| |
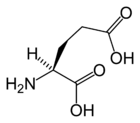
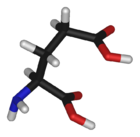
Glutamic acid | |
| Systematic (IUPAC) name | |
| (2S)-2-aminopentanedioic acid | |
| Identifiers | |
| CAS number | 56-86-0 |
| PubChem | 611 |
| Chemical data | |
| Formula | C5H9NO4 |
| Mol. weight | 147.13 |
| SMILES | N[C@@H](CCC(O)=O)C(O)=O |
| Complete data | |
Glutamic acid, also referred to as glutamate (the salt of glutamic acid), is an acidic, α-amino acid that is found in many proteins, and that in free form functions as an important neurotransmitter, a metabolic intermediate in the Krebs cycle, and a compound that can combine with an help in the elimination of toxic ammonia from the body. The salt monosodium glutamate (MSG) is a common food additive and flavor enhancer. Along with aspartic acid, to which is behaves similarly, glutamic acid is classified as an acidic amino acid.
The L-isomer, which is the only form that is involved in protein synthesis, is one of the 20 standard amino acids common in animal proteins and required for normal functioning in humans. However, it is not considered to be an "essential" amino acid since it does not have to be taken in with the diet, but can be synthesized by the human body from other compounds through chemical reactions.
Glutamic acid is responsible for one of the human senses of taste, termed umami, adding to the classical taste sensations of sweet, salty, sour, and bitter. Umami applies to the sensation of savoriness, the detection of glutamates in such foods as meats, cheese, and other protein-heavy foods. Beyond its practical value, taste adds to the human enjoyment of creation, joining such diverse senses as being able to see various colors, hear different sounds, smell a vast array of odors, and so forth. Such senses allow interaction with nature and touch upon the inner aspect of people. As a source for umami, MSG, the sodium salt of glutamic acid, is used to enhance the flavor of foods.
Glutamic acid's three letter code is Glu, its one letter code is E, and its systematic name is 2-Aminopentanedioic acid(IUPAC-IUB 1983). A three-letter designation for either glutamic acid (Glu) or the amino acid glutamine (Gln) is Glx—this is often used in cases in which peptide sequencing reactions may convert glutamine to glutamate (or vice versa), leaving the original identity of the amino acid in doubt.
Structure
In biochemistry, the term amino acid is frequently used to refer specifically to alpha amino acids: those amino acids in which the amino and carboxylate groups are attached to the same carbon, the so-called α–carbon (alpha carbon). The general structure of these alpha amino acids is:
R
|
H2N-C-COOH
|
H
where R represents a side chain specific to each amino acid.
Most amino acids occur in two possible optical isomers, called D and L. The L amino acids represent the vast majority of amino acids found in proteins. They are called proteinogenic amino acids. As the name "proteinogenic" (literally, protein building) suggests, these amino acid are encoded by the standard genetic code and participate in the process of protein synthesis. In glutamic acid, only the L-stereoisomer is involved in protein synthesis in mammals.
Glutamic acid's chemical formula is HOOC-CH(NH2)-(CH2)2-COOH (very similar to aspartic acid's formula, HOOC-CH(NH2)-CH2-COOH), but with an extra CH2), or more generally C5H9NO4. (Aspartic acid's general forumula is C4H7NO4.)
Glutamic acid behaves similar to aspartic acid, but has a longer, slightly more flexible side chain. As its name indicates, it is acidic, with a carboxylic acid component to its side chain. Generally either the amino group will be protonated or one or both of the carboxylic groups will be deprotonated. At neutral pH all three groups are ionized and the species has a charge of -1. The pKa value for Glutamic acid is 4.1. This means that at pH below this value it will be protonated (COOH) and at pH above this value it will be deprotonated (COO-).
