Uracil
| Uracil | |
|---|---|
| |
| General | |
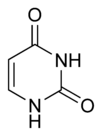
| Systematic name | Pyrimidine-2,4(1H,3H)-dione |
| Other names | Uracil, 2-oxy-4-oxy pyrimidine, 2,4(1H,3H)-pyrimidinedione, 2,4-dihydroxypryimidine, 2,4-pyrimidinediol |
| Molecular formula | C4H4N2O2 |
| Molar mass | 112.08676 g/mol |
| Appearance | Solid |
| CAS number | [66-22-8] |
| Properties | |
| Density and phase | |
| Solubility in water | Soluble. |
| Melting point | 335 °C (608 K) |
| Boiling point | N/A |
| Acidity (pKa) | basic pKa = -3.4, acidic pKa = 9.389. |
| Structure | |
| Molecular shape | pyrimidine |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | carcinogen & tetratogen with chronic exposure |
| NFPA 704 | |
| Flash point | non flammable |
| R/S statement | R |
| RTECS number | YQ8650000 |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other cations | |
| Related compounds | Thymine |
| Except where noted otherwise, data are given for materials in their standard state (at 25°C, 100 kPa) | |
Uracil is one of the five main nucleobases found in the nucleic acids DNA and RNA. The others are adenine, cytosine, guanine, and thymine. However, while the other four are found in DNA, Uracil is usually only found in RNA. Uracil (U), thymine (T), and cytosine (C) are pyrimidine derivatives, and guanine (G) and adenine (A) are purine derivatives.
In DNA, thymine and cytosine form hydrogen bonds with their complementary purine derivatives, adenine and cytosine, respectively. In RNA, uracil replaces thymine as the usual complement of adenine. Thus, thymine is usually seen only in DNA and uracil only in RNA. Methylation of uracil produces thymine, providing a mechanism for repair and protection of DNA and improvement of DNA replication. One of the common mutations of DNA involves two adjacent thymines.
Uracil is common and naturally occurring (Garrett and Grisham, 1997). Uracil was originally discovered in 1900 and it was isolated by hydrolysis of yeast nuclein that was found in bovine thymus and spleen, herring sperm, and wheat germ (Brown 1994).
Uracil is used in cells to carry out the synthesis of important enzymes and the biosynthesis of polysaccharides. Derivatives of uracil, developed through human creativity, include compounds that can be used as a pharmaceutical drug with anticancer properties, and those used in agriculture as a herbicide and pesticide.
Properties
As a pyrimidine nucleobase, uracil is a heterocyclic aromatic organic compound. Heterocyclic compounds are organic compounds (those containing carbon) that contain a ring structure containing atoms in addition to carbon, such as sulfur, oxygen, or nitrogen, as part of the ring. Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone.
Uracil is a planar, unsaturated compound that has the ability to absorb light (Horton 2002).
Found in RNA, it base pairs with adenine through hydrogen bonding and is replaced by thymine in DNA. Uracil can base pair with any of the bases depending on how the molecule arranges itself on the helix, but readily pairs with adenine because the methyl group is repelled into a fixed position. Uracil is the hydrogen bond acceptor and can form up to three hydrogen bonds. Uracil can also bind with a ribose sugar to form a ribonucleoside, uridine. When a phosphate attaches to uridine, uridine 5'-monophosphate is produced (Horton et al. 2002).
Uracil undergoes keto-enol tautomeric shifts because of its resonance structures due to the NH2 substitutents and OH substitutents. In organic chemistry, a substituent is an atom or group of atoms substituted in place of a hydrogen atom on the parent chain of a hydrocarbon. Any nuclear instability the molecule may have from the lack of formal aromaticity is compensated by the cyclic-amidic stability (Brown 1994). The keto tautomer is referred to as the lactam structure, while the enol tautomer is referred to as the lactim structure. These tautomeric forms are predominant at pH of 7. The lactam structure is the most common form of uracil.
