Sulfuric acid
| Sulfuric acid | |
|---|---|
| |
| General | |
| Systematic name | sulfuric acid |
| Other names | oil of vitriol |
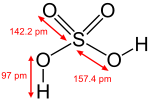
| Molecular formula | H2SO4 (aq) |
| Molar mass | 98.08 g mol−1 |
| Appearance | clear, colorless, odorless liquid |
| CAS number | [7664-93-9] |
| Properties | |
| Density and phase | 1.84 g cm−3, liquid |
| Solubility in water | fully miscible (exothermic) |
| Melting point | 10 °C (283 K) |
| Boiling point | 338 °C (611 K) |
| pKa | −3 (disputed, see discussion) 1.99 |
| Viscosity | 26.7 cP at 20°C |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Corrosive (C) |
| NFPA 704 | |
| R-phrases | R35 |
| S-phrases | S1/2, S26, S30, S45 |
| Flash point | Non-flammable |
| RTECS number | WS5600000 |
| Supplementary data page | |
| Structure & properties | n, εr, etc. |
| Thermodynamic data | Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Related strong acids | Selenic acid Hydrochloric acid Nitric acid |
| Related compounds | Hydrogen sulfide Sulfurous acid Peroxymonosulfuric acid Sulfur trioxide Oleum |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Sulfuric acid (or sulphuric acid in British English) is a strong mineral acid with the chemical formula H2SO4. It is soluble in water at all concentrations. It was once known as oil of vitriol, a term coined by the eighth-century alchemist Jabir ibn Hayyan (Geber), the chemical's probable discoverer.[1]
Sulfuric acid has many applications, and is produced in greater amounts than any other chemical besides water. World production in 2001 was 165 million tonnes, with an approximate value of $8 billion. Principal uses include ore processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. Many proteins are made of sulfur-containing amino acids (such as cysteine and methionine), which produce sulfuric acid when metabolized by the body.
History of sulfuric acid
The discovery of sulfuric acid is credited to the eighth-century alchemist Jabir ibn Hayyan (Geber). It was studied later by the ninth-century physician and alchemist ibn Zakariya al-Razi (Rhases), who obtained the substance by the dry distillation of minerals, including iron(II) sulfate heptahydrate (FeSO4 • 7H2O) and copper(II) sulfate pentahydrate (CuSO4 • 5H2O). When heated, these compounds decompose to iron(II) oxide and copper(II) oxide, respectively, giving off water and sulfur trioxide. The combination of water with sulfur trioxide produced a dilute solution of sulfuric acid. This method was popularized in Europe through translations of Arabic and Persian treatises and books by European alchemists, including the thirteenth-century German Albertus Magnus.
Sulfuric acid was known to medieval European alchemists as oil of vitriol, spirit of vitriol, or simply vitriol, among other names. The word vitriol derives from the Latin vitreus (meaning "glass"), for the glassy appearance of the sulfate salts, which also carried the name vitriol. Salts that were given this name included copper(II) sulfate (blue vitriol, or occasionally Roman vitriol), zinc sulfate (white vitriol), iron(II) sulfate (green vitriol), iron(III) sulfate (vitriol of Mars), and cobalt(II) sulfate (red vitriol).

Vitriol was widely considered the most important alchemical substance, intended to be used as a philosopher's stone. Highly purified vitriol was used as a medium to react substances in. This was largely because the acid does not react with gold, often the final aim of alchemical processes. The importance of vitriol to alchemy is highlighted in the alchemical motto, a backronym,[2] Visita Interiora Terrae Rectificando Invenies Occultum Lapidem ('Visit the interior of the earth and rectifying (i.e. purifying) you will find the hidden/secret stone').[3]
In the seventeenth century, the German-Dutch chemist Johann Glauber prepared sulfuric acid by burning sulfur together with saltpeter (potassium nitrate, KNO3), in the presence of steam. As the saltpeter decomposes, it oxidizes the sulfur to SO3, which combines with water to produce sulfuric acid. In 1736, Joshua Ward, a London pharmacist, used this method to begin the first large-scale production of sulfuric acid.