Synthesis
Various methods of biosynthesis are possible.
| Reactants | Products | Enzymes |
|---|---|---|
| Glutamine + H2O | → Glu + NH3 | GLS, GLS2 |
| NAcGlu + H2O | → Glu + Acetate | (unknown) |
| α-ketoglutarate + NADPH + NH4+ | → Glu + NADP+ + H2O | GLUD1, GLUD2 |
| α-ketoglutarate + α-amino acid | → Glu + α-oxo acid | transaminase |
| 1-pyrroline-5-carboxylate + NAD+ + H2O | → Glu + NADH | ALDH4A1 |
| N-formimino-L-glutamate + FH4 | ⇌ Glu + 5-formimino-FH4 | FTCD |
Function
In addition to its role in making up a substantial portion of proteins, glutamic acid is also important in metabolism, as a transmitter, for removal of nitrogen from the body, for medical and biological research, as a commercial product for taste, and so forth.
In metabolism
Glutamate is a key molecule in cellular metabolism. In humans, dietary proteins are broken down by digestion into amino acids, which serves as metabolic fuel for other functional roles in the body. A key process in amino acid degradation is transamination, in which the amino group of an amino acid is transferred to an α-ketoacid, typically catalysed by a transaminase (enzyme). The reaction can be generalized as such:
- R1-amino acid + R2-α-ketoacid ⇌ R1-α-ketoacid + R2-amino acid
A very common α-ketoacid is α-ketoglutarate, an intermediate in the citric acid cycle. When α-ketoglutarate undergoes transamination, it always results in glutamate being formed as the corresponding amino acid product. The resulting α-ketoacid product is often a useful one as well, which can contribute as fuel or as a substrate for further metabolism processes. Examples are as follows:
- aspartate + α-ketoglutarate ⇌ oxaloacetate + glutamate
Both pyruvate and oxaloacetate are key components of cellular metabolism, contributing as substrates or intermediates in fundamental processes such as glycolysis and gluconeogenesis (generation of glucose from non-sugar carbon substrates like pyruvate, lactate, glycerol, and such glucogenic amino acids as alanine and glutamine), as well as the citric acid cycle.
Removal of ammonia
Glutamatic acid plays an important role in the body's disposal of excess or waste nitrogen in the form of ammonia.
Glutamate undergoes deamination, an oxidative reaction catalysed by glutamate dehydrogenase, as follows:
- glutamate + water + NAD+ → α-ketoglutarate + NADH + ammonia + H+
Ammonia is a metabolic product of amino acid deamination (removal of an amine group) and is toxic to the body. In humans, ammonia is quickly converted to urea, which is much less toxic. Essentially, glutuamic acid coupled with ammonia yields glutamine, which is transported to the liver, where it is converted to urea for excretion. Ammonia (as ammonium) is then excreted predominantly as urea. This urea is a major component of the dry weight of urine.
Transamination can thus be linked to deamination, effectively allowing nitrogen from the amine groups of amino acids to be removed, via glutamate as an intermediate, and finally excreted from the body in the form of urea.
As a neurotransmitter
The salt glutamate is the most abundant fast excitatory neurotransmitter in the mammalian nervous system, transmitting nerve impulses over synapses (gaps) between nerve cells (neurons) and other cells. At chemical synapses, glutamate is stored in vesicles. Nerve impulses trigger release of glutamate from the pre-synaptic cell. In the opposing post-synaptic cell, the receptor cell, on the other side of the synapse, glutamate receptors, such as the NMDA receptor, bind glutamate and are activated. Glutamate is active in the central nervous system (brain and spinal cord). Because of its role in synaptic plasticity, it is believed that glutamic acid is involved in cognitive functions like learning and memory in the brain.
Aspartate (the conjugate base of aspartic acid) also acts as a neurotransmitter and stimulates NMDA receptors, though not as strongly as glutamate does (Chen et al. 2005).
Glutamate transporters are found in neuronal and glial membranes (Shigeri et al. 2004). They rapidly remove glutamate from the extracellular space. In brain injury or disease, they can work in reverse and excess glutamate can accumulate outside cells. This process causes calcium ions to enter cells via NMDA receptor channels, leading to neuronal damage and eventual cell death and is called excitotoxicity. The mechanisms of cell death include:
- Damage to mitochondria from excessively high intracellular Ca2+ (Manev et al. 1989).
- Glu/Ca2+-mediated promotion of transcription factors for pro-apoptotic genes, or downregulation of transcription factors for anti-apoptotic genes.
