Difference between revisions of "Cocaine" - New World Encyclopedia
Rick Swarts (talk | contribs) |
Rick Swarts (talk | contribs) |
||
| Line 479: | Line 479: | ||
<ref>{{cite journal |author=Lluch J, Rodríguez-Arias M, Aguilar MA, Miñarro J |title=Role of dopamine and glutamate receptors in cocaine-induced social effects in isolated and grouped male OF1 mice |journal=Pharmacol. Biochem. Behav. |volume=82 |issue=3 |pages=478–87 |year=2005|pmid=16313950 |doi=10.1016/j.pbb.2005.10.003}}</ref> | <ref>{{cite journal |author=Lluch J, Rodríguez-Arias M, Aguilar MA, Miñarro J |title=Role of dopamine and glutamate receptors in cocaine-induced social effects in isolated and grouped male OF1 mice |journal=Pharmacol. Biochem. Behav. |volume=82 |issue=3 |pages=478–87 |year=2005|pmid=16313950 |doi=10.1016/j.pbb.2005.10.003}}</ref> | ||
| + | |||
| + | * Office of Diversion Control (ODC). 2011. [http://www.deaDiversion.usdoj.gov/fed_regs/imprt/reg/2011 Importers Notice of Registration - 2011]. ''Drug Enforcement Agency, U.S. Department of Justice''. Retrieved August 14, 2013. | ||
| + | |||
Cocaine-related sudden death not so rare | Cocaine-related sudden death not so rare | ||
Revision as of 22:44, 11 September 2013
| |
| |
Cocaine
| |
| Systematic name | |
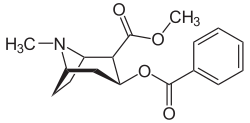
| IUPAC name methyl (1R,2R,3S,5S)-3- (benzoyloxy)-8-methyl-8-azabicyclo[3.2.1] octane-2-carboxylate | |
| Identifiers | |
| CAS number | 50-36-2 |
| ATC code | N01BC01 R02AD03, S01HA01, S02DA02 |
| PubChem | 5760 |
| DrugBank | DB00907 |
| Chemical data | |
| Formula | C17H21NO4 |
| Mol. weight | 303.353 g/mol |
| SMILES | CN1[C@H]2CC[C@@H]1[C@H]([C@H](C2)OC(=O)c3ccccc3)C(=O)OC |
| Synonyms | methylbenzoylecgonine, benzoylmethylecgonine, ecgonine methyl ester benzoate, 2b-Carbomethoxy −3b-benzoyloxy tropane |
| Physical data | |
| Melt. point | 98°C (208°F) |
| Boiling point | 187°C (369°F) |
| Solubility in water | HCl: 1800–2500 mg/mL (20°C) |
| Pharmacokinetic data | |
| Bioavailability | Oral: 33%[1] Insufflated: 60[2]–80%[3] Nasal Spray: 25[4]–43%[1] |
| Metabolism | Hepatic CYP3A4 |
| Half life | 1 hour |
| Excretion | Renal (benzoylecgonine and ecgonine methyl ester) |
| Therapeutic considerations | |
| Pregnancy cat. | C |
| Legal status | ? |
| Dependence Liability | High |
| Routes | Topical, Oral, Insufflation, IV, PO |
Cocaine is a crystalline tropane alkaloid (benzoylmethylecgonine, C17H21NO4) found in the leaves of the coca plant and best known in its concentrated form as an addictive, and generally illegal, psychoactive recreational drug.
Although the amount of cocaine in coca leaves is low, when this alkaloid is chemically extracted and concentrated it results in a powerful nervous system stimulant, which is generally used nasally, smoked, or injected. As such, cocaine can be highly addictive and have deleterious impacts on the brain, heart, respiratory system, kidneys, sexual system, and gastrointestinal tract. In most countries, the production, distribution, sale, and possession of cocaine products is restricted and/or illegal. However, cocaine also has some medical use and in some countries is available by prescription for such purposes as external application to the skin to numb pain, although derivatives such as lidocaine and novocaine have largely replaced it.
Use of concentrated cocaine yields pleasure through its interference with neurotransmitters of the sympathetic nervous system, such as blocking dopamine from being reabsorbed and thus resulting in continual stimulation. Cocaine is one of the most addictive recreational drugs and intense cravings can be created even after one use. In addition to medical problems from the drug, including sudden death, the use of cocaine can create a tolerance, requiring an increasing dose for stimulation, and the user can reach a state in which he or she has difficulty experiencing pleasure without the drug.