In 1746, in Birmingham, John Roebuck began producing sulfuric acid this way in lead-lined chambers, which were stronger, less expensive, and could be made larger than the glass containers that had been used previously. This lead chamber process allowed the effective industrialization of sulfuric acid production, and, with several refinements, remained the standard method of production for almost two centuries.
Roebuck's sulfuric acid was only about 35–40 percent sulfuric acid. Later refinements in the lead-chamber process by the French chemist Joseph-Louis Gay-Lussac and the British chemist John Glover improved this to 78 percent. However, the manufacture of some dyes and other chemical processes require a more concentrated product, and throughout the eighteenth century, this could only be made by dry distilling minerals in a technique similar to the original alchemical processes. Pyrite (iron disulfide, FeS2) was heated in air to yield iron (II) sulfate (FeSO4), which was oxidized by further heating in air to form iron(III) sulfate (Fe2(SO4)3). When iron(III) sulfate was heated to 480 °C, it decomposed to iron(III) oxide and sulfur trioxide, which could be passed through water to yield sulfuric acid in any concentration. The expense of this process prevented the large-scale use of concentrated sulfuric acid.
In 1831, the British vinegar merchant Peregrine Phillips patented a far more economical process for producing sulfur trioxide and concentrated sulfuric acid, now known as the contact process. Essentially all of the world's supply of sulfuric acid is now produced by this method.
Sulfuric acid at various concentrations
Although nearly 100 percent sulfuric acid can be made, it loses sulfur trioxide (SO3) gas at the boiling point to produce 98.3 percent acid. The 98 percent grade is more stable for storage, making it the usual form for "concentrated" sulfuric acid. Other concentrations of sulfuric acid are used for different purposes. Some common concentrations are noted below.
- Ten percent (pH 1): dilute sulfuric acid for laboratory use.
- 33.5 percent (pH 0.5): battery acid (used in lead-acid batteries).
- 62.18 percent (pH about 0.4): chamber or fertilizer acid.
- 77.67 percent (pH about 0.25): tower or Glover acid.
- 98 percent (pH about 0.1): concentrated.
Given that sulfuric acid is a strong acid, a 0.50 Molar (M) solution of this acid has a pH close to zero.
Different purities are also available. Technical grade H2SO4 is impure and often colored, but it is suitable for making fertilizer. Pure grades, such as US Pharmacopoeia (USP) grade, are used for making pharmaceuticals and dyestuffs.
When high concentrations of SO3(g) are added to sulfuric acid, H2S2O7 is formed. It is called pyrosulfuric acid, fuming sulfuric acid, or oleum. A less common name is Nordhausen acid. Concentrations of oleum are expressed either in terms of percent SO3 (called percent oleum) or percent H2SO4 (the amount made if H2O were added). Common concentrations are 40 percent oleum (109 percent H2SO4) and 65 percent oleum (114.6 percent H2SO4). Pure H2S2O7 is a solid, with a melting point of 36 °C.
Physical properties
Anhydrous H2SO4 is a very polar liquid, with a dielectric constant of around 100. This property arises from the fact that it can dissociate by protonating itself, a process known as autoprotolysis.[4] This protonation occurs to a high degree, more than ten billion times the level seen in water. It may be represented as follows:
- 2 H2SO4 → H3SO4+ + HSO4−
This process allows protons to be highly mobile in H2SO4. It also makes sulfuric acid an excellent solvent for many reactions. In fact, the chemical equilibrium is more complex than that shown above. At equilibrium, 100 percent H2SO4 contains the following species (figures in parentheses indicate amounts in terms of moles per kilogram solvent):
- HSO4− (15.0)
- H3SO4+ (11.3)
- H3O+ (8.0)
- HS2O7− (4.4)
- H2S2O7 (3.6)
- H2O (0.1).
