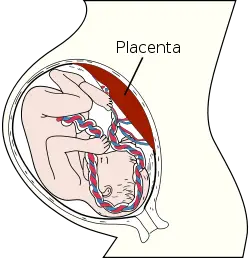
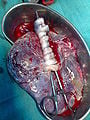
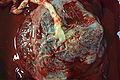
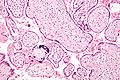
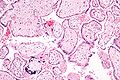
Placenta
| Placenta | ||
|---|---|---|
| Placenta | ||
| Precursor | decidua basalis, chorion frondosum | |
In zoology, the placenta is a temporary vascular organ that forms in the uterus of female placental mammals during pregnancy and, via an umbilical cord, connects the mother to the developing fetus. While this "true placenta" is a defining characteristic of eutherian or placental mammals, the term also is used in zoology to refer to organs with similar functions in certain nonmammalian animals, including various snakes, lizards, and sharks, up to Prototherial (egg-laying) and metatherial (marsupial) mammals. In botany, the term is used in reference to a part of a flowering plant ovary to which ovules are attached. This article will focus on the mammalian placenta.
The placenta facilitates important exchanges between the mother and fetus, including the transfer of nutrients and oxygen from the mother to the fetus and the removal of waste products from the fetus. In essence, the placenta acts as the lungs (oxygen/carbon dioxide exchange), digestive system (providing nutrients), and kidneys (removing waste products) for the fetus, until these systems can function on their own. In humans, the placenta is also a source of production of hormones important for pregnancy and allows antibodies to pass through to confer protection to the fetus. After birth of the fetus and fulfillment of its function, the placenta is expelled.
The classical plural is placentae, but the form placentas is common in modern English and probably has the wider currency at present.
Structure
Structurally and functionally, the placenta of diverse placental (eutherian) mammals share many common attributes. However, the different taxonomic groups also exhibit a number of pronounced differences in terms of the placenta’s macroscopic shape, the area of contact between maternal and fetal tissue, the number of layers of tissue between the maternal and fetal vascular systems, and the development of the villa structure. Classifications have been developed based on these differences (Bowen 2011; Benirschke 2007).
For example, prior to the placenta’s formation, there are six layers of tissue separating the fetal and maternal blood, with three layers of fetal extraembryonic membranes and three on the maternal side (the endometrial epithelial cells, connective tissue of the endometrium, and endometrial epithelial cells). Different taxa retain different maternal tissues during placentation. In humans, all three layers of the maternal tissue erode and the fetal chorionic epithelium is bathed in maternal blood. This is classified as hemochorial. Dogs and cats retain the maternal uterine endothelium, such that the maternal tissue is in contact with the chorionic epithelium of the fetus, and this is known as endotheliochorial. Horses, swine, and ruminants retain all three maternal layers and are classified as epitheliochorial (Bowen 2011).
In terms of macroscopic shape, a human placenta, as well as primates in general and rodents, is classified as discoid (discoid in shape, single placenta formed). The macroscopic shape of pigs and horses is classified as diffuse (the formation of the placenta involves almost the entire surface of the alantochorion), while ruminants are classified as cotyledonary (multiple, discrete areas of attachment). Dogs, cats, bears, whales, and elephants have placentas that are classified as zonary (the shape of an incomplete or complete band of tissue surrounding the fetus) (Bowen 2011; Benirschke 2007).
The development of the villi structure may take the form of lamellar (carnivores), folded (pig), labyrinthine (rodents, lagomorphs), trabecular (some primates) and villous (human) (Benirschke 2007)
Thus humans are classified as discoid, hemochorial, and villous, while rodents are classified as discoid, hemochorial, and labyrinthine, and pigs are classified as diffuse, epitheliochorial, and folded. Carnivores are classified as zonary, endotheliochorial, and lamellar.
In humans, the placenta averages 22 cm (9 inch) in length and 2–2.5 cm (0.8–1 inch) in thickness (greatest thickness at the center and become thinner peripherally). It typically weighs approximately 500 grams (1 lb). It has a dark reddish-blue or crimson color. It connects to the fetus by an umbilical cord of approximately 55–60 cm (22–24 inch) in length that contains two arteries and one vein (Yetter 1998).
