Nitric acid
| Nitric acid | |
|---|---|
| |
| General | |
| Systematic name | Nitric acid |
| Other names | Aqua fortis Spirit of nitre Salpetre acid |
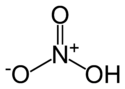
| Molecular formula | HNO3 |
| SMILES | [N+](=O)(OH)[O-] |
| Acidic Ion Concentration | pH = -2 (1 N) |
| Molar mass | 63.01 g/mol |
| Appearance | Clear, colorless liquid |
| CAS number | 7697-37-2 |
| Properties | |
| Density and phase | 1.51 g/cm³ |
| Solubility in water | miscible |
| Melting point | -42°C (231 K) |
| Boiling point | 83°C (356 K) |
| Acidity (pKa) | -2 |
| Viscosity | ? cP at ? °C |
| Structure | |
| Molecular shape | trigonal planar |
| Dipole moment | ? D |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Oxidant (O) Corrosive (C) |
| NFPA 704 (≤40%) | |
| NFPA 704 (> 40%) | |
| NFPA 704 (fuming) | |
| R-phrases | R8, R35 |
| S-phrases | S1/2, S23, S26, S36, S45 |
| Flash point | not applicable |
| RTECS number | QU5775000 |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Related compounds | Nitrous acid Dinitrogen pentoxide |
| Except where noted otherwise, data are given for materials in their standard state (at 25°C, 100 kPa) | |
Nitric acid (chemical formula HNO3) is one of the most important inorganic acids. Eighth-century alchemists called it aqua fortis (strong water), aqua valens (powerful water), or spirit of nitre. It is a highly corrosive and toxic acid that can cause severe burns. Colorless when pure, older samples tend to acquire a yellow cast due to the accumulation of oxides of nitrogen. Nitric acid is miscible in water in all proportions, forming hydrates at low temperature.
This acid is a common laboratory reagent and an important industrial commodity. It is mainly used in making ammonium nitrate (NH4NO3) for fertilizers. It is also used for the manufacture of explosives (such as nitroglycerine), nitrocotton or guncotton, plastics, and dyes.
History
The earliest known written description of the method of synthesizing nitric acid is credited to the alchemist Jabir ibn Hayyan (Geber). It says:
Take a pound of Cyprus vitriol, a pound and a half of Saltpetre, and a quarter of a pound of alum. Submit the whole to distillation, in order to withdraw a liquor which has a high solvent action. The dissolving power of the acid is greatly augmented if it be mixed with some sal ammoniac, for it will then dissolve gold, silver and sulphur.[1]
Later a Dutch chemist, Johann Rudolf Glauber, was the first to make nitric acid by distillation of saltpeter with sulfuric acid, or oil of vitriol as he called it. The product (sodium sulfate decahydrate) is named “Glauber’s salt” to commemorate him.
Aqua regia (Latin for "royal water") is one of the chemicals the ancient scientists concocted. It is a highly corrosive, fuming yellow or red solution. The mixture is formed by mixing concentrated nitric and hydrochloric acid, usually in a volumetric ratio of one to three. It is one of the few reagents that can dissolve gold and platinum, the so-called royal, or noble metals—hence the name “royal water.” The effectiveness of aqua regia is partly because of the presence of both chlorine and nitrosyl chloride. Aqua regia is used in etching and certain analytical processes, and in laboratories to clean glassware of organic and metallic compounds.
Physical properties
Laboratory reagent nitric acid contains only 68 percent HNO by weight. This concentration corresponds to the constant boiling mixture of HNO3 with water, which has the atmospheric pressure of 68.4 percent by weight, boiling at 121.9°C. Pure anhydrous nitric acid (100 percent) is a colorless liquid with a density of 1522 kg/m3 at 25°C, which solidifies at -41.6°C to form white crystals and boils at 86°C. When boiling in light, even at room temperature, there is a partial decomposition with the formation of nitrogen dioxide following the reaction:
- 4HNO3 → 2H2O + 4NO2 + O2 (72°C)
which means that anhydrous nitric acid should be stored below 0°C to avoid decomposition. The nitrogen dioxide (NO2) remains dissolved in the nitric acid, coloring it yellow, or red at higher temperatures. While the pure acid tends to give off white fumes when exposed to air, acid with dissolved nitrogen dioxide gives off reddish-brown vapors, leading to the common name "red fuming acid" or "fuming nitric acid."
- Nitric acid is miscible with water in all proportions and distillation gives an azeotrope with a concentration of 68 percent HNO3 and a boiling temperature of 120.5°C at 1 atm. Two solid hydrates are known: The monohydrate (HNO3.H2O) and the trihydrate (HNO3.3H2O).
- Nitrogen oxides (NOx) are soluble in nitric acid and this property influences more or less all the physical characteristics depending on the concentration of the oxides. These mainly include the vapor pressure above the liquid and the boiling temperature, as well as the color mentioned above.
- Nitric acid is subject to thermal or light decomposition with increasing concentration and this may give rise to some non-negligible variations in the vapor pressure above the liquid because the nitrogen oxides produced dissolve partly or completely in the acid.