Uracil also recycles itself to form nucleotides by undergoing a series of phophoribosyltransferase reactions (Garrett and Grisham, 1997). Degradation of uracil produces substrates, aspartate, carbon dioxide, and ammonia (Garrett and Grisham 1997).
- C4H4N2O2 → H3NCH2CH2COO- + NH4 + CO2
Oxidative degradation of uracil produces urea and maleic acid in the presence of H2O2]] and Fe2+ or in the presence of diatomic oxygen and Fe2+.
Uracil is a weak acid. The first site of ionization of uracil is not known (Zorbach 1973). The negative charge is placed on the oxygen anion and produces a pKa of less than or equal to 12. The basic pKa = -3.4, while the acidic pKa = 9.389. In the gas phase, uracil has four sites that are more acidic than water (Kurinovich and Lee, 2002).
Synthesis
There are many laboratory syntheses of uracil that can be performed.
The simplest of the methods of synthesis is to add water to cytosine to produce uracil and ammonia (Garrett and Grisham, 1997). The most common way to synthesize uracil is by the condensation of maleic acid with urea in fuming sulfuric acid (Brown 1994), which is also depicted below. Uracil can also be synthesized by a double decomposition of thiouracil in aqueous chloroacetic acid (Brown 1994).
- C4H5N3O + H2O → C4H4N2O2 + NH3
- C4H4O4 + CH4N2O → C4H4N2O2 + 2 H2O + CO
Photodehydrogenation of 5,6-diuracil, which is synthesized by beta-alanine reacting with urea, produces uracil (Chittenden and Schwartz 1976).
Reactions
Uracil readily undergoes regular reactions including oxidation, nitration, and alkylation. While in the presence of Phenol/Sodium hypochlorite (PhOH/NaOCl), uracil can be visualized in the blue region of UV light (Brown 1994). Uracil also has the capability to react with elemental halogens because of the presence of more than one strongly electron donating group (Brown 1994).
Uracil readily undergoes an addition to ribose sugars and phosphates to partake in synthesis and further reactions in the body. Uracil becomes Uridine-monophosphate (UMP), uridine-diphosphate (UDP), uridine-triphosphate (UTP), and uracil-diphosphate glucose (UDP-glucose). Each one of these molecules in synthesized in the body and has specific functions.
When uracil reactes with anhydrous hydrazine, a first order kinetic reaction occurs and the ring of uracil opens up (Kochetkov and Budovskii 1972). If the pH of the reaction increases to >10.5, the uracil anion forms making the reaction go much slower, the same slowing of the reaction occurs if the pH decreases because of the protonation of the hydrazine (Kochetkov and Budovskii 1972). The reactivity of uracil is unchanged even if the temperature changes (Kochetkov and Budovskii 1972).
Uses
Uracil can be used for as a pharmaceutical drug with anticancer properties. When elemental fluorine is reacted with uracil, 5-fluorouracil is produced. 5-Fluorouracil is an anticancer drug (antimetabolite) used to masquerade as uracil during the nucleic acid replication process (Garrett and Grisham 1997). The molecule also fools the enzymes that help in this process to incorporate this compound in the replication and not uracil, resulting in the biological polymer (cancer) failing to continue synthesizing (Garrett and Grisham).
Uracil is used in animals and plants to help carry out the synthesis of many enzymes necessary for cell function through bonding with riboses and phosphates (Garrett and Grisham). Uracil serves as an allosteric regulator and coenzyme for reactions in the human body and in plants (Brown 1998). UMP controls the activity of carbamoyl phosphate synthetase and aspartate transcarbamoylase in plants, while UDP and UTP requlate CPSase II activity in animals. UDP-glucose regulates the conversion of glucose to galactose in the liver and other tissues in the process of carbohydrate metabolism (Brown 1998).
Uracil is also involved in the biosynthesis of polysaccharides and the transportation of sugars containing aldehydes (Brown 1998).