There is a huge worldwide market for cocaine. The United Nations Office of Drugs and Crime estimated that in 2009 the US cocaine market was $37 billion and the West and Central European cocaine market was US$ 33 billion.
For the plant, cocaine seems to serve a valuable function as an effective insecticide, limiting damage from herbivorous insects.
Overview
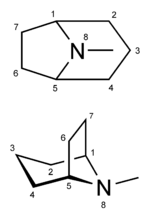
Cocaine is a tropane alkaloid. Tropane alkaloids are a class of alkaloids (naturally occurring chemical compounds that contain mostly basic nitrogen atoms) and secondary metabolites in which the chemical structure includes a tropane ring (nitrogenous bicyclic organic structure). Well-known alkaloids include caffeine, nicotine, morphine, theobromine, mescaline, strychnine, quinine, and codeine. Well-known tropane alkaloids, in addition to cocaine, include atropine and ecgonine (a precursor and metabolite of cocaine). Cocaine has the chemical formula C17H21NO4 and is also known as benzoylmethylecgonine or methyl benzoyl ecgonine.
Cocaine is found in coca plants, which are indigenous to South America. There are four varieties of these tropical plants that are cultivated: Erythroxylum coca var. coca (Bolivian or Huánuco coca), E. coca var. ipadu (Amazonian coca), E. novogranatense var. novogranatense (Colombian coca), and E. novogranatense var. truxillense (Trujillo coca). The name cocaine comes from the name of the coca plant plus the alkaloid suffix -ine.
Cocaine is the most concentrated of the dozen or more alkaloids that have been identified in the coca plant. Concentrations vary by variety and region, but leaves have been reported as having between 0.25% and 0.77% (Plowman and Rivier 1983), between 0.35% and 0.72% by dry weight (Nathanson et al. 1993), and between 0.3% and 1.5% and averaging 0.8% in fresh leaves (Casale and Klein 1993). In unprocessed form, coca leaves have been used for thousands of years in South America for various religious, social, medicinal, and nutritional purposes, including to control hunger and combat the impacts of high altitudes. However, since the alkaloid cocaine is present in only trace amounts in the leaves, it does not cause the euphoric and psychoactive effects associated with use of the drug.
When processed and concentrated by chemical extraction from large quantities of coca leaves, cocaine is a powerful stimulant. The extract from the leaves is hydrolysed and esterified with methanol and benzoic acid to produce the hydrochloride salt of cocaine.
Biologically, cocaine acts as a serotonin–norepinephrine–dopamine reuptake inhibitor, also known as a triple reuptake inhibitor (TRI). For example, Marieb and Hoehn (2010) note the impact of cocaine hooking up to the dopamine reuptake transporter protein, thus blocking reabsorption of dopamine. With this neurotransmitter remaining in the synapse, the post-synaptic receptor cells are stimulated again and again, allowing the body to experience over and over this reward system and associated high, along with increased heart rate, sexual appetite, and blood pressure. However, as a result, the system releases less and less dopamine and the reward system goes dry, and the cocaine user, in addition to becoming anxious, find himself or herself "in a very real sense, unable to experience pleasure without the drug." However, more cocaine just suppresses dopamine release even more.
Unlike most molecules, cocaine has pockets with both high hydrophilic and lipophilic efficiency, violating the rule of hydrophilic-lipophilic balance. This causes it to cross the blood–brain barrier far better than other psychoactive chemicals and may even induce blood-brain barrier breakdown (Sharma et al. 2009; Dietrich 2009). Marieb and Hoehn (2010) note that one way that might be developed to tackle addiction would be to prompt the immune system to bind cocaine molecules and prevent them from entering the brain.
Cocaine is best known worldwide for its illegal use as a recreational drug. This concentrated form of cocaine is used nasally (nasal insufflation is also known as "snorting," "sniffing," or "blowing" and involves absorption through the mucous membranes lining the sinuses), injected (the method that produces the highest blood levels in the shortest time), or smoked (notably the cheaper, more potent form called "crack"). It may also be administered orally (rubbed on gums). Among forms of cocaine use are cocaine hydrochloride, natural leaf, cocaine paste, or freebase.
