Krill
| Euphausiacea | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
 A northern krill (Meganyctiphanes norvegica)
| ||||||||||||
| Scientific classification | ||||||||||||
| ||||||||||||
|
Krill (singular and plural) or euphausiids are small, shrimp-like marine crustaceans that belong to the order (or suborder) Euphausiacea. These small invertebrates are important members of the zooplankton, particularly as food for baleen whales, manta rays, whale sharks, crabeater seals and other pinnipeds, and a few seabird species that feed almost exclusively on them. The name krill also may be used in a limited sense to refer to the genus Euphausia or even just the species Euphausia superba.
As this taxonomic group advances in its individual need of growth, maintenance, and survival, it also fulfills a larger purpose for the ecosystem and for humans. Krill are considered keystone species near the bottom of the food chain because they feed on phytoplankton and to a lesser extent other zooplankton, converting these into a form suitable for many larger animals for whom krill makes up the largest part of their diet. In the Southern Ocean (or Antarctic Ocean), one species, the Antarctic Krill Euphausia superba, makes up a biomass of hundreds of millions of tons, similar to the entire human consumption of animal protein. Over half of this biomass is eaten by whales (such as blue whales and fin whales), seals, penguins, squid, and fish each year, and replaced by growth and reproduction. Most of the species display large daily vertical migrations making a significant amount of biomass available as food for predators near the surface at night and in deeper waters during the day.
In providing for a very rich ecosystem, these small creatures also add to the aesthetic pleasure of humans, who delight in sightings of whales, seals and other marine life. But krill also provide direct economic benefit for people. Commercial fishing of krill is done in the Southern Ocean and in the waters around Japan. The total global production amounts to 150 – 200,000 metric tons annually, most of this from the Scotia Sea. Most krill is used for aquaculture and aquarium feeds, as bait in sport fishing, or in the pharmaceutical industry. In Japan and Russia, krill is also used for human consumption and known as okiami in Japan.
The name krill comes from the Norwegian word krill meaning “young fry of fish.”
Distribution
Krill occur worldwide in all oceans. Most species have transoceanic distribution and several species have endemic or neritic restricted distribution. Species of the genus Thysanoessa occur in both the Atlantic and the Pacific Ocean, which is also home to Euphausia pacifica. Northern krill occurs across the Atlantic, from the north to the Mediterranean Sea. The four species of the genus Nyctiphanes are highly abundant along the upwelling regions of the California, Humbolt, Benguela, and Canarias Current Systems, where occur most of the largest fisheries' activities of fish, mollusks, and crustaceans.
In the Antarctic, seven species are known (Brueggeman 2007): one of the genus Thysanoessa (T. macrura) and six species of the genus Euphausia. The Antarctic krill (Euphausia superba) commonly lives at depths up to 100 meters (MarineBio 2007), whereas ice krill (Euphausia crystallorophias) has been recorded at a depth of 4,000 meters, but commonly lives in depths at most 300 to 600 meters deep (Kirkwood 1984). Both are found at latitudes south of 55° S; with E. crystallorophias dominating south of 74° S (Sala et al. 2002) and in regions of pack ice. Other species known in the Southern Ocean are E. frigida, E. longirostris, E. triacantha, and E. vallentini (Hosie et al. 2003).
Taxonomy
Crustacea, the taxonomic unit to which krill belong, is generally considered a subphylum of Arthropoda, but sometimes is considered a class or superclass. One of the divisions of Crustacea is the class (or order) Malacostraca, which includes the krill. This is the largest class of crustaceans, with such familiar animals as crabs, lobsters, shrimp, krill and woodlice. The order Euphausiacea (or suborder, depending on whether Malacostraca is treated as a class or order) comprises the krill.
Euphausiacea is split into two families. The family Bentheuphausiidae has only one species, Bentheuphausia amblyops, a bathypelagic krill living in deep waters below 1,000 meters. It is considered the most primitive living species of all krill (Brinton 1962). The other family—the Euphausiidae—contains ten different genera with a total of about 85 known species. Of these, the genus Euphausia is the largest, with 31 species (ITIS 2007).
Well-known species—mainly because they are subject to commercial krill fishery—include Antarctic krill (Euphausia superba), Pacific krill (Euphausia pacifica), and Northern krill (Meganyctiphanes norvegica).