Excitotoxicity due to glutamate occurs as part of the ischemic cascade and is associated with stroke and diseases like amyotrophic lateral sclerosis, lathyrism, and Alzheimer's disease.
Glutamic acid has been implicated in epileptic seizures. Microinjection of glutamic acid into neurons produces spontaneous depolarisations around one second apart, and this firing pattern is similar to what is known as paroxysmal depolarizing shift in epileptic attacks. This change in the resting membrane potential at seizure foci could cause spontaneous opening of voltage activated calcium channels, leading to glutamic acid release and further depolarization.
GABA precursor
Glu also serves as the precursor for the synthesis of the inhibitory gamma-aminobutyric acid (GABA) in GABA-ergic neurons. (GABA is an inhibitory neurotransmitter found in the nervous systems of widely divergent species.) This reaction is catalyzed by GAD, glutamic acid decarboxylase, which is most abundant in cerebellum and pancreas.
Stiff-man syndrome is a neurologic disorder caused by anti-GAD antibodies, leading to a decrease in GABA synthesis and therefore, impaired motor function such as muscle stiffness and spasm. Since the pancreas is also abundant for the enzyme GAD, a direct immunological destruction occurs in the pancreas and the patients will have diabetes mellitus.
Medical and biochemical research
Experimental techniques to detect glutamate in intact cells include using a genetically-engineered nanosensor (Okumoto et al. 2005). The sensor is a fusion of a glutamate-binding protein and two fluorescent proteins. When glutamate binds, the fluorescence of the sensor under ultraviolet light changes by resonance between the two fluorophores. Introduction of the nanosensor into cells enables optical detection of the glutamate concentration. Synthetic analogs of glutamic acid that can be activated by ultraviolet light have also been described (Corrie et al. 1993). This method of rapidly uncaging by photostimulation is useful for mapping the connections between neurons, and understanding synapse function.
Extracellular glutamate in Drosophila brains has been found to regulate postsynaptic glutamate receptor clustering, via a process involving receptor desensitization (Augustin et al. 2007). A gene expressed in glial cells actively transports glutamate into the extracellular space (Augustin et al. 2007), while in the nucleus accumbens, stimulating group II metabotropic glutamate receptors was found to reduce extracellular glutamate levels (Zheng 2002). This raises the possibility that this extracellular glutamate plays an "endocrine-like" role as part of a larger homeostatic system.
Taste and diet
Glutamic acid is present in a wide variety of foods and is responsible for one of the human senses of taste, umami, especially in its physiological form, the sodium salt of glutamate in a neutral pH. Classical taste sensations include sweet, salty, sour, and bitter. However, more recently, psychophysicists and neuroscientists have suggested other taste categories (umami and fatty acid taste, most prominently.) Umami applies to the sensation of savoriness—specifically, to the detection of glutamates, which are especially common in meats, cheese and other protein-heavy foods.
As a source for umami, the sodium salt of glutamic acid, monosodium glutamate (MSG) is used as a food additive to enhance the flavor of foods, although an identical effect can be achieved by mixing and cooking together different ingredients rich in this amino acid and other umami substances as well.
Another source of MSG is fruits, vegetables and nuts that have been sprayed with Auxigro. Auxigro is a growth enhancer that contains 30% glutamic acid.
China-based Fufeng Group Limited is the largest producer of glutamic acid in the world, with capacity increased to 300,000 tons at the end of 2006, putting them at 25-30 percent of the Chinese market. Meihua is the second largest Chinese producer. Together, the top five producers have roughly 50 percent share in China. Chinese demand is roughly 1.1 million tons per year, while global demand, including China, is 1.7 million tons per year.
Ninety-five percent of the dietary glutamate is metabolized by intestinal cells in a first pass (Reeds et al. 2000). Overall, glutamic acid is the single largest contributor to intestinal energy.
Pharmacology
Glutamate transport and supply are obvious targets for the treatment of epilepsy. In particular Glutamate Restriction Diets are now claiming success anecdotally, by limiting or eliminating intake of wheat, peanut, soy, and bean. No similar diets for schizophrenia are known.