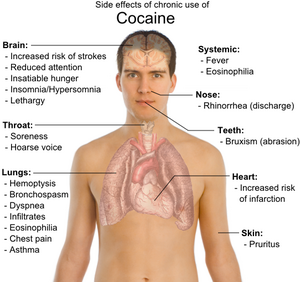
Cocaine use can be highly addictive, causing intense cravings for the drug, and can have deleterious impacts on the brain, heart, respiratory system, kidneys, sexual system, and gastrointestinal tract (WebMD 2013a). For example, it can result in a heart attack or strokes, even in young people,and it can cause ulcers and sudden kidney failure, and it can impair sexual function (WebMD 2013a).
The possession, distribution, and sale of cocaine products is illegal for non-medicinal / non-government sanctioned purposes in virtually all parts of the world. Internationally, it is regulated by the Single Convention on Narcotic Drugs, and the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances. In the United States, the manufacture, importation, possession, and distribution of cocaine is additionally regulated by the 1970 Controlled Substances Act. Cocaine is generally treated as a 'hard drug', with severe penalties for possession and trafficking.
The United Nations Office of Drugs and Crime estimated that in 2009, the US cocaine market was $37 billion (and shrinking over the past ten years) and the West and Central European Cocaine market was US$ 37 billion (and increasing over the past ten years) (USODC 2011).
The coca leaves have been used unprocessed for thousands of years in South America for various religious, social, medicinal, and nutritional purposes, including in the Andean countries to make a herbal tea with mild stimulant effects. However, since the alkaloid cocaine is present in only trace amounts in the leaves, it does not cause the euphoric and psychoactive effects associated with use of the drug. The Coca-Cola company uses a cocaine-free coca extract. In the early days of the manufacture of Coca-Cola beverage, the formulation did contain some cocaine, although within a few years of its introduction it already was only trace amounts. Cocaine is available as a prescription for such purposes as external application to the skin to numb pain.
For the plant, cocaine is believed to serve as a naturally occurring insecticide, with the alkaloid exerting such effects at concentrations normally found in the leaves (Nathanson et. al. 1993). It has been observed that compared to other tropical plants, coca seems to be relatively pest free, with little observed damage to the leaves and rare observations of herbivorous insects on plants in the field (Nathanson et al. 1993).
Medical effects
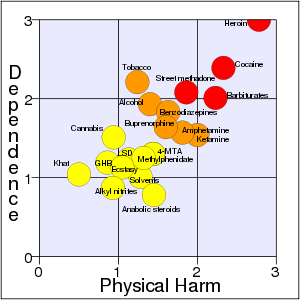
Cocaine acts in the brain on areas that acts to reward people with pleasure for behaviors important to survival individually and as a species, such as food, sex, and healthy pleasure (Marieb and Hoehn 2010; WebMD 2013a; Spanage and Weiss 1999). Involving the brain neurotransmitters in this area, it is a powerful nervous system stimulant (WHO 2004). Its effects can last from 15–30 minutes to an hour or two, depending on dosage and the route of administration (WHO 2007; WebMD 2013a). However, it can have serious negative effects on the heart, brain, lungs, and emotions, including the danger of sudden death (WebMD). It was ranked the second most addictive and harmful recreational drug (of 20 studied) by Nutt et al. (2007), exceeded only by heroin.
On the one hand, users of cocaine report a euphoria (feeling "high"), with an increased sense of alertness, feelings of well-being, competence, and "supremacy," enhanced energy and motor activity, and sexuality (WebMD 2013a).
On the other hand, some users report the high as also being accompanied with anxiety, irritability, paranoia, and restlessness, particularly during the comedown (WebMd 2013a). With excessive dosage or prolonged use, itching, tachycardia, tremors, convulsions, hallucinations, and paranoid delusions can result (WHO 2004; Zhao 2008). Overdoses cause hyperthermia (elevated body temperature) and a marked elevation of blood pressure. Cocaine constricts blood vessels, dilates the pupils, and increases heart rate and blood pressure.
