen ion, forming ascorbic acid, or joined to a metal ion, forming a mineral ascorbate.
When L-ascorbate carries out its reducing function, it is converted to its oxidized form, [[Dehydroascorbic acid|L-dehydroascorbate, which can then be converted back to the active L-ascorbate form by specialized enzymes and the peptide glutathione in the body.
Biological functions
Vitamin C is a cofactor in the synthesis of collagen
In humans, Vitamin C acts as an electron donor for eight different enzymes in the synthesis of important biochemicals (Levine, et al., 2000). Three of these enzymes participate in the synthesis of collagen, the major protein of connective tissues, such as skin, bone, cartilage, and tendon (Prockop, et al., 1995; Peterofsky, 1991; Kivirikko and Myllyla, 1985). These reactions add hydroxyl groups to the amino acids proline or lysine in the collagen molecule, yielding hydroproline, an amino acid that gives allows the collagen molecule to assume its triple helix structure by forming intrastrand hydrogen bonds. Collagen synthesized in the absence of ascorbate has a lower melting temperature than that of the normal protein, making the molecule less stable. The abnormal fibers formed by insufficiently hydroxylated collagen contribute to the skin lesions and blood vessel fragility found in scurvy. Thus, vitamin C is essential to the development and maintenance of scar tissue, blood vessels, and cartilage (McGee, date).
Vitamin C is a beneficial antioxidant
Along with vitamins A and E, and a and a group of related compounds called coenzyme Q, vitamin C may also act as a general (i.e., non-enzyme-specific) antioxidant. Antioxidants work by making themselves available for energetically favorable oxidation. Free radicals, which may be produced by the body or generated by environmental conditions, such as exposure to ultraviolet light and tobacco smoke, contain an unpaired electron and thus are highly reactive. They may, for example, oxidize (take electrons from) the lipid molecules that make up cell membranes and other vital tissues, altering their function. Antioxidants like ascorbate react readily with these free radicals before they can react with other compounds in the body. Radicals oxidize ascorbate first to monodehydroascorbate and then dehydroascorbate. The radicals are reduced to water while the oxidized forms of ascorbate are relatively stable and unreactive, and do not cause cellular damage.
Biosynthesis
The vast majority of animals and plants are able to convert glucose into vitamin C through a sequence of four enzyme-driven steps. The glucose needed to produce ascorbate in the liver of mammals and perching birds is extracted from glycogen (the storage form of glucose) (Bánhegyi and Mándl, 2001). In reptiles and birds, the biosynthesis of ascorbate is carried out in the kidneys.
Among the animals that have lost the ability to synthesize vitamin C are simians, guinea pigs, the red-vented bulbul,and fruit-eating bats (Expert Group on Vitamins and Minerals, 2003). Thus, along with other members of the ape family, humans have no capability to manufacture vitamin C. The cause of this phenomenon is that the final enzyme in the synthesic process, L-gulonolactone oxidase, cannot be produced due to a defective gene for the enzyme (Harris, 1996). This genetic mutation, which occurred in the course of evolution, has not led to the extinction of these species because vitamin C is prevalent in their food sources, with many of their natural diets consisting largely of fruit.
It has been noted that the loss of the ability to synthesize ascorbate strikingly parallels the evolutionary loss of the ability to break down uric acid. Uric acid and ascorbate are both strong reducing agents. This observation has led to the suggestion that in higher primates, uric acid may have taken over some of the functions of ascorbate (Proctor, 1970).
An adult goat, a typical example of a vitamin C-producing animal, will manufacture more than 13,000 mg of vitamin C per day in normal health and as much as 100,000 mg daily when faced with life-threatening disease, trauma or stress (Stone, 1978). Trauma and injury have also been demonstrated to use up large quantities of vitamin C in humans (Long, et al., year).