Chemical properties
Reaction with water
The reaction of sulfuric acid with water (called a hydration reaction) produces a large amount of heat, and it is therefore called an exothermic reaction. If water is added to concentrated sulfuric acid, it can boil and spit dangerously. One reason for this behavior is related to the relative densities of the two liquids. Water is less dense than sulfuric acid and will tend to float above the acid.
To dilute the acid safely, one should always add the acid to the water (in small increments) rather than the water to the acid.[5]
The reaction is best thought of as forming hydronium ions, as follows:
- H2SO4 + H2O → H3O+ + HSO4−
And then:
- HSO4− + H2O → H3O+ + SO42−
Because the hydration of sulfuric acid is thermodynamically favorable,[6] sulfuric acid is an excellent dehydrating agent and is used to prepare many dried fruits. The affinity of sulfuric acid for water is sufficiently strong that it will remove hydrogen and oxygen atoms from other compounds. For example, mixing starch (C6H12O6)n and concentrated sulfuric acid will give elemental carbon and water, which is absorbed by the sulfuric acid (which becomes slightly diluted):
- (C6H12O6)n → 6C + 6H2O.
The effect of this can be seen when concentrated sulfuric acid is spilled on paper; the starch reacts to give a burned appearance, the carbon appears as soot would in a fire. A more dramatic illustration occurs when sulfuric acid is added to a tablespoon of white sugar in a cup when a tall rigid column of black porous carbon smelling strongly of caramel emerges from the cup.
Other reactions of sulfuric acid
As an acid, sulfuric acid reacts with most bases to give the corresponding sulfates. For example, copper(II) sulfate, the familiar blue salt of copper used for electroplating and as a fungicide, is prepared by the reaction of copper(II) oxide with sulfuric acid:
- CuO + H2SO4 → CuSO4 + H2O
Sulfuric acid can be used to displace weaker acids from their salts. For example its reaction with sodium acetate gives acetic acid:
H2SO4 + CH3COONa → NaHSO4 + CH3COOH
Likewise, the reaction of sulfuric acid with potassium nitrate can be used to produce nitric acid, along with a precipitate of potassium bisulfate. With nitric acid itself, sulfuric acid acts as both an acid and a dehydrating agent, forming the nitronium ion NO2+, which is important in nitration reactions involving electrophilic aromatic substitution. This type of reaction where protonation occurs on an oxygen atom, is important in many reactions in organic chemistry, such as Fischer esterification and the dehydration of alcohols.
Sulfuric acid reacts with most metals in a single displacement reaction to produce hydrogen gas and the metal sulfate. Dilute H2SO4 attacks iron, aluminum, zinc, manganese, and nickel, but tin and copper require hot concentrated acid. Lead and tungsten, however, are resistant to sulfuric acid. The reaction with iron is typical for most of these metals, but the reaction with tin is unusual in that it produces sulfur dioxide rather than hydrogen. These reactions are shown here:
Manufacture
Sulfuric acid is produced from sulfur, oxygen, and water via the contact process.
In the first step, sulfur is burned to produce sulfur dioxide.
This product is then oxidized to sulfur trioxide using oxygen in the presence of a vanadium(V) oxide catalyst.
Finally, the sulfur trioxide is treated with water (usually as 97-98 percent H2SO4 containing two to three percent water) to produce 98-99 percent sulfuric acid.
Note that directly dissolving SO3 in water is impractical, because the reaction is highly exothermic and generates mists instead of a liquid.
An alternative method is to absorb SO3 into H2SO4, to produce oleum (H2S2O7). The oleum is then diluted to form sulfuric acid.
- (3) H2SO4(l) + SO3 → H2S2O7(l)
Oleum is reacted with water to form concentrated H2SO4.