Human Placental Development and Circulation
Development
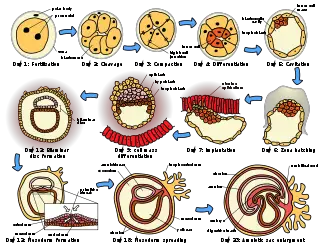
The placenta begins to develop upon implantation of the blastocyst into the maternal endometrium. The outer layer of the blastocyst becomes the trophoblast, which forms the outer layer of the placenta. This outer layer is divided into two further layers: the underlying cytotrophoblast layer and the overlying syncytiotrophoblast layer. The syncytiotrophoblast is a multinucleated continuous cell layer that covers the surface of the placenta. It forms as a result of differentiation and fusion of the underlying cytotrophoblast cells, a process that continues throughout placental development. The syncytiotrophoblast (otherwise known as syncytium), thereby contributes to the barrier function of the placenta.
The placenta grows throughout pregnancy. Development of the maternal blood supply to the placenta is complete by the end of the first trimester of pregnancy (approximately 12–13 weeks).
As the womb grows, the placental also moves, commonly starting low in the womb in early pregnancy but eventually moving to the top of the womb. In order for the cervix to be open for delivery, the placenta should be near the top of the womb by the third trimester. When the placenta is cervix is obstructed by the placenta, it is a condition known as previa. (Vorvick and Storck 2011).
Circulation
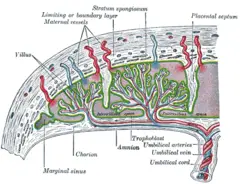
Maternal placental circulation
In preparation for implantation, the uterine endometrium undergoes "decidualization." Spiral arteries in decidua are remodeled so that they become less convoluted and their diameter is increased. The increased diameter and straighter flow path both act to increase maternal blood flow to the placenta. The relatively high pressure as the maternal blood fills intervillous space through these spiral arteries bathes the fetal villi in blood, allowing an exchange of gases to take place. In humans and other "hemochorial placentals," (see structure above) the maternal blood comes into direct contact with the fetal chorion, though no fluid is exchanged. As the pressure decreases between pulses, the deoxygenated blood flows back through the endometrial veins.
Maternal blood flow in humans is approx 600–700 ml/min at term.
Fetoplacental circulation
Deoxygenated fetal blood passes through umbilical arteries to the placenta. At the junction of umbilical cord and placenta, the umbilical arteries branch radially to form chorionic arteries. Chorionic arteries, in turn, branch into cotyledon arteries. In the villi, these vessels eventually branch to form an extensive arterio-capillary-venous system, bringing the fetal blood extremely close to the maternal blood; but no intermingling of fetal and maternal blood occurs ("placental barrier") (Schöni-Affolter et al. 2008).
Endothelin and prostanoids cause vasoconstriction in placental arteries, while nitric oxide vasodilation. On the other hand, there is no neural vascular regulation, and catecholamines have only little effect (Kiserud and Acharya 2004).
Functions
The placenta allows exchange between the developing fetus and the mother as a result of the thin tissue separating the fetal blood from the maternal blood, while at the same time not allowing the bloodstreams to intermix. This allows nutrients, oxygen, antibodies, and other constituents of the mother's blood to support the development of the fetus and for wastes (carbon dioxide, nitrogenous wastes) to diffuse out of the fetal blood into the mother's circulatory system for removal. The fetal blood flows via the umbilical cord to and from the placenta.
Nutrition
The perfusion of the intervillous spaces of the placenta with maternal blood allows the transfer of nutrients and oxygen from the mother to the fetus and the transfer of waste products and carbon dioxide back from the fetus to the maternal blood supply. Nutrient transfer to the fetus occurs via both active and passive transport. Active transport systems allow significantly different plasma concentrations of various large molecules to be maintained on the maternal and fetal sides of the placental barrier (Wright and Sibley 2011).
Adverse pregnancy situations, such as those involving maternal diabetes or obesity, can increase or decrease levels of nutrient transporters in the placenta resulting in overgrowth or restricted growth of the fetus.
Excretion
Waste products excreted from the fetus such as urea, uric acid, and creatinine are transferred to the maternal blood by diffusion across the placenta.
Immunity
IgG antibodies can pass through the human placenta, thereby providing protection to the fetus in utero (Simister and Story, 1997).
Furthermore, the placenta functions as a selective maternal-fetal barrier against transmission of microbes to the fetus. However, insufficiency in this function may still cause mother-to-child transmission of infectious diseases.
