Air pollution
| |||
| Air pollution | |||
| Acid rain • Air Pollution Index • Air Quality Index • Atmospheric dispersion modeling • Chlorofluorocarbon • Global dimming • Global warming • Haze • Indoor air quality • Ozone depletion • Particulate • Smog • Roadway air dispersion | |||
| Water pollution | |||
| Eutrophication • Hypoxia • Marine pollution • Ocean acidification • Oil spill • Ship pollution • Surface runoff • Thermal pollution • Wastewater • Waterborne diseases • Water quality • Water stagnation | |||
| Soil contamination | |||
| Bioremediation • Herbicide • Pesticide •Soil Guideline Values (SGVs) | |||
| Radioactive contamination | |||
| Actinides in the environment • Environmental radioactivity • Fission product • Nuclear fallout • Plutonium in the environment • Radiation poisoning • radium in the environment • Uranium in the environment | |||
| Other types of pollution | |||
| Invasive species • Light pollution • Noise pollution • Radio spectrum pollution • Visual pollution | |||
| Government acts | |||
| Clean Air Act • Clean Water Act • Kyoto Protocol • Water Pollution Control Act • Environmental Protection Act 1990 | |||
| Major organizations | |||
| DEFRA • Environmental Protection Agency • Global Atmosphere Watch • Greenpeace • National Ambient Air Quality Standards | |||
| Related topics | |||
| Natural environment |
Air pollution is the human introduction into the atmosphere of chemicals, particulate matter, or biological materials that cause harm or discomfort to humans or other living organisms, or damages the environment.[1] Air pollution causes deaths and respiratory disease.[2] Air pollution is often identified with major stationary sources, but the greatest source of emissions is mobile sources, mainly automobiles.[3] Gases such as carbon dioxide, which contribute to global warming, have recently been labeled as pollutants by climate scientists, while they also recognize that carbon dioxide is essential for plant life through photosynthesis.
The atmosphere is a complex, dynamic natural gaseous system that is essential to support life on planet Earth. Stratospheric ozone depletion due to air pollution has long been recognized as a threat to human health as well as to the Earth's ecosystems.
Pollutants

There are many substances in the air which may impair the health of plants and animals (including humans), or reduce visibility. These arise both from natural processes and human activity. Substances not naturally found in the air or at greater concentrations or in different locations from usual are referred to as pollutants.
Pollutants can be classified as either primary or secondary. Primary pollutants are substances directly emitted from a process, such as ash from a volcanic eruption, the carbon monoxide gas from a motor vehicle exhaust or sulfur dioxide released from factories.
Secondary pollutants are not emitted directly. Rather, they form in the air when primary pollutants react or interact. An important example of a secondary pollutant is ground level ozone - one of the many secondary pollutants that make up photochemical smog.
Note that some pollutants may be both primary and secondary: that is, they are both emitted directly and formed from other primary pollutants.
Major primary pollutants produced by human activity include:
- Sulfur oxides (SOx) especially sulfur dioxide are emitted from burning of coal and oil.
- Nitrogen oxides (NOx) especially nitrogen dioxide are emitted from high temperature combustion. Can be seen as the brown haze dome above or plume downwind of cities.
- Carbon monoxide is colorless, odorless, non-irritating but very poisonous gas. It is a product by incomplete combustion of fuel such as natural gas, coal or wood. Vehicular exhaust is a major source of carbon monoxide.
- Carbon dioxide (CO2), a greenhouse gas emitted from combustion.
- Volatile organic compounds (VOC), such as hydrocarbon fuel vapors and solvents.
- Particulate matter (PM), measured as smoke and dust. PM10 is the fraction of suspended particles 10 micrometers in diameter and smaller that will enter the nasal cavity. PM2.5 has a maximum particle size of 2.5 µm and will enter the bronchi’s and lungs.
- Toxic metals, such as lead, cadmium and copper.
- Chlorofluorocarbons (CFCs), harmful to the ozone layer emitted from products currently banned from use.
- Ammonia (NH3) emitted from agricultural processes.