- (4) H2S2O7(l) + H2O(l) → 2 H2SO4(l)
Uses
Sulfuric acid is a very important commodity chemical, and indeed a nation's sulfuric acid production is a good indicator of its industrial strength.[7] The major use (60 percent of total worldwide) for sulfuric acid is in the "wet method" for the production of phosphoric acid, used for manufacture of phosphate fertilizers and trisodium phosphate for detergents. This method involves the use of phosphate rock, and more than 100 million metric tons is processed annually. This raw material, shown below as fluorapatite (Ca5F(PO4)3) (though the exact composition may vary), is treated with 93 percent sulfuric acid to produce calcium sulfate (CaSO4), hydrogen fluoride (HF), and phosphoric acid (H3PO4). The HF is removed as hydrofluoric acid. The overall process can be represented as follows:
- Ca5F(PO4)3 + 5 H2SO4 + 10 H2O → 5 CaSO4•2 H2O + HF + 3 H3PO4
Sulfuric acid is used in large quantities in iron and steel making principally as pickling-acid used to remove oxidation, rust and scale from rolled sheet and billets prior to sale into the automobile and white-goods business. The used acid is often re-cycled using a Spent Acid Regeneration (SAR) plant. These plants combust the spent acid with natural gas, refinery gas, fuel oil or other suitable fuel source. This combustion process produces gaseous sulfur dioxide (SO2) and sulfur trioxide (SO3) which are then used to manufacture "new" sulfuric acid. These types of plants are common additions to metal smelting plants, oil refineries, and other places where sulfuric acid is consumed on a large scale, as operating a SAR plant is much cheaper than purchasing the commodity on the open market.
Ammonium sulfate, an important nitrogen fertilizer is most commonly produced as a by-product from coking plants supplying the iron and steel making plants, Reacting the ammonia produced in the thermal decomposition of coal with waste sulfuric acid allows the ammonia to be crystallized out as a salt (often brown because of iron contamination) and sold into the agro-chemicals industry.
Another important use for sulfuric acid is for the manufacture of aluminium sulfate, also known as papermaker's alum. This can react with small amounts of soap on paper pulp fibers to give gelatinous aluminium carboxylates, which help to coagulate the pulp fibers into a hard paper surface. It is also used for making aluminum hydroxide, which is used at water treatment plants to filter out impurities, as well as to improve the taste of the water. Aluminum sulfate is made by reacting bauxite with sulfuric acid:
Sulfuric acid is used for a variety of other purposes in the chemical industry. For example, it is the usual acid catalyst for the conversion of cyclohexanoneoxime to caprolactam, used for making nylon. It is used for making hydrochloric acid from salt via the Mannheim process. Much H2SO4 is used in petroleum refining, for example as a catalyst for the reaction of isobutane with isobutylene to give isooctane, a compound that raises the octane rating of gasoline (petrol). Sulfuric acid is also important in the manufacture of dyestuffs.
A mixture of sulfuric acid and water is sometimes used as the electrolyte in various types of lead-acid battery where it undergoes a reversible reaction where lead and lead dioxide are converted to lead(II) sulfate. Sulfuric acid is also the principal ingredient in some drain cleaners, used to clear blockages consisting of paper, rags, and other materials not easily dissolved by caustic solutions.
Sulfuric acid is also used as a general dehydrating agent in its concentrated form. See Reaction with water.
Sulfur-iodine cycle
The sulfur-iodine cycle is a series of thermochemical processes used to obtain hydrogen. It consists of three chemical reactions whose net reactant is water and whose net products are hydrogen and oxygen.
The sulfur and iodine compounds are recovered and reused, hence the consideration of the process as a cycle. This process is endothermic and must occur at high temperatures, so energy in the form of heat has to be supplied.
The sulfur-iodine cycle has been proposed as a way to supply hydrogen for a hydrogen-based economy. It does not require hydrocarbons like current methods of steam reforming.