Endocrine function
In humans, aside from serving as the conduit for oxygen and nutrients for fetus, the placenta secretes hormones (secreted by syncytial layer/syncytiotrophoblast of chorionic villi) that are important during pregnancy.
Hormones:
Human Chorionic Gonadotropin (hCG): The first placental hormone produced is hCG, which can be found in maternal blood and urine as early as the first missed menstrual period (shortly after implantation has occurred) through about the 100th day of pregnancy. This is the hormone analyzed by pregnancy test; a false-negative result from a pregnancy test may be obtained before or after this period. Women's blood serum will be completely negative for hCG by one to two weeks after birth. hCG testing is proof that all placental tissue is delivered. hCG is present only during pregnancy because it is secreted by the placenta, which is present only during pregnancy (Pillitteri, 2010). hCG also ensures that the corpus luteum continues to secrete progesterone and estrogen. Progesterone is very important during pregnancy because, when its secretion decreases, the endometrial lining will slough off and pregnancy will be lost. hCG suppresses the maternal immunologic response so that the placenta is not rejected.
Human Placental Lactogen (hPL [Human Chorionic Somatomammotropin]): This hormone is lactogenic and has growth-promoting properties. It promotes mammary gland growth in preparation for lactation in the mother. It also regulates maternal glucose, protein, and fat levels so that this is always available to the fetus.
Estrogen is referred to as the "hormone of women" because it stimulates the development of secondary female sex characteristics. It contributes to the woman's mammary gland development in preparation for lactation and stimulates uterine growth to accommodate growing fetus.
Progesterone is necessary to maintain endometrial lining of the uterus during pregnancy. This hormone prevents preterm labor by reducing myometrial contraction. Levels of progesterone are high during pregnancy.
Cloaking from immune system of mother
The placenta and fetus may be regarded as a foreign allograft inside the mother, and thus must evade from being attacked by the mother's immune system.
For this purpose, the placenta uses several mechanisms:
- It secretes Neurokinin B-containing phosphocholine molecules. This is the same mechanism used by parasitic nematodes to avoid detection by the immune system of their host (BBC 2007).
- There is the presence of small lymphocytic suppressor cells in the fetus that inhibit maternal cytotoxic T cells by inhibiting the response to interleukin 2 (Clark et al. 1986).
However, the placental barrier is not the sole means to evade the immune system, as foreign foetal cells also persist in the maternal circulation, on the other side of the placental barrier (Williams et al. 2008).
Other functions
The placenta also provides a reservoir of blood for the fetus, delivering blood to it in case of hypotension and vice versa, comparable to a capacitor (Assad et al. 2001).
Birth
Placental expulsion begins as a physiological separation from the wall of the uterus. The period from just after the fetus is expelled until just after the placenta is expelled is called the third stage of labor. The placenta is usually expelled within 15–30 minutes of the baby's being born.
Placental expulsion can be managed actively, for example by giving oxytocin via intramuscular injection followed by cord traction to assist in delivering the placenta. As an alternative, it can be managed expectantly, allowing the placenta to be expelled without medical assistance.
The habit is to cut the cord immediately after the baby is born, but it has been stated that there is no medical reason to do so immediately and that, on the contrary, it seems that not cutting the cord helps the baby in his adaptation to extra uterine life, especially in preterm infants (Mercier and Vohr, 2010).
Pathology
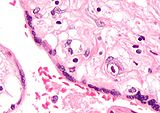
Numerous pathologies can affect the placenta:
- Placenta accreta
- Placenta praevia
- Placental abruption/abruptio placentae
- Placental insufficiency
Infections involving the placenta:
- Placentitis, such as the TORCH infections.
- Chorioamnionitis.
Placenta accretia is the condition when the placenta implants too deeply, into actual muscle of uterine wall.
Placenta abruptio, or placenta abruption, is a condition where the placenta separates from the uterine wall before the baby is delivered (Vorvick and Storck 2010a).
Placenta previa is the condition where the placenta stays in the lowest part of the uterus and covers part or all of the opening to the cervix, which is the opening to the birth canal. Normally, the placenta is low in early pregnancy, but moves to the top of the womb as pregnancy develops. When it stays low in the womb, it does not allow the cervix to be open for delivery (Vorvick and Storck 2011).
Placental insufficiency is a condition where the placental is unable to bring enough oxygen and nutrients to the fetus. Conditions that can lead to this include diabetes, high blood pressure, and smoking (Vorvick and Storck 2010b).