Anatomy and morphology
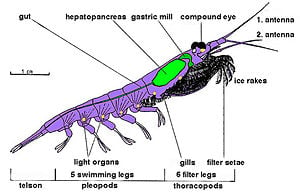
Krill are crustaceans and as such they have a chitinous exoskeleton made up of three segments: the cephalon (head), thorax, and the abdomen. In krill, the first two segments are fused into one segment, the cephalothorax. This outer shell of krill is transparent in most species.
Most krill are about 1 to 2 centimeters long as adults, but a few species grow to sizes of the order of 6 to 15 centimeters. The largest krill species is the mesopelagic Thysanopoda spinicauda (Brinton 1953). Krill can be easily distinguished from other crustaceans, such as true shrimps, by their externally visible gills.
As with other crustaceans, krill have compound eyes and two antennae. Krill eyes are intricate; some species can adapt to different lighting conditions through the use of screening pigments (Gaten 2006).
Krill have several pairs of thoracic legs called pereiopods or thoracopods (so named because they are attached to the thorax; their number varies among genera and species). These thoracic legs include the feeding legs and the grooming legs. Additionally, all species have five swimming legs called pleopods or "swimmerets," very similar to those of the common freshwater lobster.
Many krill are filter feeders: their front-most extremities, the thoracopods, form very fine combs with which they can filter out their food from the water. These filters can be very fine indeed in those species (such as Euphausia spp.) that feed primarily on phytoplankton, in particular on diatoms, which are unicellular algae. However, it is believed that all krill species are mostly omnivorous (feeding on both plants and animals) and some few species are carnivorous, preying on small zooplankton and fish larvae.
Except for the Bentheuphausia amblyops species, krill are bioluminescent animals, having organs called photophore that are able to emit light. The light is generated by an enzyme-catalyzed chemiluminescence reaction, wherein a luciferin (a kind of pigment) is activated by a luciferase enzyme. Studies indicate that the luciferin of many krill species is a fluorescent tetrapyrrole similar but not identical to dinoflagellate luciferin (Shimomura 1995) and that the krill probably do not produce this substance themselves but acquire it as part of their diet that contains dinoflagellates (Dunlap et al. 1980). Krill photophores are complex organs with lenses and focusing abilities, and they can be rotated by muscles (Herring and Widder 2001).
The precise function of the photophore organs is as yet unknown. They might have a purpose in mating, social interaction, or in orientation. Some researchers (e.g. Lindsay and Latz (1999) or Johnsen (2005) have proposed that krill use the light as a form of counter-illumination camouflage to compensate their shadow against the ambient light from above to make themselves more difficult to be seen by predators from below.
Behavior
Most krill are swarming animals; the size and density of such swarms vary greatly depending on the species and the region. Of Euphausia superba, there have been reports of swarms of up to 10,000 to 30,000 individuals per cubic meter (Kils and Marshall 1995). Swarming is considered to be a defensive mechanism, confusing smaller predators that would like to pick out single individuals.
Krill typically follow a diurnal vertical migration. They spend the day at greater depths and rise during the night towards the surface. The deeper they go, the more they reduce their activity (Jaffe et al. 1999), apparently to reduce encounters with predators and to conserve energy. Some species (e.g. Euphausia superba, E. pacifica, E. hanseni, Pseudeuphausia latifrons, or Thysanoessa spinifera) also form surface swarms during the day for feeding and reproductive purposes even though such behavior is dangerous because it makes them extremely vulnerable to predators.
Dense swarms may elicit a feeding frenzy among predators such as fish or birds, especially near the surface, where escape possibilities for the krill are limited. When disturbed, a swarm scatters, and some individuals have even been observed to molt instantaneously, leaving the exuvia (shed exoskeleton) behind as a decoy (Howard 2005).