The sulfur-iodine cycle is currently being researched as a feasible method of obtaining hydrogen, but the concentrated, corrosive acid at high temperatures poses currently insurmountable safety hazards if the process were built on large-scale.
Environmental aspects
Sulfuric acid is a constituent of acid rain, being formed by atmospheric oxidation of sulfur dioxide in the presence of water, i.e. oxidation of sulfurous acid. Sulfur dioxide is the main product when the sulfur in sulfur-containing fuels such as coal or oil is burned.
Sulfuric acid is formed naturally by the oxidation of sulfide minerals, such as iron sulfide. The resulting water can be highly acidic and is called Acid Rock Drainage (ARD). The acidic water so formed can dissolve metals present in sulfide ores, resulting in brightly colored and toxic streams. The oxidation of iron sulfide pyrite by molecular oxygen produces iron(II), or Fe2+:
The Fe2+ can be further oxidized to Fe3+, according to:
and the Fe3+ so produced can be precipitated as the hydroxide or hydrous oxide. The equation for the formation of the hydroxide is:
The iron(III) ion ("ferric iron," in casual nomenclature) can also oxidize pyrite. When iron(III) oxidation of pyrite occurs, the process can become rapid and pH values below zero have been measured in ARD from this process.
ARD can also produce sulfuric acid at a slower rate, so that the Acid Neutralization Capacity (ANC) of the aquifer can neutralize the produced acid. In such cases, the Total Dissolved solids (TDS) concentration of the water can be increased form the dissolution of minerals from the acid-neutralization reaction with the minerals.
Extraterrestrial sulfuric acid
Sulfuric acid is produced in the upper atmosphere of Venus by the Sun's photochemical action on carbon dioxide, sulfur dioxide, and water vapor. Ultraviolet photons of wavelengths less than 169 nm can photodissociate carbon dioxide into carbon monoxide and atomic oxygen. Atomic oxygen is highly reactive; when it reacts with sulfur dioxide, a trace component of the Venerian atmosphere, the result is sulfur trioxide, which can combine with water vapor, another trace component of Venus' atmosphere, to yield sulfuric acid.
In the upper, cooler portions of Venus's atmosphere, sulfuric acid can exist as a liquid, and thick sulfuric acid clouds completely obscure the planet's surface from above. The main cloud layer extends from 45–70 km above the planet's surface, with thinner hazes extending as low as 30 and as high as 90 km above the surface.
Infrared spectra from NASA's Galileo mission show distinct absorptions on Europa, a moon of Jupiter, that have been attributed to one or more sulfuric acid hydrates. The interpretation of the spectra is somewhat controversial. Some planetary scientists prefer to assign the spectral features to the sulfate ion, perhaps as part of one or more minerals on Europa's surface.
Safety
Laboratory hazards
The corrosive properties of sulfuric acid are accentuated by its highly exothermic reaction with water. Hence burns from sulfuric acid are potentially more serious than those of comparable strong acids (e.g. hydrochloric acid), as there is additional tissue damage due to dehydration and particularly due to the heat liberated by the reaction with water, i.e. secondary thermal damage. The danger is obviously greater with more concentrated preparations of sulfuric acid, but it should be remembered that even the normal laboratory "dilute" grade (approx. one M, ten percent) will char paper by dehydration if left in contact for a sufficient length of time. The standard first aid treatment for acid spills on the skin is, as for other corrosive agents, irrigation with large quantities of water: Washing should be continued for a sufficient length of time—at least ten to fifteen minutes—in order to cool the tissue surrounding the acid burn and to prevent secondary damage. Contaminated clothing must be removed immediately and the underlying skin washed thoroughly.