In terms of the circulatory system, the increase of heart rate and blood pressure, while restricting arteries supplying blood, can result in a heart attack, even in youth without heart disease (WebMD 2013a). An abnormal heart rhythm called arrhythmia can be triggered. In terms of the brain, the constriction of blood vessels in the brain can cause strokes, even in young people without other stroke risk factors (WebMD 2013a). Cocaine can double both the risks of hemorrhagic and ischemic strokes (Jeffrey and Vega 2008) and increase the risk of other infarctions, such as myocardial infarction (Vasica and Tennant 2002). Cocaine can cause seizures. Sudden death has been known to occur, such as the case of Len Bias, considered by some as one of the greatest American college basketball athletes, who died two days after being drafted by the Boston Celtics because of a cardiac arrhythmia induced by use of cocaine.
The constriction of blood vessels supplying the gastrointestinal tract can lead to oxygen starvation and the development of ulcers or perforation of the stomach and intestines (WebMD 2013a). Cocaine use can also cause a wide array of kidney diseases and renal failure (Jaffe and Kimmel 2006; van der Woude 2000). Kidney failure can suddenly occur through a process known as rhabdomyolysis (WebMD 2013a).
While sexual appetite may be increased, cocaine use can impair sexual function in men and women, including impaired ejaculation in men (WebMd 2013a).
In terms of the lungs and respiratory system, physical side effects from chronic smoking of cocaine include hemoptysis, bronchospasm, pruritus, fever, diffuse alveolar infiltrates without effusions, pulmonary and systemic eosinophilia, chest pain, lung trauma, sore throat, asthma, hoarse voice, dyspnea (shortness of breath), and an aching, flu-like syndrome. Permanent lung damage can result in some users.
The experience of insatiable hunger, aches, insomnia/oversleeping, lethargy, and persistent runny nose are often described as very unpleasant. Depression with suicidal ideation may develop in very heavy users.
Chronic intranasal usage can degrade the cartilage separating the nostrils (the septum nasi), leading eventually to its complete disappearance. Due to the absorption of the cocaine from cocaine hydrochloride, the remaining hydrochloride forms a dilute hydrochloric acid.[6]
Cocaine may also greatly increase this risk of developing rare autoimmune or connective tissue diseases such as lupus, Goodpasture's disease, vasculitis, glomerulonephritis, Stevens–Johnson syndrome and other diseases (Trozak and Gould 1984; Peces et al. 1999; Moore and Richardson 1998).
Cocaine often is a cause of involuntary tooth grinding, known as bruxism, which can deteriorate tooth enamel and lead to gingivitis (Baigent 2003). Additionally, stimulants like cocaine, methamphetamine, and even caffeine cause dehydration and dry mouth. Since saliva is an important mechanism in maintaining one's oral pH level, chronic stimulant abusers who do not hydrate sufficiently may experience demineralization of their teeth due to the pH of the tooth surface dropping too low (below 5.5).
Chronic cocaine intake causes brain cells to adapt functionally to strong imbalances of transmitter levels in order to compensate extremes. Thus, receptors disappear from the cell surface or reappear on it, resulting more or less in an "off" or "working mode" respectively, or they change their susceptibility for binding partners (ligands) – mechanisms called down-/upregulation. Marieb and Hoehn (2010) state that the blocking of dopamine uptake by repeated use of cocaine causes the reward system to effectively go dry, as the system releases less and less dopamine, and "the cocaine user becomes anxious and, in a very real sense, unable to experience pleasure without the drug." As the postsynaptic cells sprout new receptors to pick up the dopamine signals, a vicious cycle begins where cocaine "is needed to experience pleasure, but using it suppresses dopamine release even more" (Marieb and Hoehn 2010). A loss of vesicular monoamine transporters, neurofilament proteins, and other morphological changes appear to indicate a long term damage of dopamine neurons. All these effects contribute a rise in tolerance thus requiring a larger dosage to achieve the same effect (Lowinson et al. 2004). On the other hand, a study by D'Haenen et al. (2002) suggests cocaine abusers do not show normal age-related loss of striatal dopamine transporter (DAT) sites, suggesting cocaine has neuroprotective properties for dopamine neurons.
Cocaine can often cause reduced food intake, many chronic users lose their appetite and can experience severe malnutrition and significant weight loss.
The lack of normal amounts of serotonin and dopamine in the brain is the cause of the dysphoria and depression felt after the initial high.