Chemical properties
Nitric acid is formed by the reaction of both dinitrogen pentoxide (N2O3) and nitrogen dioxide (NO2) with water. If the solution contains more than 86 percent nitric acid, it is referred to as fuming nitric acid. Fuming nitric acid is characterized as white fuming nitric acid and red fuming nitric acid, depending on the amount of nitrogen dioxide present.
Nitric acid is a strong, monobasic acid, a powerful oxidizing agent which also nitrates many organic compounds, and a monoprotic acid because there is only one dissociation.
Acidic properties
Being a typical acid, nitric acid reacts with alkalis, basic oxides, and carbonates to form salts, the most important of which is ammonium nitrate. Due to its oxidizing nature, nitric acid does not (with some exceptions) liberate hydrogen on reaction with metals and the resulting salts are usually in the higher oxidized state. For this reason, heavy corrosion can be expected and should be guarded against by the appropriate use of corrosion-resistant metals or alloys.
Nitric acid is a strong acid with an acid dissociation constant (pKa) of −2: in aqueous solution, it completely ionizes into the nitrate ion NO3− and a hydrated proton, known as a hydronium ion, H3O+.
- HNO3 + H2O → H3O+ + NO3-
Oxidizing properties
Nitric acid is a strong oxidizing agent as shown by its large positive E◦ values.
- NO3- (aq) + 2H+ (aq) e- → NO2 (g) + H2O (l) E◦ = 0.79 V
- NO3- (aq) + 4H+ + 3e- → NO (g) 2H2 (l) E◦ = 0.96 V
Being a powerful oxidizing agent, nitric acid reacts violently with many non-metallic compounds and the reactions may be explosive. Depending on the acid concentration, temperature and the reducing agent involved, the end products can be variable. Reaction takes place with all metals except the precious metal series and certain alloys. As a general rule, oxidizing reactions occur primarily with the concentrated acid, favoring the formation of nitrogen dioxide (NO2).
Reactions with metals
Nitric acid dissolves most metals including iron, copper, and silver, with generally the liberation of lower oxides of nitrogen rather than hydrogen. It can also dissolve the noble metals with the addition of hydrochloric acid.
- Cu + 4HNO3 → Cu(NO3)2 + 2NO2 + 2H2O
The acidic properties tend to dominate with dilute acid, coupled with the preferential formation of nitrogen oxide (NO).
- 3Cu + 8HNO3 → 3Cu(NO3)2 + 2NO + 4H2O
Since nitric acid is an oxidizing agent, hydrogen (H) is rarely formed. Only magnesium (Mg) and calcium (Ca) react with cold, dilute nitric acid to give hydrogen:
- Mg(s) + 2HNO3 (aq) → Mg(NO3)2 (aq) + H2 (g)
Reactions with nonmetals
Reaction with non-metallic elements, with the exception of silicon and halogen, usually oxidizes them to their highest oxidation states as acids with the formation of nitrogen dioxide for concentrated acid and nitrogen oxide for dilute acid.
- C + 4HNO3 → CO2 + 4NO2 + 2H2O
or
- 3C + 4HNO3 → 3CO2 + 4NO + 2H2O
Passivation
Although chromium (Cr), iron (Fe), and aluminum (Al) readily dissolve in dilute nitric acid, the concentrated acid forms a metal oxide layer that protects the metal from further oxidation, which is called passivation.
Synthesis and production
Nitric acid is made by mixing nitrogen dioxide (NO2) with water in the presence of oxygen or air to oxidize the nitrous acid also produced by the reaction. Dilute nitric acid may be concentrated by distillation up to 68 percent acid, which is an azeotropic mixture with 32 percent water. Further concentration involves distillation with sulfuric acid which acts as a dehydrating agent. On a laboratory scale, such distillation must be done in all glass apparatus at reduced pressure, to prevent decomposition of the acid. Rubber and cork fittings should also be avoided as nitric acid attacks these materials. Commercial grade nitric acid solutions are usually between 52 percent and 68 percent nitric acid. Commercial production of nitric acid is via the Ostwald process, named after Wilhelm Ostwald.
The first process is a gas phase catalytic reaction—the primary process of oxidation of ammonia to nitric acid at around 900°C over a platinum-rhodium catalyst.
- 4 NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (g)
The second step is the rapid oxidation of nitric oxide to nitrogen dioxide. This is a relatively slow reaction, i.e., the rate-determining step in the sequence of reactions.
- 2NO (g) + O,2 (g) → 2NO2 (g)
Finally the disproportionation of NO2 in water gives one molecule of nitric oxide for every two molecules of nitric acid.
- 3NO2 (g) + H2O (l) → 2HNO3 (aq) + NO (g)
Further removal of water is required to obtain pure nitric acid that is colorless and boils at 83°C.