Vitamin-C deficiency is linked to scurvy

Scurvy is a disease caused by vitamin-C deficiency in humans and other animals incapable of synthesizing ascorbic acid. Since the body cannot store vitamin C, supplies are quickly depleted if fresh supplies are not consumed through the digestive system (McGee, 2007). As mentioned above, collagen synthesized in vitro in the absence of the vitamin C is too unstable to meet its function, which results in the skin lesions and fragile blood vessels characteristic of scurvy.
Throughout history, scurvy was common among those with poor access to fresh fruit and vegetables, such as remote, isolated sailors and soldiers. For example, in 1536, the French explorer Jacques Cartier, exploring the St. Lawrence River, relied on the local natives' medicinal knowledge to save his men who were dying of scurvy. He boiled the needles of the arbor vitae tree to make a tea that was later shown to contain 50 mg of vitamin C per 100 grams (Martini, 2002).

While the earliest documented case of scurvy was described by Hippocrates around the year 400 B.C.E., the first attempt to provide a scientific explanation for the disease came in 1747 from the Scottish physician James Lind, a ship's surgeon in the British Royal Navy. Lind's work was slow to be noticed; some 50 years elapsed from the publication of Lind's treatise before the British navy adopted lemons as standard issue at sea. After limes were substituted in 1856, British sailors began to be known as “limeys.”

The name "antiscorbutic" was used in the eighteenth and nineteenth centuries as a general term for those foods known to prevent scurvy, even though the underlying compound responsible for its effectiveness was not yet understood. Between 1928 and 1933, the Hungarian research team of Joseph L Svirbely and Albert Szent-Györgyi and, independently, the American Charles Glen King, first isolated ascorbic acid.
Natural and artificial dietary sources
The richest natural sources of vitamin C are fruits and vegetables; of the former, the camu camu fruit and the Kakadu plum contain the highest concentration of the vitamin. It is also present in some cuts of meat, especially liver, but not in significant quantities.
Since the stability of ascorbic acid decreases with increases in temperature and pH, the nutritional value of foods containing the vitamin may be lost or dimnished in high-temperature processes such as pressure cooking, roasting, frying and grilling. Longer cooking times also add to this effect, as will copper food vessels, which catalyze the molecule's decomposition by oxidation.
Another cause of vitamin C loss from food is a process called leaching, during which the water-soluble vitamin dissolves into the cooking water. However, vitamin C does not leach from all vegetables at the same rate; research shows that broccoli seems to retain more of the nutrient than other greens (Combs, 2001). Research has also shown that fresh-cut fruit does not lose significant nutrients when stored in the refrigerator for a few days (Hitt, 2006).
Plant sources
While plants are generally a good source of vitamin C, the amount depends on the precise variety of the plant, the soil condition, the climate in which it grew, the length of time since it was picked, the storage conditions, and the method of preparation (Danish Veterinary and Food Administration, 2007).
The following table shows the relative abundance of vitamin C in different raw plant sources (Nutrient Database, date). The amount is given in milligrams per 100 grams of fruit or vegetable and is a rounded average from multiple authoritative sources (Nutrient Data Laboratory, 2007; Healthy Eating Club, 2001; Natural Food Hub, 2001):
| Plant source | Amount (mg / 100g) |
|---|---|
| Kakadu plum | 3150 |
| Camu Camu | 2800 |
| Rose hip | 2000 |
| Acerola | 1600 |
| Amla | 720 |
| Jujube | 500 |
| Baobab | 400 |
| Blackcurrant | 200 |
| Red pepper | 190 |
| Parsley | 130 |
| Seabuckthorn | 120 |
| Guava | 100 |
| Kiwifruit | 90 |
| Broccoli | 90 |
| Loganberry | 80 |
| Redcurrant | 80 |
| Brussels sprouts | 80 |
| Lychee | 70 |