Cocaine is extensively metabolized, primarily in the liver, with only about 1% excreted unchanged in the urine. The metabolism is dominated by hydrolytic ester cleavage, so the eliminated metabolites consist mostly of benzoylecgonine (BE), the major metabolite, and other significant metabolites in lesser amounts such as ecgonine methyl ester (EME) and ecgonine. Further minor metabolites of cocaine include norcocaine, p-hydroxycocaine, m-hydroxycocaine, p-hydroxybenzoylecgonine (pOHBE), and m-hydroxybenzoylecgonine (Kolbrich et al. 2006).
Cocaine has been held responsible for more visits to US emergency rooms than any other illegal drug (WebMD 2013a). The amount of sudden-deaths from cocaine also is not a rare phenomenon and in one study the cause of cocaine-related sudden death was found to cardiovascular in 62% of the cases, cerebrovascular in 14%, excited delirium in 14%, respiratory in 5%, and and metabolic in 5% (Nainggolan 2010). Drs. Richard Lange and L David Hillis of the University of Texas Health Science Center note "The notion that recreational cocaine use is 'safe' should be dispelled, since even small amounts may have catastrophic consequences" (Nainggolan 2010).
Addiction
Cocaine dependence (or addiction) is psychological dependency on the regular use of cocaine. Cocaine dependency may result in physiological damage, lethargy, psychosis, depression, akathisia, and fatal overdose.
Physical withdrawal is not dangerous. Physiological changes caused by cocaine withdrawal include vivid and unpleasant dreams, insomnia or hypersomnia, increased appetite and psychomotor retardation or agitation (Lowinson et al. 2004).
Among the notable cocaine-related deaths from cocaine have been basketball player Len Bias, baseball player Ken Caminiti, Quiet Riot singer Kevin DuBrow, The Who musician John Entwistle, actor Chris Farley (along with morphine), model Katy French, The Righteous Brothers musician Bobby Hatfield, the Pretenders musician James Honeyman-Scott, Blind Melon singer Shannon Hoon, actress/singer Whitney Houston (drowning under the influence), Grateful Dead musician Brent Mydland, actor River Phoenix (along with heroin), the Temptations musician David Ruffin, baseball player Rod Scurry, and musician Ike Turner.
Mechanism of action
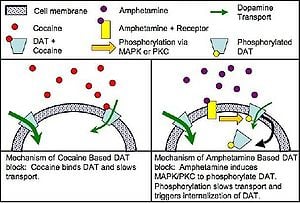
The human brain appears to be hardwired with a reward system that provides pleasure when humans engage in various behaviors that are important to individual or species survival, such as romantic love, sex, and food. Humans ability to feel good involves brain neurotransmiters in this reward system, including dopamine released by neurons in areas known as the ventral tegmental area (VTA), the amygdala, and the nucleus accumbens (Marieb and Hoehn 2010; Spanage and Weiss 1999).
Various drugs of abuse can subvert this reward system, one of which is cocaine. These drugs can cause an addictive pleasure flush by flooding the brain with neurotransmitter-like chemicals or causing a build-up of neurotransmitters such as dopamine. However, this short-lived pleasure also comes with some serious side-effects, including the brain making and releasing less neurotransmitters on its own.
A major effect of cocaine on the central nervous system is the blockade of the dopamine reuptake transporter protein and thus blocking the reabsorption of dopamine. Dopamine transmitter released during neural signaling is normally recycled via the transporter protein; in other words, the transporter binds the transmitter and pumps it out of the synaptic cleft back into the presynaptic neuron, where it is taken up into storage vesicles. By binding tightly with the dopamine transporter, cocaine forms a complex that blocks the transporter's function. The dopamine transporter can no longer perform its reuptake function, and thus dopamine accumulates in the synaptic cleft. This results in an enhanced and prolonged postsynaptic effect of dopaminergic signaling at dopamine receptors on the receiving neuron. In other words, by the dopamine remaining in the synapse, the post-synaptic receptor cells are triggered again and again, allowing a prolonged pleasure flush.
When the dopamine uptake is blocked by repeated use of cocaine, the system reacts by releasing less and less dopamine and "the reward system effectively goes dry" (Marieb and Hoehn 2010). In other words, prolonged exposure to cocaine leads to homeostatic dysregulation of normal dopaminergic signaling via down-regulation of dopamine receptors and enhanced signal transduction. The decreased dopaminergic signaling after chronic cocaine use may contribute to depressive mood disorders and sensitize this important brain reward circuit to the reinforcing effects of cocaine (for example, enhanced dopaminergic signaling only when cocaine is self-administered). This sensitization contributes to the intractable nature of addiction and relapse.