Krill normally swim at a pace of a few centimeters per second (0.2 – 10 body lengths per second) (Ignatyev 1999), using their swimmerets for propulsion. Their larger migrations are subject to the currents in the ocean. When in danger, they show an escape reaction called lobstering: flipping their caudal appendages, i.e., the telson and uropods, they move backwards through the water relatively quickly, achieving speeds in the range of 10 to 27 body lengths per second (Ignatyev 1999), which for large krill such as E. superba means around 0.8 meters per second (Kils 1982). Their swimming performance has led many researchers to classify adult krill as micro-nektonic lifeforms, i.e., small animals capable of individual motion against (weak) currents. Larval forms of krill are generally considered [plankton|zooplankton]] (Nicol and Endo. 1997).
Ecology and life history

Krill are an important element of the food chain. Antarctic krill feed directly on phytoplankton, converting the primary production energy into a form suitable for consumption by larger animals that cannot feed directly on the minuscule algae, but that can feed upon krill. Some species like the Northern krill have a smaller feeding basket and hunt for copepods and larger zooplankton. Many other animals feed on krill, ranging from smaller animals like fish or penguins to larger ones like seals and even baleen whales.
Disturbances of an ecosystem resulting in a decline of the krill population can have far-reaching effects. During a coccolithophore bloom in the Bering Sea in 1998 (Weier 1999), for instance, the diatom concentration dropped in the affected area. However, krill cannot feed on the smaller coccolithophores, and consequently the krill population (mainly E. pacifica) in that region declined sharply. This in turn affected other species: the shearwater population dropped, and the incident was even thought to have been a reason for salmon not returning to the rivers of western Alaska in that season (Brodeur et al. 1998).
Other factors besides predators and food availability also can influence the mortality rate in krill populations. There are several single-celled endoparasitoidic ciliates of the genus Collinia that can infect different species of krill and cause mass dying in affected populations. Such diseases have been reported for Thysanoessa inermis in the Bering Sea, but also for E. pacifica, Thysanoessa spinifera, and T. gregaria off the North-American Pacific coast (Roach 2003). There are also some ectoparasites of the family Dajidae (epicaridean isopods) that afflict krill (and also shrimps and mysids). One such parasite is Oculophryxus bicaulis, which has been found on the krill Stylocheiron affine and S. longicorne. It attaches itself to the eyestalk of the animal and sucks blood from its head; it is believed that it inhibits the reproduction of its host as none of the afflicted animals found reached maturity (Shields and Gómez-Gutiérrez 1996).
Life history
The general life-cycle of krill has been the subject of several studies (e.g., Guerny 1942 (Gurney 1942) or Mauchline and Fisher (1969) performed on a variety of species and is thus relatively well understood, although there are minor variations in details from species to species. In common with other crustaceans, krill have a stiff exoskeleton, which must be shed to allow the animal to grow (ecdysis). Despite the diversity of forms, crustaceans, including krill, are united by the special larval form known as the nauplius.
When krill hatch from the eggs, they go through several larval stages called the nauplius, pseudometanauplius, metanauplius, calyptopsis, and furcilia stages, each of which is sub-divided into several sub-stages. The pseudometanauplius stage is exclusive of species that lay their eggs within an ovigerous sac (so-called sac-spawners). The larvae grow and molt multiple times during this process, shedding their rigid exoskeleton and growing a new one whenever it becomes too small. Smaller animals molt more frequently than larger ones. Until and including the metanauplius stage, the larvae nourish on yolk reserves within their body. Only by the calyptopsis stages, differentiation has progressed far enough for them to develop a mouth and a digestive tract, and they begin to feed upon phytoplankton. By that time, the larvae must have reached the photic zone, the upper layers of the ocean where algae flourish, for their yolk reserves are exhausted by then and they would starve otherwise.
During the furcilia stages, segments with pairs of swimmerets are added, beginning at the frontmost segments. Each new pair becomes functional only at the next molt. The number of segments added during any one of the furcilia stages may vary even within one species depending on environmental conditions (Knight 1984).
After the final furcilia stage, the krill emerges in a shape similar to an adult, but is still immature. During the mating season, which varies depending on the species and the climate, the male deposits a sperm package at the genital opening (named thelycum) of the female. The females can carry several thousand eggs in their ovary, which may then account for as much as one third of the animal's body mass (Ross and Quetin 1986). Krill can have multiple broods in one season, with interbrood periods of the order of days.