| Cloudberry | 60 |
| Persimmon | 60 |
| Papaya | 60 |
| Plant source | Amount (mg / 100g) |
|---|---|
| Strawberry | 60 |
| Orange | 50 |
| Lemon | 40 |
| Melon, cantaloupe | 40 |
| Cauliflower | 40 |
| Grapefruit | 30 |
| Raspberry | 30 |
| Tangerine | 30 |
| Mandarin orange | 30 |
| Passion fruit | 30 |
| Spinach | 30 |
| Cabbage raw green | 30 |
| Lime | 20 |
| Mango | 20 |
| Potato | 20 |
| Melon, honeydew | 20 |
| Mango | 16 |
| Tomato | 10 |
| Blueberry | 10 |
| Pineapple | 10 |
| Pawpaw | 10 |
| Plant source | Amount (mg / 100g) |
|---|---|
| Grape | 10 |
| Apricot | 10 |
| Plum | 10 |
| Watermelon | 10 |
| Banana | 9 |
| Carrot | 9 |
| Avocado | 8 |
| Crabapple | 8 |
| Peach | 7 |
| Apple | 6 |
| Blackberry | 6 |
| Beetroot | 5 |
| Pear | 4 |
| Lettuce | 4 |
| Cucumber | 3 |
| Eggplant | 2 |
| Fig | 2 |
| Bilberry | 1 |
| Horned melon | 0.5 |
| Medlar | 0.3 |
Vitamin C supplements
Vitamin C is the most widely taken nutritional supplement (Diet Channel, 2007). It is available in many forms including caplets, tablets, capsules, drink-mix packets, and as part of multi-vitamin formulas. In supplements, vitamin C most often comes in the form of various mineral ascorbates, as they are easier to absorb, more easily tolerated, and provide a source of several dietary minerals. Absorption of the vitamin is slowed by large quantities of sugar in the intestines or bloodstream.
Ascorbic acid was the first vitamin to be artificially produced. Between 1933 and 1934, the British chemists Sir Walter Norman Haworth and Sir Edmund Hirst and, independently, the Polish chemist Tadeus Reichstein, succeeded in synthesizing the vitamin. Only Haworth was awarded the 1937 Nobel Prize in Chemistry for this work, but the process for vitamin C retained Reichstein's name. In 1934 Hoffmann–La Roche became the first pharmaceutical company to mass-produce synthetic vitamin C, under the brand name of Redoxon.
Vitamin C is produced from glucose by two main routes. The Reichstein process, developed in the 1930s, uses a single pre-fermentation followed by a purely chemical route. The modern two-step fermentation process, originally developed in China in the 1960s, uses additional fermentation to replace part of the later chemical stages. Both processes yield approximately 60% vitamin C from the glucose feed (Competition Commission, 2001).
Research is underway at the Scottish Crop Research Institute to create a strain of yeast that can synthesise vitamin C in a single fermentation step from galactose, a technology expected to reduce manufacturing costs considerably.
Daily requirements
The North American Dietary Reference Intake recommends 90 milligrams per day for ran adult male, and and no more than 2 grams per day (2000 milligrams per day) (us rda). Other related species sharing the same inability to produce vitamin C and requiring exogenous vitamin C consume 20 to 80 times this reference intake. There is continuing debate within the scientific community over the best dose schedule (the amount and frequency of intake) of vitamin C for maintaining optimal health in humans (newswire, 2004). It is generally agreed that a balanced diet without supplementation contains enough vitamin C to prevent acute scurvy in an average healthy adult, while those who are pregnant, smoke tobacco, or are under stress require slightly more (us rda).
High doses (thousands of milligrams) may result in diarrhea, which is harmless if the dose is reduced immediately.
Recommendations for vitamin C intake have been set by various national agencies:
- 40 milligrams per day — the United Kingdom's Food Standards Agency
- 45 milligrams per day — the World Health Organization (who, 2004)
- 60-95 milligrams per day — United States' National Academy of Sciences
Vitamin C as macronutrient
Most simians consume the vitamin in amounts 10 to 20 times higher than that recommended by governments for humans (Milton, 1999). It is thought that the human Vitamin C requirement is far lower than that of Vitamin C-synthesizing mammals due to increased Vitamin-C recycling efficiency (Linster and van Schaftingen, 2006).