- Odors, such as from garbage, sewage, and industrial processes
- Radioactive pollutants produced by nuclear explosions and war explosives, and natural processes such as radon.
Secondary pollutants include:
- Particulate matter formed from gaseous primary pollutants and compounds in photochemical smog, such as nitrogen dioxide.
- Ground level ozone (O3) formed from NOx and VOCs.
- Peroxyacetyl nitrate (PAN) similarly formed from NOx and VOCs.
Minor air pollutants include:
- A large number of minor hazardous air pollutants. Some of these are regulated in USA under the Clean Air Act and in Europe under the Air Framework Directive.
- A variety of persistent organic pollutants, which can attach to particulate matter.
Sources


Sources of air pollution refer to the various locations, activities or factors which are responsible for the releasing of pollutants in the atmosphere. These sources can be classified into two major categories which are:
Anthropogenic sources (human activity) mostly related to burning different kinds of fuel
- "Stationary Sources" as smoke stacks of power plants, manufacturing facilities, municipal waste incinerators.
- "Mobile Sources" as motor vehicles, aircraft etc.
- Marine vessels, such as container ships or cruise ships, and related port air pollution.
- Burning wood, fireplaces, stoves, furnaces and incinerators .
- Oil refining, and industrial activity in general.
- Chemicals, dust and controlled burn practices in agriculture and forestry management, (see Dust Bowl).
- Fumes from paint, hair spray, varnish, aerosol sprays and other solvents.
- Waste deposition in landfills, which generate methane.
- Military, such as nuclear weapons, toxic gases, germ warfare and rocketry.
Natural sources
- Dust from natural sources, usually large areas of land with little or no vegetation.
- Methane, emitted by the digestion of food by animals, for example cattle.
- Radon gas from radioactive decay within the Earth's crust.
- Smoke and carbon monoxide from wildfires.
- Volcanic activity, which produce sulfur, chlorine, and ash particulates.
Emission factors
Air pollutant emission factors are representative values that attempt to relate the quantity of a pollutant released to the ambient air with an activity associated with the release of that pollutant. These factors are usually expressed as the weight of pollutant divided by a unit weight, volume, distance, or duration of the activity emitting the pollutant (e.g., kilograms of particulate emitted per megagram of coal burned). Such factors facilitate estimation of emissions from various sources of air pollution. In most cases, these factors are simply averages of all available data of acceptable quality, and are generally assumed to be representative of long-term averages.
The United States Environmental Protection Agency has published a compilation of air pollutant emission factors for a multitude of industrial sources.[4] Other countries have published similar compilations, as has the European Environment Agency.[5] [6] [7]
Indoor air quality (IAQ)
A lack of ventilation indoors concentrates air pollution where people often spend the majority of their time. Radon (Rn) gas, a carcinogen, is exuded from the Earth in certain locations and trapped inside houses. Building materials including carpeting and plywood emit formaldehyde (H2CO) gas. Paint and solvents give off volatile organic compounds (VOCs) as they dry. Lead paint can degenerate into dust and be inhaled. Intentional air pollution is introduced with the use of air fresheners, incense, and other scented items. Controlled wood fires in stoves and fireplaces can add significant amounts of smoke particulates into the air, inside and out. Indoor pollution fatalities may be caused by using pesticides and other chemical sprays indoors without proper ventilation.
Carbon monoxide (CO) poisoning and fatalities are often caused by faulty vents and chimneys, or by the burning of charcoal indoors. Chronic carbon monoxide poisoning can result even from poorly adjusted pilot lights. Traps are built into all domestic plumbing to keep sewer gas, hydrogen sulfide, out of interiors. Clothing emits tetrachloroethylene, or other dry cleaning fluids, for days after dry cleaning.