Uracil can also increase the risk for cancer in cases where the body is extremely deficient in folate (Mashiyama et al. 2004). The deficiency in folate leads to increased ratio of deoxyuracilmonophosphates (dUMP) to deoxythyminemonophosphates (dTMP) and uracil misincorporation into DNA and eventually low production of DNA (Mashiyama 2004).
Uracil has a number of agricultural uses. It can be used to determine microbial contamination of tomatoes, since only after lactic acid bacteria have contaminated the fruit does uracil appear (Hidalgo et al. 2005). Uracil's derivatives, which contain a diazine ring, are used in pesticides (Pozharskii et al. 1997). More often, it is used as an anti-photosynthetic herbicide to destroy weeds among cotton, sugar beet, turnip, soya, pea, and sunflower crops, as well as in vineyards, berry plantations, and orchards (Pozharskii 1997).
ReferencesISBN links support NWE through referral fees
- Brown, D. J. Heterocyclic Compounds: The Pyrimidines, Vol 52. New York: Interscience, 1994.
- Brown, E. G. Ring Nitrogen and Key Biomolecules: The Biochemistry of N-Heterocycles. Boston: Lluwer Academic Publishers, 1998.
- Chittenden, G. J. F., and A. W. Schwartz. “Possible pathway for prebiotic uracil synthesis by photodehvdroention.” Nature 263(5575): 350-351, 1976.
- Garrett, R. H., and C. M. Grisham. Principals of Biochemistry with a Human Focus. United States: Brooks/Cole Thomson Learning, 1997.
- Hidalgo, A., C. Pompei, A. Galli, and S. Cazzola. “Uracil as an Index of Lactic Acid Bacteria Contamination of Tomato Products.” Journal of Agricultural and Food Chemistry 53(2): 349-355, 2005.
- Horton, R. H. et al. Principles of Biochemistry, 3rd ed. Upper Saddle River, NJ: Prentice Hall, 2002.
- Kochetkov, N. K., and E. I. Budovskii. Organic Chemistry of Nucleic Acids, Part B. New York: Plenum Press, 1972.
- Kurinovich, M. A., and J. K. Lee. “The Acidity of uracil and uracil analogs in the gas phase: Four surprisingly acidic sites and biological implications.” J. Am. Soc. Mass. Spectrom. 13: 985-995, 2002.
- Mashiyama, S. T., C. Courtemanche, I. Elson-Schwab, J. Crott, B. L. Lee, C. N. Ong, M. Fenech, and B. N. Ames. “Uracil in DNA, determined by an improved assay, is increased when deoxynucleosides are added to folate-deficient cultured human lymphocytes.” Anal Biochem. 330(1): 58-69, 2004.
- Pozharskii, A. F. et al. Heterocycles in Life and Society: An Introduction to Heterocyclic Chemistry and Biochemistry and the Role of Heterocycles in Science, Technology, Medicine, and Agriculture. New York: John Wiley and Sons, 1997.
- Zorbach, W. W. Synthetic Procedures in Nucleic Acid Chemistry: Physical and Physicochemical Aids in Determination of Structure. Vol 2. New York: Wiley-Interscience, 1973.
| Nucleic acids edit |
|---|
| Nucleobases: Adenine - Thymine - Uracil - Guanine - Cytosine - Purine - Pyrimidine |
| Nucleosides: Adenosine - Uridine - Guanosine - Cytidine - Deoxyadenosine - Thymidine - Deoxyguanosine - Deoxycytidine |
| Nucleotides: AMP - UMP - GMP - CMP - ADP - UDP - GDP - CDP - ATP - UTP - GTP - CTP - cAMP - cGMP |
| Deoxynucleotides: dAMP - dTMP - dUMP - dGMP - dCMP - dADP - dTDP - dUDP - dGDP - dCDP - dATP - dTTP - dUTP - dGTP - dCTP |
| Nucleic acids: DNA - RNA - LNA - PNA - mRNA - ncRNA - miRNA - rRNA - siRNA - tRNA - mtDNA - Oligonucleotide |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
