Valine
| Valine | |
|---|---|
| |
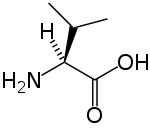
| Systematic name | (S)-2-amino-3-methyl-butanoic acid |
| Abbreviations | Val V |
| Chemical formula | C5H11NO2 |
| Molecular mass | 117.15 g mol-1 |
| Melting point | 315 °C |
| Density | 1.230 g cm-3 |
| Isoelectric point | 5.96 |
| pKa | 2.27 9.52 |
| PubChem | 1182 |
| CAS number | [72-18-4] |
| EINECS number | 200-773-6 |
| SMILES | CC(C)[C@H](N)C(O)=O |
Valine is an α-amino acid that is found in most proteins and is essential in the human diet. It is similar to leucine and isoleucine in being a branched-chain amino acid and whose buildup in the blood and urine, due a particular enzyme deficiency, causes the serious metabolic disorder maple syrup urine disease.
The L-isomer of valine, which is the only form that is involved in protein synthesis, is one of the 20 standard amino acids common in animal proteins and required for normal functioning in humans. Valine is also classified as an "essential amino acid" since it cannot be synthesized by the human body from other compounds through chemical reactions and thus has to be taken in with the diet.
The precision and complex coordination in the universe is revealed in valine's role in proteins. Similar to leucine and isoleucine, valine's structure makes it important for the correct folding of proteins. The functionality of a protein is dependent upon its ability to fold into a precise three-dimensional shape. In sickle-cell disease, valine substitutes for the hydrophilic (binds with water) amino acid glutamic acid in hemoglobin. Because valine is hydrophobic (repelled by water), the hemoglobin does not fold correctly.
In the case of essential amino acids, it is important for individuals to have disciplined eating habits in order to get proper amounts. This is emphasized in the case of maple syrup urine disorder, where one must obtain minimal levels of valine (and leucine and isoleucine) without consuming too much to result in the symptoms.
Valine's three letter code is Val, its one letter code is V, its codons are GUU, GUC, GUA, and GUG, and its systematic name is 2-Amino-3-methylbutanoic acid (IUPAC-IUB 1983). Valine is named after the plant valerian.
Structure
In biochemistry, the term amino acid is frequently used to refer specifically to alpha amino acids: those amino acids in which the amino and carboxylate groups are attached to the same carbon, the so-called α–carbon (alpha carbon). The general structure of these alpha amino acids is:
R | H2N-C-COOH | H
where R represents a side chain specific to each amino acid.
Most amino acids occur in two possible optical isomers, called D and L. The L amino acids represent the vast majority of amino acids found in proteins. They are called proteinogenic amino acids. As the name "proteinogenic" (literally, protein building) suggests, these amino acid are encoded by the standard genetic code and participate in the process of protein synthesis. In valine, only the L-stereoisomer is involved in synthesis of mammalian proteins.
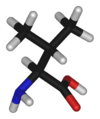
Valine's chemical formula is (CH3)2CH-CH(NH2)-COOH, or in general form C5H11NO2 (IUPAC-IUB 1983).
Like isoleucine and leucine, valine has large aliphatic hydrophobic side chains. Its molecules are rigid, and its mutual hydrophobic interactions are important for the correct folding of proteins, as these chains tend to be located inside of the protein molecule.
Sources
Nutritional sources of valine include cottage cheese, fish, poultry, peanuts, sesame seeds, and lentils.
In a 1994 report released by five top cigarette companies, valine is one of the 599 additives to cigarettes. Like most cigarette additives, the reason for its inclusion is unknown.
Biosynthesis
As an essential amino acid, valine is not synthesized in animals, hence it must be ingested, usually as a component of proteins. Valine is synthesized in plants via several steps starting from pyruvic acid. The initial part of the pathway also leads to leucine.
The intermediate α-ketovalerate undergoes reductive amination with glutamate. Enzymes involved in this biosynthesis include (Lehninger 2000):
- acetolactate synthase
- acetohydroxy acid isomeroreductase
- dihydroxyacid dehydratase
- valine aminotransferase
Maple syrup urine disease
Maple syrup urine disease (MSUD) is an autosomal recessive metabolic disorder of amino acid metabolism. It is also called branched chain ketoaciduria.
This amino acid acidopathy is due to a deficiency of the metabolic enzyme branched chain α-keto acid dehydrogenase (BCKDH) leading to a buildup of the branched-chain amino acids valine, leucine, and isoleucine in the blood and urine.
MSUD is characterized by an infant with sweet-smelling urine with an odor similar to that of maple syrup. Infants with this disease seem healthy at birth but if left untreated suffer severe brain damage and eventually die. Because of a genetic bottleneck effect, MSUD has a much higher prevalence in children of Amish and Mennonite descent.
From early infancy, the condition is characterized by poor feeding, vomiting, lack of energy (lethargy), seizures, and mental health issues. The urine of affected infants has a distinctive sweet odor, much like burned caramel, that gives the condition its name.
Treatment of the MSUD, like diabetes, requires careful monitoring of blood chemistry and involves both special diet and frequent testing. A diet with minimal levels of the amino acids valine, leucine, and isoleucine must be maintained in order to prevent neurological damage. Usually patients or parents of patients are assisted by a physician or dietician. This diet must be adhered to strictly and permanently. However, with proper treatment those afflicted are able to live healthy, normal lives and not suffer the severe neurological damage that characterizes the untreated disease.
ReferencesISBN links support NWE through referral fees
- Doolittle, R. F. 1989. “Redundancies in protein sequences.” In Prediction of Protein Structures and the Principles of Protein Conformation. Edited by G. D. Fasman. New York: Plenum Press. ISBN 0306431319
- International Union of Pure and Applied Chemistry and International Union of Biochemistry and Molecular Biology (IUPAC-IUB) Joint Commission on Biochemical Nomenclature. 1983. “Nomenclature and symbolism for amino acids and peptides: Recommendations on organic & biochemical nomenclature, symbols & terminology.” IUPAC-IUB. Retrieved June 14, 2007.
- Lehninger, A. L., D. L. Nelson, and M. M. Cox. 2000. Lehninger Principles of Biochemistry, 3rd ed. New York: Worth Publishing. ISBN 1572591536
External links
All links retrieved May 3, 2023.
| Major families of biochemicals | ||
| Peptides | Amino acids | Nucleic acids | Carbohydrates | Nucleotide sugars | Lipids | Terpenes | Carotenoids | Tetrapyrroles | Enzyme cofactors | Steroids | Flavonoids | Alkaloids | Polyketides | Glycosides | ||
| Analogues of nucleic acids: | The 20 Common Amino Acids | Analogues of nucleic acids: |
| Alanine (dp) | Arginine (dp) | Asparagine (dp) | Aspartic acid (dp) | Cysteine (dp) | Glutamic acid (dp) | Glutamine (dp) | Glycine (dp) | Histidine (dp) | Isoleucine (dp) | Leucine (dp) | Lysine (dp) | Methionine (dp) | Phenylalanine (dp) | Proline (dp) | Serine (dp) | Threonine (dp) | Tryptophan (dp) | Tyrosine (dp) | Valine (dp) | ||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.