Though its use has now been banned in many countries, the extensive use of asbestos in industrial and domestic environments in the past has left a potentially very dangerous material in many localities. Asbestosis is a chronic inflammatory medical condition affecting the tissue of the lungs. It occurs after long-term, heavy exposure to asbestos from asbestos-containing materials in structures. Sufferers have severe dyspnea (shortness of breath) and are at an increased risk regarding several different types of lung cancer. As clear explanations are not always stressed in non-technical literature, care should be taken to distinguish between several forms of relevant diseases. According to the World Health Organization (WHO), these may defined as; asbestosis, lung cancer, and mesothelioma (generally a very rare form of cancer, when more widespread it is almost always associated with prolonged exposure to asbestos).
Biological sources of air pollution are also found indoors, as gases and airborne particulates. Pets produce dander, people produce dust from minute skin flakes and decomposed hair, dust mites in bedding, carpeting and furniture produce enzymes and micrometer-sized fecal droppings, inhabitants emit methane, mold forms in walls and generates mycotoxins and spores, air conditioning systems can incubate Legionnaires' disease and mold, and houseplants, soil and surrounding gardens can produce pollen, dust, and mold. Indoors, the lack of air circulation allows these airborne pollutants to accumulate more than they would otherwise occur in nature.
Health effects
Health effects caused by air pollutants may range from subtle biochemical and physiological changes to difficulty in breathing, wheezing, coughing, and aggravation of existing respiratory and cardiac conditions. These effects can result in increased medication use, increased doctor or emergency room visits, more hospital admissions and premature death. The human health effects of poor air quality are far-reaching, but principally affect the body's respiratory system and the cardiovascular system. Individual reactions to air pollutants depend on the type of pollutant a person is exposed to, the degree of exposure, the individual's health status, and genetics.
The World Health Organization states that 2.4 million people die each year from causes directly attributable to air pollution, with 1.5 million of these deaths attributable to indoor air pollution.[2] A study by the University of Birmingham has shown a strong correlation between pneumonia-related deaths and air pollution from motor vehicles.[8] Direct causes of deaths related to air pollution include aggravated asthma, bronchitis, emphysema, lung and heart diseases, and respiratory allergies.
The worst short-term civilian pollution crisis in India was the 1984 Bhopal Disaster.[9] Leaked industrial vapors from the Union Carbide factory, belonging to Union Carbide, Inc., U.S.A., killed more than 2,000 people outright and injured anywhere from 150,000 to 600,000 others, some 6,000 of whom would later die from their injuries. The United Kingdom suffered its worst air pollution event when the December 4 Great Smog of 1952 formed over London. An accidental leak of anthrax spores from a biological warfare laboratory in the former USSR in 1979 near Sverdlovsk is believed to have been the cause of hundreds of civilian deaths. The worst single incident of air pollution to occur in the United States of America occurred in Donora, Pennsylvania in late October, 1948, when 20 people died and over 7,000 were injured.[10]
Effects on children
In cities around the world with high levels air pollutants, children have a higher probability of developing asthma, pneumonia, and other lower respiratory infections. Because children spend more time outdoors and have higher minute ventilation, they are more susceptible to the dangers of air pollution.
Research by the World Health Organization shows that the highest concentrations of particulate matter can be found in countries with low economic strength and high poverty and population rates. Examples of these countries include Egypt, Sudan, Mongolia, and Indonesia. Protective measures to ensure the health of youth are being undertaken in cities such as New Delhi, where buses now use compressed natural gas to help eliminate the “pea-soup” fog.[11]
In the U.S., the Clean Air Act was passed in 1970. However, in 2002, at least 146 million Americans were living in areas that did not meet at least one of the “criteria pollutants” laid out in the 1997 National Ambient Air Quality Standards.[12] Those pollutants included: ozone, particulate matter, sulfur dioxide, nitrogen dioxide, carbon monoxide, and lead.
Cystic fibrosis
Cystic fibrosis patients are born with decreased lung function. For them, everyday pollutants such as smoke emissions from automobiles, tobacco smoke, and improper use of indoor heating devices can more severely affect lung function.[13]
A study from 1999 to 2000 by the University of Washington showed that patients near and around particulate matter air pollution had an increased risk of pulmonary exacerbations and decrease in lung function.[14] Patients were examined before the study for amounts of specific pollutants like P. aeruginosa or B. cepacia, as well as their socioeconomic standing. During the time of the study, 117 deaths were associated with air pollution. A trend was noticed that patients living in large metropolitan areas had higher level of pollutants in their system because of greater emission levels in larger cities.
Chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) includes illnesses such as chronic bronchitis, emphysema, and some forms of asthma.[15] Two researchers, Holland and Reid, conducted research on 293 male postal workers in London during the time of the Great Smog of 1952 (see below) and 477 male postal workers in the rural setting. The amount of the pollutant FEV1 was significantly lower in urban employees however lung function was decreased due to city pollutions such as car fumes and increased amount of cigarette exposure.[16]
It is believed that, much like cystic fibrosis, serious health problems become more apparent among people living in a more urban environment. Studies have shown that in urban areas, patients suffer mucus hypersecretion, lower levels of lung function, and more self-diagnosis of chronic bronchitis and emphysema.[17]
The Great Smog of 1952
In a span of four days, a combination of dense fog and sooty black coal smoke covered the London area.[18] The fog was so dense that residents of London could not see in front of them. The extreme reduction in visibility was accompanied by an increase in criminal activity as well as transportation delays and a virtual shutdown of the city. During the four-day period of the fog, 12,000 people are believed to have been killed.[19]
Environmental impacts
The greenhouse effect is a phenomenon whereby greenhouse gases create a condition in the upper atmosphere causing a trapping of heat and leading to increased surface and lower tropospheric temperatures. It shares this property with many other gases, the largest overall forcing on Earth coming from water vapor. Other greenhouse gases include methane, hydrofluorocarbons, perfluorocarbons, chlorofluorocarbons, NOx, and ozone. Many greenhouse gases, contain carbon, and some of that from fossil fuels.
This effect has been understood by scientists for about a century, and technological advancements during this period have helped increase the breadth and depth of data relating to the phenomenon. Currently, scientists are studying the role of changes in composition of greenhouse gases from natural and anthropogenic sources for the effect on climate change.
A number of studies have also investigated the potential for long-term rising levels of atmospheric carbon dioxide to cause slight increases in the acidity of ocean waters and the possible effects of this on marine ecosystems. However, carbonic acid is a very weak acid, and is utilized by marine organisms during photosynthesis.
Reduction efforts
There are various air pollution control technologies and urban planning strategies available to reduce air pollution.
Efforts to reduce pollution from mobile sources includes primary regulation (many developing countries have permissive regulations), expanding regulation to new sources (such as cruise and transport ships, farm equipment, and small gas-powered equipment such as lawn trimmers, chainsaws, and snowmobiles), increased fuel efficiency (such as through the use of hybrid vehicles), conversion to cleaner fuels (such as bioethanol, biodiesel, or conversion to electric vehicles).
Control devices
The following items are commonly used as pollution control devices by industry or transportation devices. They can either destroy contaminants or remove them from an exhaust stream before it is emitted into the atmosphere.
- Particulate control
- Mechanical collectors (dust cyclones, multicyclones)
- Electrostatic precipitators
- Baghouses
- Particulate scrubbers
- Scrubbers
- Baffle spray scrubber
- Cyclonic spray scrubber
- Ejector venturi scrubber
- Mechanically aided scrubber
- Spray tower
- Wet scrubber
- NOx control
- Low NOx burners
- Selective catalytic reduction (SCR)
- Selective non-catalytic reduction (SNCR)
- NOx scrubbers
- Exhaust gas recirculation
- Catalytic converter (also for VOC control)
- VOC abatement
- Adsorption systems, such as activated carbon
- Flares
- Thermal oxidizers
- Catalytic oxidizers
- Biofilters
- Absorption (scrubbing)
- Cryogenic condensers
- Vapor recovery systems
- Acid Gas/SO2 control
- Wet scrubbers
- Dry scrubbers
- Flue gas desulfurization
- Mercury control
- Sorbent Injection Technology
- Electro-Catalytic Oxidation (ECO)
- K-Fuel
- Dioxin and furan control
- Miscellaneous associated equipment
- Source capturing systems
- Continuous emissions monitoring systems (CEMS)
Atmospheric dispersion models
The basic technology for analyzing air pollution is through the use of a variety of mathematical models for predicting the transport of air pollutants in the lower atmosphere. The principal methodologies are noted below.