There is a strong advocacy movement for large doses of vitamin C, promoting a great deal of added benefits. Many pro-vitamin C organizations promote usage levels well beyond the current Dietary Reference Intake. The movement is led by scientists and doctors such as Robert Cathcart, Ewan Cameron, Steve Hickey, Irwin Stone and the twice Nobel Prize laureate Linus Pauling and the more controversial Matthias Rath. There is an extensive and growing scientific literature critical of governmental agency dose recommendations (Forman, 1981). The biological halflife for vitamin C is fairly short, about 30 minutes in blood plasma, a fact which high dose advocates say that mainstream researchers have failed to take into account (Sardi, 2004).
Stone and Pauling calculated, based on the diet of our primate cousins (similar to what our common descents are likely to have consumed when the gene mutated), that the optimum daily requirement of vitamin C is around 2300 milligrams for a human requiring 2500 kcal a day (Stone, 1972; Pauling, date; Milton, 2003). The established RDA has been criticized by Pauling to be one that will prevent acute scurvy, and is not necessarily the dosage for optimal health.
Since its discovery vitamin C has been considered by some enthusiastic proponents a "universal panacea", although this led to suspicions by others of it being over-hyped. Other proponents of high dose vitamin C consider that if it is given "in the right form, with the proper technique, in frequent enough doses, in high enough doses, along with certain additional agents and for a long enough period of time, it can prevent and, in many cases, cure, a wide range of common and/or lethal diseases, notably the common cold and heart disease (Levy, 2002; Rath, date; Pauling, date). Some proponents issued controversial statements involving it being a cure for AIDS,[1] bird flu, and SARS.[2][3][4]
Probably the most controversial issue, the putative role of ascorbate in the management of AIDS, is still unresolved, more than 16 years after the landmark study published in the prestigious Proceedings of National Academy of Sciences (USA) showing that non toxic doses of ascorbate suppress HIV replication in vitro.[5] Other studies expanded on those results, but still, no large scale trials have yet been conducted.[6][7][8]
A 1986 study indicates that vitamin C may be important in regulation of endogenous cholesterol synthesis.[9]
In January 2007 the US Food and Drug Administration approved a new trial of intravenous vitamin C as a cancer treatment for "patients who have exhausted all other conventional treatment options." Additional studies over several years would be needed to demonstrate whether it is effective.[10]
ReferencesISBN links support NWE through referral fees
- Atkins, P. and L. Jones. 2005. Chemical Principles, 3rd ed. New York: W.H. Freeman.
- Bánhegyi, G. and J. Mándl. 2001. The hepatic glycogenoreticular system. Pathol Oncol Res 7(2):107-10. PMID 11458272
- Combs, G.F. 2001. The Vitamins, Fundamental Aspects in Nutrition and Health, 2nd ed. San Diego, CA: Academic Press.
- Competition Commission. 2001. [cite web |url=http://www.competition-commission.org.uk/rep_pub/reports/2001/fulltext/456a4.2.pdf The production of vitamin C.] Retrieved February 20, 2007.
- Danish Veterinary and Food Administration. 2007. The vitamin and mineral content is stable. Retrieved March 7, 2007.
- The Diet Channel. 2007. Vitamin C: General Info. Retrieved June 30, 2007.
- Expert Group on Vitamins and Minerals. 2003. Vitamin C – Risk Assessment. UK Food Standards Agency. Retrieved February 19, 2007.
- Forman, R. 1981. Medical Resistance to Innovation. Medical Hypotheses 7(8):1009-17. Retrieved February 23, 2007
- Harris, J.R. 1996. Ascorbic Acid: Subcellular Biochemistry. New York: Springer. ISBN 0-306-45148-4
- Healthy Eating Club. 2001. Vitamin C Food Data Chart. Retrieved March 7, 2007.
- Hediger, M.A. 2002. New view at C. Nature Medicine 8:445-6.
- Hitt, Miranda. 2006. Fresh-Cut Fruit May Keep Its Vitamins. Retrieved February 25, 2007.