Dopamine-rich brain regions such as the ventral tegmental area, nucleus accumbens, and prefrontal cortex are frequent targets of cocaine addiction research. Of particular interest is the pathway consisting of dopaminergic neurons originating in the ventral tegmental area that terminate in the nucleus accumbens. This projection may function as a "reward center," in that it seems to show activation in response to drugs of abuse like cocaine in addition to natural rewards like food or sex (Spanage and Weiss 1999). While the precise role of dopamine in the subjective experience of reward is highly controversial among neuroscientists, the release of dopamine in the nucleus accumbens is widely considered to be at least partially responsible for cocaine's rewarding effects. This hypothesis is largely based on laboratory data involving rats that are trained to self-administer cocaine. If dopamine antagonists are infused directly into the nucleus accumbens, well-trained rats self-administering cocaine will initially increase responding only to stop completely, thereby indicating that cocaine is no longer reinforcing (i.e. rewarding) the drug-seeking behavior.
Cocaine also effects seratonin (5-hydroxytryptamine, 5-HT), a monoamine neurotransmitter widely thought to be a contributor to feelings of well-being and happiness. Cocaine has been shown to inhibit the re-uptake of 5-HT3. The overabundance of 5-HT3 receptors in cocaine conditioned rats display this trait; however, the exact effect of 5-HT3 in this process is unclear (Carta et al. 2003). The 5-HT2 receptor (particularly the subtypes 5-HT2AR, 5-HT2BR and 5-HT2CR) show influence in the evocation of hyperactivity displayed in cocaine use (Filip et al. 2004).
Sigma receptors are affected by cocaine, as cocaine functions as a sigma ligand agonist (NIH/NIDA 2003). Sigma receptors are proteins found in the brain (and other parts of the body). The impact of cocaine on these sigma receptions may be part of the reason for cocaine's suppression of the immune system (NIH/NIDA 2003). Another specific receptor cocaine has been demonstrated to function on is NMDA (Lluch et al. 2005).
Cocaine also blocks sodium channels, thereby interfering with the propagation of action potentials; thus, like lignocaine and novocaine, it acts as a local anesthetic. It also functions on the binding sites to the dopamine and serotonin sodium dependent transport area as targets as separate mechanisms from its reuptake of those transporters; unique to its local anesthetic value, which makes it in a class of functionality different from both its own derived phenyltropanes analogues (which have that removed) and the amphetamine class of stimulants (which as well altogether lack that). In addition to this cocaine has some target binding to the site of the Kappa-opioid receptor as well. Cocaine also causes vasoconstriction, thus reducing bleeding during minor surgical procedures. The locomotor enhancing properties of cocaine may be attributable to its enhancement of dopaminergic transmission from the substantia nigra.
The impact of the neurotransmitter glutamate is also believed to be important to maintaining addiction, as glutamate signaling seems to cause permanent brain changes that lead to "compulsive drug-seeking behavior elicited by external cues" (Marieb and Hoehn 2010). Mice lacking a particular glutamate receptor are willing to try cocaine but do not become addicted (Marieb and Hoehn 2010). These combined dopamine and glutamate systems are so strong that years later, certain setting can create intense cravings for cocaine (Marieb and Hoehn 2010).
Because nicotine increases the levels of dopamine in the brain, many cocaine users find that consumption of tobacco products during cocaine use enhances the euphoria. This, however, may have undesirable consequences, such as uncontrollable chain smoking during cocaine use (even users who do not normally smoke cigarettes have been known to chain smoke when using cocaine), in addition to the detrimental health effects and the additional strain on the cardiovascular system caused by tobacco.
Forms
Cocaine in its purest form is a white, pearly product. Cocaine appearing in powder form is a salt, typically cocaine hydrochloride. Street market cocaine is frequently adulterated or “cut” with various powdery fillers to increase its weight; the substances most commonly used in this process are baking soda; sugars, such as lactose, dextrose, inositol, and mannitol; and local anesthetics, such as lidocaine or benzocaine, which mimic or add to cocaine's numbing effect on mucous membranes. Cocaine may also be "cut" with other stimulants such as methamphetamine. Adulterated cocaine is often a white, off-white or pinkish powder.