Cultural practices and beliefs
The placenta often plays an important role in various cultures, with many societies conducting rituals regarding its disposal (Deelah).
In the Western world, the placenta is most often incinerated (BBC 2006). However, some cultures bury the placenta. The Māori of New Zealand traditionally bury the placenta from a newborn child to emphasize the relationship between humans and the earth (Metge 2005) Likewise, the Navajo bury the placenta and umbilical cord at a specially chosen site, particularly if the baby dies during birth (Francisco 2004; Shepardson 1978). In Cambodia and Costa Rica, burial of the placenta is believed to protect and ensure the health of the baby and the mother (Buckley 2006). If a mother dies in childbirth, the Aymara of Bolivia bury the placenta in a secret place so that the mother's spirit will not return to claim her baby's life (Davenport, A. 2005).
The placenta is believed by some communities to have power over the lives of the baby or its parents. The Kwakiutl of British Columbia bury girls' placentas to give the girl skill in digging clams, and expose boys' placentas to ravens to encourage future prophetic visions. In Turkey, the proper disposal of the placenta and umbilical cord is believed to promote devoutness in the child later in life. In Ukraine, Transylvania, and Japan, interaction with a disposed placenta is thought to influence the parents' future fertility.
Several cultures believe the placenta to be or have been alive, often a relative of the baby. Nepalese think of the placenta as a friend of the baby; Malaysian Orang Asli regard it as the baby's older sibling. The Ibo of Nigeria consider the placenta the deceased twin of the baby, and conduct full funeral rites for it (Buckley 2006). Native Hawaiians believe that the placenta is a part of the baby, and traditionally plant it with a tree that can then grow alongside the child (BBC 2006). Various cultures in Indonesia, such as Javanese, believe that the placenta has a spirit and needs to be buried outside the family house.
In some cultures, the placenta is eaten, a practice known as placentophagy. In some eastern cultures, such as China and Hong Kong, the dried placenta (紫河車) is thought to be a healthful restorative and is sometimes used in preparations of traditional Chinese medicine and various health products.
Placenta in non-placental animals
A "true placenta" is a defining characteristic of eutherian or placental mammals. However, the term has also been applied to various nonmammlian animals (snakes, lizards, and sharks) as well as egg-laying and marsupial mammals.
Note, however, that the homology of such structures in various viviparous organisms is debatable at best and, in invertebrates such as Arthropoda, is definitely analogous at best. However, a 2012 publication describes what amounts to a phylogenetically analogous, but physiologically and functionally almost identical structure in a skink. In some senses, it is not particularly surprising, because many species are ovoviviparous and some are known as examples of various degrees of viviparous matrotrophy. However, the latest example is the most extreme to date, of a purely reptilian placenta directly comparable to a eutherian placenta (Blackburn and Flemming 2012).
Prototherial (egg-laying) and metatherial (marsupial) mammals produce a choriovitelline placenta that, while connected to the uterine wall, provides nutrients mainly derived from the egg sac.
Additional images
Micrograph of a placental infection (CMV placentitis).
See also
- Embryo
- Childbirth
- Uterus
ReferencesISBN links support NWE through referral fees
- Assad, R. S., F. Y. Lee, and F. L. Hanley. 2001. Placental compliance during fetal extracorporeal circulation. Journal of applied physiology 90(5): 1882–1886. PMID 11299282.
- BBC. 2007. Placenta "fools body's defences." BBC News November 10, 2007. Retrieved June 23, 2012.
- BBC. 2006. Why eat a placenta? BBC News April 18, 2006. Retrieved June 23, 2012.
- Benirschke, K. 2007. ‘’Comparative Placentation.’’ San Diego, CA: University of California at San Diego.
- Blackburn, D. G., and A. F. Flemming. 2012. Invasive implantation and intimate placental associations in a placentotrophic african lizard, Trachylepis ivensi (scincidae). Journal of Morphology 273(2): 137-59. Retrieved June 23, 2012.
- Brown, T. L. 2008. Placenta. Pages 2170-2171 in Magill's Medical Guide, 4th Edition, Vol. IV Pasadena, CA: Salem Press. ISBN 9781587653841.
- Buckley, S. J. 2006. Placenta rituals and folklore from around the world. Midwifery Today Int Midwife 80:58-9. PMID 17265840. Retrieved June 23, 2012. (Also in Mothering, which is the archived article in the link.)