There are two types of spawning mechanisms (Gómez-Gutiérrez 2002a). The 57 species of the genera Bentheuphausia, Euphausia, Meganyctiphanes, Thysanoessa, and Thysanopoda are "broadcast spawners": the female eventually just releases the fertilized eggs into the water, where they usually sink into deeper waters, disperse, and are on their own. These species generally hatch in the nauplius 1 stage, but recently have been discovered to hatch sometimes as metanauplius or even as calyptopis stages (Gómez-Gutiérrez 2002b). The remaining 29 species of the other genera are “sac spawners,” where the female carries the eggs with her attached to its rearmost pairs of thoracopods until they hatch as metanauplii, although some species like Nematoscelis difficilis may hatch as nauplius or pseudometanauplius (Brinton et al. 2000).
Some high latitude species of krill can live up to more than six years (e.g., Euphausia superba). Others, such as the as mid-latitude species Euphausia pacifica, live only for two years (Nicol and Endo 1997). Subtropical or tropical species' longevity is still smaller, like e.g., Nyctiphanes simplex that usually lives only for six to eight months (Gómez-Gutiérrez 2007).
Molting occurs whenever the animal outgrows its rigid exoskeleton. Young animals, growing faster, therefore molt more often than older and larger ones. The frequency of molting varies wildly from species to species and is, even within one species, subject to many external factors such as the latitude, the water temperature, or the availability of food. The subtropical species Nyctiphanes simplex, for instance, has an overall intermolt period in the range of two to seven days: larvae molt on the average every three days, while juveniles and adults do so on the average every five days. For E. superba in the Antarctic sea, intermolt periods ranging between 9 and 28 days depending on the temperature between -1°C to 4°C have been observed, and for Meganyctiphanes norvegica in the North Sea the intermolt periods range also from 9 and 28 days but at temperatures between 2.5°C to 15°C (Buchholz 2003. E. superba is known to be able to reduce its body size when there is not enough food available, molting also when its exoskeleton becomes too large (Shin and Nicol 2002). Similar shrinkage has also been observed for E. pacifica (a species occurring in the Pacific Ocean from polar to temperate zones) as an adaptation to abnormally high water temperatures, and has been postulated for other temperate species of krill, too (Marinovic and Mangel 1999).
Economy
Krill has been harvested as a food source for both humans (okiami) and their domesticated animals since the 19th century, in Japan maybe even earlier. Large-scale fishing developed only in the late 1960s and early 1970s, and now occurs only in Antarctic waters and in the seas around Japan. Historically, the largest krill fishery nations were Japan and the Soviet Union, or, after the latter's dissolution, Russia and Ukraine.
A peak in krill harvest had been reached in 1983 with more than 528,000 metric tons in the Southern Ocean alone (of which the Soviet Union produced 93 percent). In 1993, two events led to a drastic decline in krill production: first, Russia abandoned its operations, and second, the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR) defined maximum catch quotas for a sustainable exploitation of Antarctic krill. Nowadays, the largest krill fishing nations in the Antarctic are Japan, followed by South Korea, Ukraine, and Poland (Nicol and Endo 1997). The annual catch in Antarctic waters seems to have stabilized around 100,000 tons of krill, which is roughly one fiftieth of the catch quota (CCAMLR 2005). The main limiting factor is probably the high cost associated with Antarctic operations. The fishery around Japan appears to have saturated at some 70,000 tons (Nicol and Foster 2003).
Experimental small-scale harvesting is being carried out in other areas also, for example fishing for Euphausia pacifica off British Columbia or harvesting Meganyctiphanes norvegica, Thysanoessa raschii, and Thysanoessa inermis in the Gulf of St. Lawrence. These experimental operations produce only a few hundred tons of krill per year. Nicol and Foster (2003) consider it unlikely that any new large-scale harvesting operations in these areas will be started due to the opposition from local fishing industries and conservation groups.
Krill taste salty and somewhat stronger than shrimp. For mass-consumption and commercially prepared products, they must be peeled because their exoskeleton contains fluorides, which are toxic in high concentrations (Haberman 1997). Excessive intake of okiami may cause diarrhea.
ReferencesISBN links support NWE through referral fees
- Brinton, E. 1953. Thysanopoda spinicauda, a new bathypelagic giant euphausiid crustacean, with comparative notes on T. cornuta and T. egregia. J. Wash. Acad. Sci. 43: 408-412.