- Point source dispersion, used for industrial sources.
- Line source dispersion, used for airport and roadway air dispersion modeling.
- Area source dispersion, used for forest fires or duststorms.
- Photochemical models, used to analyze reactive pollutants that form smog.
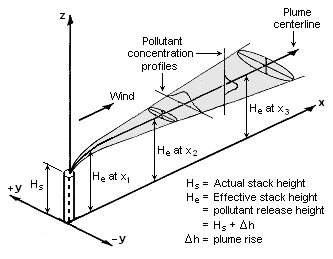
The point source problem is the best understood, since it involves simpler mathematics and has been studied for a long period of time, dating back to about the year 1900. It uses a Gaussian dispersion model to forecast air pollution plumes (as shown in the figure on the right), with consideration given to wind velocity, stack height, emission rate and stability class (a measure of atmospheric turbulence).[20][21]
The roadway air dispersion model was developed starting in the late 1950s and early 1960s in response to requirements of the National Environmental Policy Act and the U.S. Department of Transportation (then known as the Federal Highway Administration) to understand impacts of proposed new highways upon air quality, especially in urban areas. Several research groups were active in this model development, among which were the Environmental Research and Technology (ERT) group in Lexington, Massachusetts, the ESL Inc. group in Sunnyvale, California and the California Air Resources Board group in Sacramento, California.
Area source models were developed in 1971 through 1974 by the ERT and ESL groups, but addressed a smaller fraction of total air pollution emissions, so that their use and need was not as widespread as the line source model, which enjoyed hundreds of different applications as early as the 1970s.
Likewise, photochemical models were developed primarily in the 1960s and 1970s. Their use was constrained to regional needs, such as understanding smog formation in Los Angeles, California.
Legal regulations in some nations
In general, there are two types of air quality standards. The first class of standards (such as the U.S. National Ambient Air Quality Standards) set maximum atmospheric concentrations for specific pollutants. Environmental agencies enact regulations which are intended to result in attainment of these target levels. The second class (such as the North American Air Quality Index) take the form of a scale with various thresholds, which is used to communicate to the public the relative risk of outdoor activity. The scale may or may not distinguish between different pollutants.
Canada
In Canada, air quality is typically evaluated against standards set by the Canadian Council of Minister for the Environment (CCME), an inter-governmental body of federal, provincial and territorial Ministers responsible for the environment. The CCME set Canada Wide Standards(CWS).[22]
European Union
National Emission Ceilings (NEC) for certain atmospheric pollutants are regulated by Directive 2001/81/EC (NECD).[23] As part of the preparatory work associated with the revision of the NECD, the European Commission is assisted by the NECPI working group (National Emission Ceilings – Policy Instruments).
United Kingdom
Air quality targets set by the UK's Department for Environment, Food and Rural Affairs (DEFRA) are mostly aimed at local government representatives responsible for the management of air quality in cities, where air quality management is the most urgent. The UK has established an air quality network where levels of the key air pollutants are published by monitoring centers. Air quality in Oxford, Bath and London[24] is particularly poor. One controversial study performed by the Calor Gas company and published in the Guardian newspaper compared walking in Oxford on an average day to smoking over sixty light cigarettes.[25]
More precise comparisons can be collected from the UK Air Quality Archive[26] which allows the user to compare a cities management of pollutants against the national air quality objectives set by DEFRA in 2000.
Localized peak values are often cited, but average values are also important to human health. The UK National Air Quality Information Archive offers almost real-time monitoring of "current maximum" air pollution measurements for many UK towns and cities. This source offers a wide range of constantly updated data, including:
- Hourly Mean Ozone (µg/m³)
- Hourly Mean Nitrogen dioxide (µg/m³)
- Maximum 15-Minute Mean Sulphur dioxide (µg/m³)
- 8-Hour Mean Carbon monoxide (mg/m³)
- 24-Hour Mean PM10 (µg/m³ Grav Equiv)
DEFRA acknowledges that air pollution has a significant effect on health and has produced a simple banding index system is used to create a daily warning system that is issued by the BBC Weather Service to indicate air pollution levels. DEFRA has published guidelines for people suffering from respiratory and heart diseases.