- Institute of Medicine of the National Academies. 2001. US Recommended Dietary Allowance (RDA). Retrieved February 19, 2007.
- Kivirikko, K.I. and R. Myllyla. 1985. Post-translational processing of procollagens. Ann NY Acad Sci 460:187–201.
- Levy, T.E. 2002. Curing the Incurable: Vitamin C, Infectious Diseases, and Toxins. Livon Books. ISBN 1-4010-6963-0
- Linster, C. and E. Van Schaftingen. 2006. Vitamin C: Biosynthesis, recycling and degradation in mammals. Retrieved April 30, 2007.
- Long, C. et al. 2003. Ascorbic acid dynamics in the seriously ill and injured. Journal of Surgical Research 109(2):144–8.
- Martini, E. 2002. Jacques Cartier witnesses a treatment for scurvy. Vesalius 8(1):2-6. Retrieved February 25, 2007.
- McGee, W. 2007. Ascorbic acid. Medical Encyclopedia. Retrieved June 30, 2007.
- Meister, A. 1994. Glutathione-ascorbic acid antioxidant system in animals. J Biol Chem 269(213): 9397-400. PMID 8144521
- Milton, K. 2003. Micronutrient intakes of wild primates: are humans different? Comp Biochem Physiol 136(1):47-59. PMID 14527629
- Milton, K. 1999. Nutritional characteristics of wild primate foods: do the diets of our closest living relatives have lessons for us? Nutrition 15(6):488-98.
- The Natural Food Hub. 2001. Natural food-Fruit Vitamin C Content. March 7, 2007.
- Nutrient Data Laboratory of the US Agricultural Research Service. 2007. National Nutrient Database. Retrieved March 7, 2007.
- Padayatty, S., Katz, A., Wang, Y., Eck, P., Kwon, O., Lee, J., Chen, S., Corpe, C., Dutta, A., Dutta, S., and M. Levine. 2003. Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr 22(1):18-35.
- Pauling, L. 1970. Evolution and the need for ascorbic acid. Proc Natl Acad Sci 67(4):1643-8.
- Peterkofsky, B. 1991. Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am J Clin Nutr 54:1135S–40S.
- Prockop, D.J. and K.I. Kivirikko. 1995. Collagens: molecular biology, diseases, and potentials for therapy. Annu Rev Biochem 64:403–34.
- Proctor, P. 1970. Similar functions of uric acid and ascorbate in man? Nature 228(5274): 868.
- Sardi, B. 2004. Linus Pauling Vindicated; Researchers Claim RDA For Vitamin C is Flawed. Knowledge of Health. Retrieved February 20, 2007.
- Svirbelf, J.L. and A. Szent-Gyorgyi. 1932. The Chemical Nature Of Vitamin C. The National Library of Medicine. Accessed June 30, 2007.
- Sardi, B. 2004. The Vitamin C Fanatics Were Right All Along. Knowledge of Health. Retrieved February 22, 2007.
- Stipanuk, M.H. 2000. "Biochemical and Physiological Aspects of Human Nutrition." Philadelphia: Saunders.
- Stone, I. 1979. Eight Decades of Scurvy. The Case History of a Misleading Dietary Hypothesis. Orthomolecular Psychiatry 8(2):58-62. Retrieved April 4, 2007.
- Stone, I. 1972. The Healing Factor: Vitamin C Against Disease. New York:Grosset and Dunlap. ISBN 0-448-11693-6
- Stryer, L. 1995. Biochemistry, 4th edition. New York: W.H. Freeman. ISBN 0716720094
- World Health Organization. 2004. Vitamin and mineral requirements in human nutrition, 2nd ed. Retrieved February 20, 2007.
.
Further reading
- Pauling, Linus (1970). Vitamin C and the Common Cold. W. H. Freeman & Company. ISBN 071670160X.
- Pauling, Linus (1976). Vitamin C, the Common Cold, and the Flu. W H Freeman & Co. ISBN 0716703610.
- Cameron, Ewan and Linus Pauling, (1979). Cancer and Vitamin C. Pauling Institute of Science and Medicine. ISBN 0393500004.