In the laboratory, nitric acid can be made from Copper(II) nitrate or by reacting approximately equal masses of potassium nitrate (KNO3) with 96 percent sulfuric acid (H2SO4), and distilling this mixture at nitric acid's boiling point of 83°C until only a white crystalline mass, potassium hydrogen sulfate (KHSO4), remains in the reaction vessel. The obtained red fuming nitric acid may be converted to the white nitric acid. Note that in a laboratory setting, it is necessary to use all-glass equipment, ideally a one-piece retort, because anhydrous nitric acid attacks cork, rubber, and skin, and leaks can be extremely dangerous.
- H2SO4 + KNO3 → KHSO4 + HNO3
The dissolved NOx are readily removed using reduced pressure at room temperature (10-30 min. at 200 mm Hg or 27 kPa). Obtained white fuming nitric acid has a density of 1.51 g/cm³. This procedure can also be performed under reduced pressure and temperature in one step in order to produce less nitrogen dioxide gas.
The acid can also be synthesized by oxidizing ammonia, but the product is diluted by the water also formed as part of the reaction. However, this method is important in producing ammonium nitrate from ammonia derived from the Haber process, because the final product can be produced from nitrogen, hydrogen, and oxygen as the sole feedstocks.
White fuming nitric acid, also called 100 percent nitric acid or WFNA, is very close to the anhydrous nitric acid product. One specification for white fuming nitric acid is that it has a maximum of 2 percent water and a maximum of 0.5 dissolved NO2. Red fuming nitric acid, or RFNA, contains substantial quantities of dissolved nitrogen dioxide (NO2), leaving the solution with a reddish-brown color. One formulation of RFNA specifies a minimum of 17 percent NO2, another specifies 13 percent NO2. In either event, an inhibited fuming nitric acid (either IWFNA, or IRFNA) can be made by the addition of 0.6 to 0.7 percent hydrogen fluoride, HF. This fluoride is added for corrosion resistance in metal tanks (the fluoride creates a metal fluoride layer that protects the metal).
Uses
Commonly used as a laboratory reagent, nitric acid is used in the manufacture of explosives including nitroglycerin, trinitrotoluene (TNT), and cyclotrimethylenetrinitramine (RDX), as well as fertilizers such as ammonium nitrate.
Also, in ICP-MS and ICP-AES techniques, nitric acid (with a concentration from 0.5 percent to 2.0 percent) is used as a matrix compound for determining metal traces in solutions. Ultrapure acid is required for such determination, because small amounts of metal ions could affect the result of the analysis.
It has additional uses in metallurgy and refining as it reacts with most metals, and in organic syntheses. When combined with hydrochloric acid, it forms aqua regia, one of the few reagents capable of dissolving gold and platinum.
Nitric acid is a component of acid rain.
Nitric acid is a powerful oxidizing agent, and the reactions of nitric acid with compounds such as cyanides, carbides, and metallic powders can be explosive. Reactions of nitric acid with many organic compounds, such as turpentine, are violent and hypergolic (i.e., self-igniting).
Concentrated nitric acid dyes human skin yellow due to a reaction with the protein keratin. These yellow stains turn orange when neutralized.
One use for IWFNA is as an oxidizer in liquid fuel rockets.
One use for nitric acid is in a colorimetric test to distinguish heroin and morphine.
Nitric acid is also used in school laboratories to perform experiments involving the testing of chloride. The sample is added with silver nitrate solution and nitric acid to see if a white precipitate, silver chloride, remains.
Safety precautions
Nitric acid is a dangerous chemical and should be handled with regard to its corrosive and oxidizing properties. Avoid contact with acid and use protective equipment, most particularly eye protection. If spilled on the skin, it can cause yellow discoloration, and larger quantities or concentrations can cause fatal burns. Do not breathe fumes given off when mixed with metals or organic compounds—the effects may be delayed but still fatal. Stay away from any red-brown fumes! Nitric acid does not itself burn, but it oxidizes organic matter and makes it highly flammable.
Related topics
Notes
- ↑ Thomas H. Chilton, Strong Water; Nitric Acid: Sources, Methods of Manufacture, and Uses (Cambridge, MA: M.I.T. Press, 1968). OCLC 237255.
ReferencesISBN links support NWE through referral fees
- Chilton, Thomas H. 1968. Strong Water; Nitric Acid: Sources, Methods of Manufacture, and Uses. Cambridge, MA: M.I.T. Press. OCLC 237255.
- Corwin, C. H. 2001. Introductory Chemistry Concepts and Connections. 3rd ed. Upper Saddle River, NJ: Prentice Hall. ISBN 0130874701.
- Federmann, R. 1964. The Royal Art of Alchemy. Trans. R. H. Weber. New York: Chilton Book. ASIN B000J3UZJ4.
- Jolly, W. L. 1966. The Chemistry of the Non-Metals. Foundation of Modern Chemistry Series. Englewood Cliffs, NJ: Prentice Hall. ASIN B0006BNQ1I.
- McMurry, J., and Fay, R. C. 2004. Chemistry. 4th ed. Upper Saddle River, NJ: Prentice Hall. ISBN 0131402080.
External links
All links retrieved June 30, 2025.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