- Brinton, E. 1962. The distribution of Pacific euphausiids. Bull. Scripps Inst. Oceanogr. 8(2): 51-270.
- Brinton, E., M. D. Ohman, A. W. Townsend, M. D. Knight, and A. L. Bridgeman. 2000. Euphausiids of the world ocean. World Biodiversity Database CD-ROM Series. Springer Verlag. ISBN 3540146733.
- Brodeur, R.D., G. H. Kruse, et al. 1998. Draft Report of the FOCI International Workshop on Recent Conditions in the Bering Sea. NOAA.
- Brueggeman, P. 2007. Euphausia crystallorophias. Underwater Field Guide to Ross Island & McMurdo Sound, Antarctica. Retrieved April 23, 2007.
- Buchholz, F. 2003. Experiments on the physiology of Southern and Northern krill, Euphausia superba and Meganyctiphanes norvegica, with emphasis on moult and growth: A review. Marine and Freshwater Behaviour and Physiology 36(4): 229-247. Retrieved April 23, 2007.
- Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR). 2005. Harvested species: Krill (Eupausia superba). CCAMLR. Retrieved June 20, 2005.
- Dunlap J. C., J. W. Hastings, and O. Shimomura. 1980. Crossreactivity between the light-emitting systems of distantly related organisms: Novel type of light-emitting compound. Proc. Natl. Acad. Sci. USA 77(3): 1394-1397. Retrieved April 23, 2007.
- Gaten, E. 2006. Meganyctiphanes norvegica. Department of Biology, University of Leichester.
- Gómez-Gutiérrez, J. 2002a. Euphausiid reproduction rate. College of Oceanic and Atmospheric Sciences Oregon State University. Personal communication. Retrieved April 23, 2007.
- Gómez-Gutiérrez, J. 2002b. Hatching mechanism and delayed hatching of the eggs of three broadcast spawning euphausiid species under laboratory conditions]. J. of Plankton Research 24(12): 1265-1276.
- Gómez-Gutiérrez, J. 2007 Euphausiids. Oregon State University. Retrieved June 16, 2005.
- Gómez-Gutiérrez, J., W. T. Peterson, A. De Robertis, and R. D. Brodeur. 2003. Mass mortality of krill caused by parasitoid ciliates. Science 301(5631): 339f.
- Gurney, R. 1942. Larvae of decapod crustacea. Royal Society Publ. 129.
- Haberman, K. 1997. Answers to miscellaneous questions about krill. NASA Quest, February 26, 1997. Retrieved June 17, 2005.
- Howard, D. 2005. Krill in Cordell Bank National Marine Sanctuary. NOAA. Retrieved June 15, 2005.
- Hosie, G. W., M. Fukuchi, and S. Kawaguchi. 2003. Development of the Southern Ocean Continuous Plankton Recorder survey. Progress in Oceanography 58: 263-283. Retrieved April 23, 2007.
- Herring, P. J., and E. A. Widder. 2001. Bioluminescence in plankton and nekton. In J. H. Steele, S. A. Thorpe, and K. K. Turekian, eds., Encyclopedia of Ocean Science 1: 308-317. San Diego: Academic Press. Retrieved April 24, 2007.
- Ignatyev, S. M. 1999. Functional-morphological adaptations of the krill to active swimming]. Poster on the 2nd International Symposium on Krill, Santa Cruz, CA, August 23-27, 1999.
- ITIS. 2007. Taxonomy of Euphausiacea. ITIS Report. Retrieved April 24, 2007.
- Jaffe, J. S., M. D. Ohmann, and A. De Robertis. 1999. Sonar estimates of daytime activity levels of Euphausia pacifica in Saanich Inlet. Can. J. Fish. Aquat. Sci. 56: 2000-2010. Retrieved April 24, 2007.
- Johnsen, S. 2005. The red and the black: Bioluminescence and the color of animals in the deep sea. Integr. Comp. Biol. 45: 234-246. Retrieved April 24, 2007.
- Kils, U. 1982. Swimming behavior, Swimming Performance and Energy Balance of Antarctic Krill Euphausia superba. BIOMASS Scientific Series 3, BIOMASS Research Series, 1-122.