Preparation of the diluted acid can also be dangerous due to the heat released in the dilution process. It is essential that the concentrated acid is added to water and not the other way round, to take advantage of the relatively high heat capacity of water. Addition of water to concentrated sulfuric acid leads at best to the dispersal of a sulfuric acid aerosol, at worst to an explosion. Preparation of solutions greater than six M (35 percent) in concentration is the most dangerous, as the heat produced can be sufficient to boil the diluted acid: efficient mechanical stirring and external cooling (e.g. an ice bath) are essential.
Industrial hazards
Although sulfuric acid is nonflammable, contact with metals in the event of a spillage can lead to the liberation of hydrogen gas. The dispersal of acid aerosols and gaseous sulfur dioxide is an additional hazard of fires involving sulfuric acid. Water should not be used as the extinguishing agent because of the risk of further dispersal of aerosols: carbon dioxide is preferred where possible.
Sulfuric acid is not considered toxic besides its obvious corrosive hazard, and the main occupational risks are skin contact leading to burns (see above) and the inhalation of aerosols. Exposure to aerosols at high concentrations leads to immediate and severe irritation of the eyes, respiratory tract and mucous membranes: this ceases rapidly after exposure, although there is a risk of subsequent pulmonary edema if tissue damage has been more severe. At lower concentrations, the most commonly reported symptom of chronic exposure to sulfuric acid aerosols is erosion of the teeth, found in virtually all studies: indications of possible chronic damage to the respiratory tract are inconclusive as of 1997. In the United States, the permissible exposure limit (PEL) for sulfuric acid is fixed at one mg/m3: limits in other countries are similar. Interestingly there have been reports of sulfuric acid ingestion leading to vitamin B12 deficiency with subacute combined degeneration. The spinal cord is most often affected in such cases, but the optic nerves may show demyelination, loss of axons and gliosis.
See also
Notes
- ↑ Amin A. Khairallah, Outline of Arabic Contributions to Medicine, chapter 10 (Beirut, 1946).
- ↑ A backronym is a type of acronym that begins as an ordinary word and is later interpreted as an acronym.
- ↑ Mentioned in L'Azoth des Philosophes by the fifteenth-century alchemist Basilius Valentinus.
- ↑ N.N. Greenwood, and A. Earnshaw, Chemistry of the Elements (Oxford, UK: Pergamon Press, 1984, ISBN 0080220576), 837-845.
- ↑ To remember this rule, several mnemonics have been coined, such as:
- Always do things as you oughta, add the acid to the water. If you think your life's too placid, add the water to the acid.
- A.A.: Add Acid.
- Drop acid, not water.
- ↑ The change in enthalpy for the reaction is ΔH = -880 kilojoules/mole.
- ↑ Philip J. Chenier, Survey of Industrial Chemistry (New York: Springer, 2002, ISBN 0306472465), 45-57.
ReferencesISBN links support NWE through referral fees
- Agamanolis, D.P. "Metabolic and toxic disorders." In: Prayson R. (ed.) Neuropathology: a volume in the foundations in diagnostic pathology series. Philadelphia: Elsevier/Churchill Livingstone, 2005.
- Chang, Raymond. Chemistry, 9th ed. New York: McGraw-Hill Science/Engineering/Math, 2006. ISBN 0073221031
- Chenier, Philip J. Survey of Industrial Chemistry. New York: Springer, 2002. ISBN 0306472465
- Davenport, W.G., and M.J. King. Sulfuric Acid Manufacture: Analysis, Control, and Optimization. Elsevier Science, 2005. ISBN 0080444288
- Greenwood, N.N., and A. Earnshaw. Chemistry of the Elements, 2nd ed. Oxford, UK; Burlington, MA: Butterworth-Heinemann, Elsevier Science, 1998. ISBN 0750633654.
- Khairallah, Amin A. Outline of Arabic Contributions to Medicine. Beirut, 1946.
- Lide, David R. CRC Handbook of Chemistry and Physics, 87th ed. Boca Raton, FL: CRC Press, 2006. ISBN 0849304873
External links
All links retrieved February 26, 2023.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