Salts. Cocaine is a weakly alkaline compound (an "alkaloid") and can therefore combine with acidic compounds to form various salts. The hydrochloride (HCl) salt of cocaine is by far the most commonly encountered, although the sulfate (-SO4) and the nitrate (-NO3) are occasionally seen. Different salts dissolve to a greater or lesser extent in various solvents. The hydrochloride salt is polar in character and is quite soluble in water. Powdered cocaine is commonly known as "coke" or "blow" and users can snort the powder (inhale through the nose) and into the bloodstream, or dissolve in water and inject directly into the bloodstream.
Basic. As the name implies, “freebase” or "free base" is the base form of cocaine, as opposed to the salt form. It is practically insoluble in water whereas hydrochloride salt is water soluble. (Most alkaloids are unstable in their pure form and exist in ionic salt form. The salts usually exhibit greater water solubility. Common counterions include chloride, bromide, acetate and oxalate. Because of the ubiquity of chloride salts, formed from the reaction of the amine with hydrochloric acid, these amine derivatives are known as the hydrochlorides.) Pure cocaine is prepared by neutralizing its compounding salt with an alkaline solution, which will precipitate to non-polar basic cocaine. It is further refined through aqueous-solvent Liquid-liquid extraction.
The term "freebasing" means converting an ionic form into free base. It can refer to deprotonating the hydrochloride salt form of cocaine to free base form. The free base is preferred for smoking. Smoking freebase cocaine has the additional effect of releasing methylecgonidine into the user's system due to the pyrolysis of the substance (a side effect which insufflating or injecting powder cocaine does not create). Some research suggests that smoking freebase cocaine can be even more cardiotoxic than other routes of administration (Scheidweiler et al. 2003; Yang et al. 2001; Fandiño et al. 2002).
Crack cocaine. Crack is a lower purity form of free-base cocaine that is usually produced by neutralization of cocaine hydrochloride with a solution of baking soda (sodium bicarbonate, NaHCO3) and water, producing a very hard/brittle, off-white-to-brown colored, amorphous material that contains sodium carbonate, entrapped water, and other by-products as the main impurities. The color of “crack” cocaine depends upon several factors including the origin of the cocaine used, the method of preparation—with ammonia or baking soda—and the presence of impurities, but will generally range from white to a yellowish cream to a light brown. Its texture will also depend on the adulterants, origin and processing of the powdered cocaine, and the method of converting the base. It ranges from a crumbly texture, sometimes extremely oily, to a hard, almost crystalline nature.
The "freebase" and "crack" forms of cocaine are usually administered by vaporization of the powdered substance into smoke, which is then inhaled. The origin of the name "crack" comes from the "crackling" sound (and hence the onomatopoeic moniker “crack”) that is produced when the cocaine and its impurities (i.e. water, sodium bicarbonate) are heated past the point of vaporization (Nelson 1998). Pure cocaine base/crack can be smoked because it vaporizes smoothly, with little or no decomposition at 98 °C (208 °F) (Miller et al. 2009), which is below the boiling point of water. In contrast, cocaine hydrochloride does not vaporize until heated to a much higher temperature (about 197°C), and considerable decomposition/burning occurs at these high temperatures. This effectively destroys some of the cocaine, and yields a sharp, acrid, and foul-tasting smoke.
Unprocessed coca leaf. Coca leaves have been used unprocessed for thousands of years in South America for various religious, social, medicinal, and nutritional purposes, including to control hunger and combat the impacts of high altitudes. Chewing of unadulterated coca leaves has been a tradition in the Andes for thousands of years and remains practiced by millions in South America today (Cortes 2013). Individuals may suck on wads of the leaves and keep them in their cheeks for hours at a time, often combining with chalk or ask to help dissolve the alkaloids into the saliva (Boucher 1991). Unprocessed coca leaves are also commonly used in the Andean countries to make a herbal tea with mild stimulant effects. However, since the alkaloid cocaine is present in only trace amounts in the leaves, it does not cause the euphoric and psychoactive effects associated with use of the drug. (See the article coca.)
Routes of administration
Cocaine powder can be inhaled through the nose or dissolved in water and injected into the bloodstream, as well as rubbed along the gum line. The freebase form can be smoked. Cocaine can also be applied to the skin as an topical anesthetic. Coca leaf can be chewed and brewed into a tea. Injecting and smoking leads to faster absorption into the bloodstream than snorting and a quicker, stronger high, but faster absorption also tends to mean a shorter duration of the high (5-10 minutes for smoking versus 15-30 minutes for snorting)(Botany Central 2013).