- Kils, U., and P. Marshall. 1995. Der Krill, wie er schwimmt und frisst - neue Einsichten mit neuen Methoden ("The Antarctic krill - feeding and swimming performances - new insights with new methods"). Pages 201-210 in I. Hempel and G. Hempel. Biologie der Polarmeere - Erlebnisse und Ergebnisse (Biology of the Polar Oceans). Fischer. ISBN 3334609502.
- Kirkwood, J. A. 1984. A Guide to the Euphausiacea of the Southern Ocean. Australian National Antarctic Research Expedition; Australia Dept of Science and Technology, Antarctic Division.
- Knight, M. D. 1984. [1] Variation in Larval Morphogenesis within the Southern California Bight Population of Euphausia pacifica from Winter through Summer, 1977-1978. CalCOFI Report XXV. Retrieved April 24, 2007.
- Lindsay, S. M., and M. I. Latz. 1999. Experimental evidence for luminescent countershading by some euphausiid crustaceans. Poster presented at the American Society of Limnology and Oceanography (ASLO) Aquatic Sciences Meeting, Santa Fe. Retrieved April 23, 2007.
- MarineBio. 2007. Euphausia superba Krill. MarineBio.org. Retrieved April 24, 2007.
- Mauchline, J., and L. R. Fisher. 1969. The biology of euphausiids. Adv. Mar. Biol. 7.
- Marinovic, B., and M. Mangel. Krill can shrink as an ecological adaptation to temporarily unfavourable environments. Ecology Letters 2: 338-343. Retrieved April 24, 2007.
- Nicol, S., and Y. Endo. 1997. Krill fisheries of the world. FAO Fisheries Technical Paper 367. Retrieved April 24, 2007.
- Nicol, S., and J. Foster. 2003. Recent trends in the fishery for Antarctic krill. Aquat. Living Resour. 16: 42-45. Retrieved April 24, 2007.
- Roach, J. 2003. Scientists discover mystery krill killer. National Geographic News, July 17, 2003. Retrieved April 24, 2007.
- Ross, R. M., and L. B. Quetin. 1986. How productive are Antarctic krill? Bioscience 36: 264-269.
- Sala, A., M. Azzali, and A. Russo. 2002. Krill of the Ross Sea: Distribution, abundance and demography of Euphausia superba and Euphausia crystallorophias during the Italian Antarctic Expedition (January-February 2000). Scientia Marina 66(2): 123-133. Retrieved April 24, 2007.
- Shields, J. D., and J. Gómez-Gutiérrez. 1996. Oculophryxus bicaulis, a new genus and species of dajid isopod parasitic on the euphausiid Stylocheiron affine Hansen. Int'l J. for Parasitology 26(3): 261-268. Retrieved April 24, 2007.
- Shimomura, O. 1995. The roles of the two highly unstable components F and P involved in the bioluminescence of euphausiid shrimps. Jour. Biolumin. Chemilumin. 10(2): 91-101. Retrieved April 24, 2007.
- Shin, H.-C., and S. Nicol. 2002. Using the relationship between eye diameter and body length to detect the effects of long-term starvation on Antarctic krill Euphausia superba. Mar Ecol Progress Series (MEPS) 239: 157-167. Retrieved April 24, 2007.
- Weier, J. 1999. [http://earthobservatory.nasa.gov/Study/Coccoliths/bering_sea.htmlObservatory. Retrieved June 15, 2005.
Further reading
All links retrieved March 3, 2025.
- Boden, B. P., M. W. Johnson, and E. Brinton. 1955. Euphausiacea (Crustacea) of the North Pacific. Bulletin of the Scripps Institution of Oceanography 6(8).
- Brinton, E. 1962. The distribution of Pacific euphausiids. Bulletin of the Scripps Institution of Oceanography 8(2): 51-269.
- Brinton, E. 1975. Euphausiids of Southeast Asian waters. Naga Report 4 (part 5). La Jolla: University of California, Scripps Institution of Oceanography.
- Everson, I. (ed.). 2000. Krill: Biology, Ecology and Fisheries. Oxford: Blackwell Science. ISBN 0632055650.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.