- Clark, D. A., A. Chaput, and D. Tutton. 1986. Active suppression of host-vs-graft reaction in pregnant mice. VII. Spontaneous abortion of allogeneic CBA/J x DBA/2 fetuses in the uterus of CBA/J mice correlates with deficient non-T suppressor cell activity. J. Immunol. 136(5): 1668–75. PMID 2936806.
- Davenport, A. 2005. The love offer. Johns Hopkins Magazine June, 2005. Retrieved June 23, 2012.
- Deelah, C. n.d. Afterbirth, After birth. Naturallyborn.net.
- Kiserud, T., and G. Acharya. 2004. The fetal circulation. Prenatal Diagnosis 24(13): 1049–1059. PMID 15614842.
- Mercier, J. S., and B. R. Vohr. 2010. Seven-month developmental outcomes of very low bith weight infants enrolled in a randomized controlled trial of delayed versus immediate cord clamping. Journal of Perinatology 30(1):1.
- Metge, J. 2005. Working in/playing with three languages: English, Te Reo Maori, and Maori Bod Language. Sites 2(2):83-90.
- Pillitteri, A. 2010. Maternal and Child Health Nursing, 6th Edition. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. ISBN 9781582559995.
- Schöni-Affolter, F., C. Dubuis-Grieder, and E. Strauch. 2008. Placental blood circulation. Chapter 10.3 in Human Embryology. Swiss Virtual Campus.
- Shepardson, M. 1978. Changes in Navajo mortuary practices and beliefs. American Indian Quarterly 6:170-190.
- Simister, N. E., and C. M. Story. 1997. Human placental Fc receptors and the transmission of antibodies from mother to fetus. Journal of Reproductive Immunology 37: 1-23.
- Vorvick, L., and S. Storck. 2010a. Placenta abruptio. MedlinePlus November 21, 2010. Retrieved June 13, 2012.
- Vorvick, L., and S. Storck. 2010b. Placenta insufficiency. MedlinePlus June 5, 2010. Retrieved June 13, 2012.
- Vorvick, L., and S. Storck. 2011. Placenta previa. MedlinePlus September 12, 2011. Retrieved June 13, 2012.
- Williams, Z., D. Zepf, J. Longtine, et al. 2008. Foreign fetal cells persist in the maternal circulation. Fertil. Steril. 91(6): 2593–5. PMID 18384774.
- Wright, C., and C. P. Sibley. 2011. Placental transfer in health and disease. In H. Kay, M. Nelson, and Y. Wang, The Placenta: From Development to Disease. John Wiley and Sons. ISBN 9781444333664.
- Yetter, J. F. 1998. Examination of the placenta. Am Fam Physician 57(5):1045-1054. Retrieved June 21, 2011.
| Anatomy |
|---|
| Major Systems |
| Circulatory system . Digestive system . Endocrine system . Excretory system . Immune system . Integumentary system . Lymphatic system . Muscular system . Nervous system . Reproductive system . Respiratory system . Skeletal system |
| Organs |
| . Adrenal Gland . Anus . Appendix . Brain . Breast . Colon or large intestine . Diaphragm . Ear . Eye . Heart . Kidney . Larynx . Liver . Lung . Nose . Ovary . Pharynx . Pancreas . Penis . Placenta . Prostate Gland . Rectum . Skin . Small intestine . Seminal Vesicles . Spleen . Stomach . Testis . Thyroid Gland . Tongue . Uterus . Vulva |
| Bones |
| . Collar bone (clavicle) . Thigh bone (femur) . Humerus . Mandible . Patella . Radius . Skull (cranium) . Tibia . Ulna . Rib (costa) . Vertebrae . Pelvis . Sternum |
| Glands |
| Ductless gland . Mammary gland . Salivary gland . Thyroid gland . Parathyroid gland . Adrenal gland . Pituitary gland . Pineal gland |
| Tissues |
| Connective tissue . Endothelial tissue . Epithelial tissue . Glandular tissue . Lymphoid tissue |
| Body Parts |
| . Abdomen . Arm . Back . Buttock . Chest . Ear . Eye . Face . Genitals . Head . Joint . Leg . Mouth . Neck . Scalp . Skin . Teeth . Tongue |
| Other Terms |
| Artery . Coelom . Diaphragm . Gastrointestinal tract . Hair . Exoskeleton . Lip . Nerve . Peritoneum . Serous membrane . Skeleton . Skull . Spinal cord . Vein |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.