The dissociative drug phencyclidine (more commonly known as PCP), which exhibiting hallucinogenic and neurotoxic effects, antagonizes glutamic acid non-competitively at the NMDA receptor. For the same reasons, sub-anaesthetic doses of Ketamine have strong dissociative and hallucinogenic effects.
ReferencesISBN links support NWE through referral fees
- Augustin, H., Y. Grosjean, K. Chen, Q. Sheng, and D. E. Featherstone. 2007. Nonvesicular release of glutamate by glial xCT transporters suppresses glutamate receptor clustering in vivo. Journal of Neuroscience 27(1): 111-123. PMID 17202478.
- Chen, P. E., M. T. Geballe, P. J. Stansfeld, A. R. Johnston, H. Yuan, A. L. Jacob, J. P. Snyder, S. F. Traynelis, and D. J. A. Wyllie. 2005. Structural features of the glutamate binding site in recombinant NR1/NR2A N-Methyl-D-aspartate receptors determined by site-directed mutagenesis and molecular modeling. Molecular Pharmacology 67: 1470-1484. Retrieved June 25, 2007.
- Corrie, J. E. et al. 1993. Postsynaptic activation at the squid giant synapse by photolytic release of L-glutamate from a 'caged' L-glutamate. Journal of Physiology 465: 1-8. PMID 7901400. Retrieved June 26, 2007.
- Doolittle, R. F. 1989. Redundancies in protein sequences. In G. D. Fasman, ed., Prediction of Protein Structures and the Principles of Protein Conformation. New York: Plenum Press. ISBN 0306431319.
- International Union of Pure and Applied Chemistry and International Union of Biochemistry and Molecular Biology (IUPAC-IUB) Joint Commission on Biochemical Nomenclature. 1983. Nomenclature and symbolism for amino acids and peptides: Recommendations on organic & biochemical nomenclature, symbols & terminology. IUPAC-IUB. Retrieved June 14, 2007.
- Lehninger, A. L., D. L. Nelson, and M. M. Cox. 2000. Lehninger Principles of Biochemistry, 3rd ed. New York: Worth Publishing. ISBN 1572591536.
- Manev, H., M. Favaron, A. Guidotti, and E. Costa. 1989. Delayed increase of Ca2+ influx elicited by glutamate: Role in neuronal death. Mol Pharmacol. 36(1): 106-112. PMID 2568579.
- Okumoto, S., et al. 2005. Detection of glutamate release from neurons by genetically encoded surface-displayed FRET nanosensors. Proceedings of the National Academy of Sciences U.S.A 102(24): 8740-8745. PMID 15939876.
- Reeds, P. J. et al. 2000. Intestinal glutamate metabolism. Journal of Nutrition 130(4s): 978S-982S. PMID 10736365.
- Shigeri, Y., R. P. Seal, K. Shimamoto. 2004. Molecular pharmacology of glutamate transporters, EAATs and VGLUTs. Brain Res Brain Res Rev. 45(3): 250-265. PMID 15210307.
- Zheng, Xi, D. A. Baker, H. Shen, D. S. Carson, and P. W. Kalivas. 2002. Group II metabotropic glutamate receptors modulate extracellular glutamate in the nucleus accumbens. Journal of Pharmacology and Experimental Therapeutics 300(1): 162-171. PMID 11752112.
| Major families of biochemicals | ||
| Peptides | Amino acids | Nucleic acids | Carbohydrates | Nucleotide sugars | Lipids | Terpenes | Carotenoids | Tetrapyrroles | Enzyme cofactors | Steroids | Flavonoids | Alkaloids | Polyketides | Glycosides | ||
| Analogues of nucleic acids: | The 20 Common Amino Acids | Analogues of nucleic acids: |
| Alanine (dp) | Arginine (dp) | Asparagine (dp) | Aspartic acid (dp) | Cysteine (dp) | Glutamic acid (dp) | Glutamine (dp) | Glycine (dp) | Histidine (dp) | Isoleucine (dp) | Leucine (dp) | Lysine (dp) | Methionine (dp) | Phenylalanine (dp) | Proline (dp) | Serine (dp) | Threonine (dp) | Tryptophan (dp) | Tyrosine (dp) | Valine (dp) | ||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.