United States

In the 1960s, 1970s, and 1990s, the United States Congress enacted a series of Clean Air Acts which significantly strengthened regulation of air pollution. Individual U.S. states, some European nations and eventually the European Union followed these initiatives. The Clean Air Act sets numerical limits on the concentrations of a basic group of air pollutants and provide reporting and enforcement mechanisms.
In 1999, the United States EPA replaced the Pollution Standards Index (PSI) with the Air Quality Index (AQI) to incorporate new PM2.5 and Ozone standards.
The effects of these laws have been very positive. In the United States between 1970 and 2006, citizens enjoyed the following reductions in annual pollution emissions:[27]
- carbon monoxide emissions fell from 197 million tons to 89 million tons
- nitrogen oxide emissions fell from 27 million tons to 19 million tons
- sulfur dioxide emissions fell from 31 million tons to 15 million tons
- particulate emissions fell by 80 percent
- lead emissions fell by more than 98 percent
The EPA proposed, in June 2007, a new threshold of 75 ppb. This falls short of the scientific recommendation, but is an improvement over the current standard.
Polluting industries are lobbying to keep the current (weaker) standards in place. Environmentalists and public health advocates are mobilizing to support compliance with the scientific recommendations.
The National Ambient Air Quality Standards are pollution thresholds which trigger mandatory remediation plans by state and local governments, subject to enforcement by the EPA.
See also
- Acid rain
- Atmospheric chemistry
- Emission standard
- Environmental agreement
- Greenhouse effect
- Kyoto Protocol
- Particulate
- Smog
Notes
- ↑ Pollutes. Merriam-Webster Online Dictionary. Retrieved September 27, 2017.
- ↑ 2.0 2.1 Estimated deaths & DALYs attributable to selected environmental risk factors. WHO. Retrieved September 27, 2017.
- ↑ National Research Council: Committee on Air Quality Management in the United States, Board on Environmental Studies and Toxicology, Board on Atmospheric Sciences and Climate, Division on Earth and Life Studies. Air Quality Management in the United States (Washington, DC: National Academies Press, 2004, ISBN 0309089328).
- ↑ Emissions Factors & AP 42. EPA. Retrieved September 27, 2017.
- ↑ EMEP/EEA air pollutant emission inventory guidebook 2016. European Environment Agency. Retrieved September 27, 2017.
- ↑ Australian National Pollutant Inventory Emissions Estimation Technique Manuals. NPI. Retrieved September 27, 2017.
- ↑ Canada's Greenhouse Gas Inventory. Environment Canada. Retrieved September 27, 2017.
- ↑ Study links traffic pollution to thousands of deaths. The Guardian, 2008. Retrieved September 27, 2017.
- ↑ Simi Chakrabarti, 20th anniversary of world's worst industrial disaster. ABC, 2004. Retrieved September 27, 2017.
- ↑ Devra Davis, When Smoke Ran Like Water: Tales of Environmental Deception and the Battle Against Pollution (New York, NY: Basic Books, 2002, ISBN 0465015212).
- ↑ Polluted Cities: The Air Children Breathe. World Health Organization. Retrieved September 27, 2017.
- ↑ Committee on Environmental Health. "Ambient Air Pollution: Health Hazards to Children." Pediatrics 114(6) (2004): 1699-1707.
- ↑ Michael Kymisis and Konstantinos Hadjistavrou, Short-Term Effects Of Air Pollution Levels On Pulmonary Function Of Young Adults. The Internet Journal of Pulmonary Medicine 9(2) (2007). Retrieved September 27, 2017.
- ↑ Christopher H. Goss, Stacey A. Newsom, Jonathan S. Schildcrout, Lianne Sheppard and Joel D. Kaufman, "Effect of Ambient Air Pollution on Pulmonary Exacerbations and Lung Function in Cystic Fibrosis." American Journal of Respiratory and Critical Care Medicine 169 (2004):816-821.