- Kent, Saul (1980). Life Extension Revolution. Morrow.
- Pearson, Durk and Sandy Shaw (1982). Life Extension: A Practical Scientific Approach. Warner Books. ISBN 0446387355. see Part IV, Chapter 7: Vitamin C
- Pelton, Ross (1986). Mind Food and Smart Pills: How to Increase Your Intelligence and Prevent Brain Aging. T & R Pub. ISBN 0936809000. see Chapter 3: Vitamin C, The Champion Free Radical Scavenger
- Clemetson, C.A.B (1989). Vitamin C. Boca Raton, Florida: CRC Press. ISBN 0-8493-4841-2. Monograph - Volumes I, II, III.
- Levy, Thomas E. (2002). Vitamin C Infectious Diseases, & Toxins. Xlibris. ISBN 1401069630.
[11]<
External links
- Jane Higdon, "Vitamin C", Micronutrient Information Center, Linus Pauling Institute
- AscorbateWeb — a collection of twentieth century medical & scientific literature on vitamin C in the treatment and prevention of human disease at seanet.com
- Healing Thresholds — Research on Vitamin C in the treatment of autism. at healingthresholds.com
- U.S. Patent 5278189 (PDF) — "Prevention and treatment of occlusive cardiovascular disease with ascorbate and substances that inhibit the binding of lipoprotein (a)", Inventors: Matthias W. Rath and Linus C. Pauling
- vitamin C at United Kingdom Food Standards Agency
- Naidu KA (2003). Vitamin C in human health and disease is still a mystery? An overview. Nutrition journal 2: 7.
- For Doctors: Preparation of Vitamin C IV's — by Andrew W. Saul, PhD. at doctoryourself.com
- Information regarding treatment of the Bird Flu with massive doses of ascorbate. — by Robert Cathcart, M.D. at orthomed.com
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
- ↑ Nigeria: Vitamin C Can Suppress HIV/Aids Virus. allAfrica.com (2006-05-22). Retrieved 2006-06-16.
- ↑ Hemilä H (2003). Vitamin C and SARS coronavirus. J Antimicrob Chemother 52 (6): 1049-50.
- ↑ Boseley, Sarah. "Discredited doctor's 'cure' for Aids ignites life-and-death struggle in South Africa", The Guardian, 2005-05-14. Retrieved 2007-02-21.
- ↑ Rath, Matthias (2005). Open letter from Dr. Matthias Rath MD to German Chancellor Merkel. Dr. Rath Health Foundation. Retrieved 2007-02-21.
- ↑ Harakeh S, Jariwalla R, Pauling L (1990). Suppression of human immunodeficiency virus replication by ascorbate in chronically and acutely infected cells. Proc Natl Acad Sci U S A 87 (18): 7245-9.
- ↑ Harakeh S, Jariwalla R (1991). Comparative study of the anti-HIV activities of ascorbate and thiol-containing reducing agents in chronically HIV-infected cells. Am J Clin Nutr 54 (6 Suppl): 1231S-1235S.
- ↑ Harakeh S, Jariwalla R (1997). NF-kappa B-independent suppression of HIV expression by ascorbic acid. AIDS Res Hum Retroviruses 13 (3): 235-9.
- ↑ Harakeh S, Jariwalla R. Ascorbate effect on cytokine stimulation of HIV production. Nutrition 11 (5 Suppl): 684-7.
- ↑ Harwood H, Greene Y, Stacpoole P (1986). Inhibition of human leukocyte 3-hydroxy-3-methylglutaryl coenzyme A reductase activity by ascorbic acid. An effect mediated by the free radical monodehydroascorbate. J Biol Chem 261 (16): 7127-35.
- ↑ FDA OKs vitamin C trial for cancer. Physorg.com (January 12, 2007). Retrieved 2007-04-06.
- ↑ National Nutrient Database. Nutrient Data Laboratory of the US Agricultural Research Service. Retrieved 2007-03-07.