- ↑ John D. Zoidis, The Impact of Air Pollution on COPD. RT: For Decision Makers in Respiratory Care, 1999. Retrieved September 27, 2017.
- ↑ W.W. Holland, and D.D. Reid, "The urban factor in chronic bronchitis." Lancet I (1965):445-448.
- ↑ J. Sunyer,Urban air pollution and Chronic Obstructive Pulmonary disease: a review. European Respiratory Journal 17 (2001):1024-1033. Retrieved September 27, 2017.
- ↑ John Nielsen, The Killer Fog of ’52: Thousands died as Poisonous Air Smothered London. National Public Radio, 2002. Retrieved September 27, 2017.
- ↑ On this Day: 1952 London Fog Clears After days of Chaos. BBC News, 2005. Retrieved September 27, 2017.
- ↑ D.B. Turner, Workbook of atmospheric dispersion estimates: an introduction to dispersion modeling, 2nd Edition. (Boca Raton, FL: CRC Press, 1994, ISBN 156670023X.)
- ↑ M.R. Beychok, Fundamentals Of Stack Gas Dispersion, 4th Edition. (Irvine, CA: M.R. Beychok, 2005, ISBN 0964458802).
- ↑ Canada-wide Standards. CCME. Retrieved September 27, 2017.
- ↑ Directive 2001/81/EC of the European Parliament and of the Council of 23 October 2001 on national emission ceilings for certain atmospheric pollutants. Eur-Lex. Retrieved September 27, 2017.
- ↑ London. The London Air Quality Network. Retrieved September 27, 2017.
- ↑ Paul Brown, Taking the Oxford air adds up to a 60-a-day habit. The Guardian, August 28, 2004. Retrieved September 27, 2017.
- ↑ Home Page. UK Air Quality Archive. Retrieved September 27, 2017.
- ↑ Don't Be Very Worried National Center for Policy Analysis, May 25, 2006. Retrieved September 27, 2017.
ReferencesISBN links support NWE through referral fees
- Beychok, M.R. Fundamentals Of Stack Gas Dispersion, 4th Edition. Irvine, CA: M.R. Beychok, 2005. ISBN 0964458802
- Brasseur, Guy P., John J. Orlando, and Geoffrey S. Tyndall. Atmospheric Chemistry and Global Change. Oxford, UK: Oxford University Press, 1999. ISBN 0195105214.
- Davis, Devra. When Smoke Ran Like Water: Tales of Environmental Deception and the Battle Against Pollution. New York, NY: Basic Books, 2002. ISBN 0465015212
- Finlayson-Pitts, Barbara J., and James N. Pitts, Jr. Chemistry of the Upper and Lower Atmosphere: Theory, Experiments, and Applications. San Diego, CA: Academic Press, 2000. ISBN 012257060X.
- Seinfeld, John H., and Spyros N. Pandis. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 2nd ed. New York, NY: Wiley, 2006. ISBN 0471828572.
- Turner, D.B. Workbook of atmospheric dispersion estimates: an introduction to dispersion modeling, 2nd Edition. Boca Raton, FL: CRC Press, 1994. ISBN 156670023X
- Warneck, Peter. Chemistry of the Natural Atmosphere, 2nd ed. San Diego, CA: Academic Press, 2000. ISBN 0127356320.
- Wayne, Richard P. Chemistry of Atmospheres, 3rd ed. Oxford, UK: Oxford University Press, 2000. ISBN 019850375X.
External links
All links retrieved June 16, 2023.
- Climate and Clean Air Coalition.
- Stuff in the Air Standard air quality modeling procedure for industrial sources.
- Wiki on Atmospheric Dispersion Modeling. Addresses the international community of atmospheric dispersion modelers - primarily researchers, but also users of models. Its purpose is to pool experiences gained by dispersion modelers during their work.
- An Unfiltered Look at Indoor and Outdoor Air Pollution
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.