Insufflation ("snorting," "sniffing," or "blowing") involves breathing in the powder through the nose and in that manner absorption into the bloodstream. Prior to insufflation, cocaine powder is divided into very fine particles. Rolled up banknotes, hollowed-out pens, cut straws, and other such items are often used to insufflate cocaine. Upon snorting, the drug coats and is absorbed through the mucous membranes lining the sinuses. Any material not directly absorbed through the mucous membranes is collected in mucus and swallowed. When insufflating cocaine, absorption through the nasal membranes is approximately 30–60%, with higher doses leading to increased absorption efficiency.
Nasal insufflation is the most common method of ingestion of recreational powdered cocaine in the Western world. In a study of cocaine users, the average time taken to reach peak subjective effects was 14.6 minutes (Volkow et al. 2000). Physiological and psychotropic effects from nasally insufflated cocaine are sustained for approximately 40–60 minutes after the peak effects are attained (Barnett et al. 1981). Snorting involves slower absorption into the bloodstream; however, as with other means of administration sudden death remains a risk, as with the other medical complications, including potential damage to the inside of the nose due to cocaine highly constricting blood vessels and therefore blood and oxygen/nutrient flow to that area. In addition, a study by Bonkovsky and Mehta (2001) reported that, just like shared needles, the sharing of straws used to "snort" cocaine can spread blood diseases such as hepatitis C.
Injection. Injection, involving administering the drug directly to the bloodstream by the use of needles, provides the highest blood levels of drug in the shortest amount of time. Volkow et al. (2000) found that the average time taken to reach peak subjective effects was 3.1 minutes. The euphoria passes quickly. Aside from the toxic effects of cocaine, there is also danger of circulatory emboli from the insoluble substances that may be used to cut the drug. Subjective effects not commonly shared with other methods of administration include a ringing in the ears moments after injection (usually when in excess of 120 milligrams) lasting 2 to 5 minutes, including tinnitus & audio distortion. As with all injected illicit substances, there is a risk of the user contracting blood-borne infections if sterile injecting equipment is not available or used. Additionally, because cocaine is a vasoconstrictor, and usage often entails multiple injections within several hours or less, subsequent injections are progressively more difficult to administer, which in turn may lead to more injection attempts and more sequelae from improperly performed injection. An injected mixture of cocaine and heroin, known as “speedball,” is a particularly dangerous combination, as the converse effects of the drugs actually complement each other, but may also mask the symptoms of an overdose. It has been responsible for numerous deaths, including celebrities such as John Belushi, Chris Farley, Mitch Hedberg, River Phoenix and Layne Staley.
Inhalation. Inhalation or smoking involves inhaling cocaine vapor into the lungs by sublimating solid cocaine by heating. Smoking freebase or crack cocaine is most often accomplished using a pipe made from a small glass tube, often taken from "love roses," small glass tubes with a paper rose that are promoted as romantic gifts (Reist 2005). A small piece of clean heavy copper or occasionally stainless steel scouring pad may serve as a reduction base and flow modulator in which the "rock" can be melted and boiled to vapor. Crack often is smoked by placing it at the end of the pipe; a flame held close to it produces vapor, which is then inhaled by the smoker. Powdered cocaine is also sometimes smoked, though heat destroys much of the chemical. Smoking or vaporizing cocaine and inhaling it into the lungs produces an almost immediate "high" that can be very intense quite rapidly. In a Brookhaven National Laboratory medical department study, based on self reports of cocaine abusers who participated in the study, "peak high" was found at mean of 1.4min +/- 0.5 minutes (Volkow et al. 2000). While the stimulating effects may last for hours, the euphoric sensation is very brief—usually 5 to 15 minutes—prompting the user to smoke more immediately.
Application to skin. Many users rub the powder along the gum line, or onto a cigarette filter, which is then smoked, numbing the gums and teeth—hence, the colloquial names of "numbies", "gummers" or "cocoa puffs" for this type of administration. This is mostly done with the small amounts of cocaine remaining on a surface after insufflation. A medical form of cocaine, strictly regulated and available by prescription, is applied to the skin to numb eye, nose, and throat pain (WebMD 2013b).
Oral: Coca leaf chewing and infusions (tea). Unadulterated coca leaves have been chewed for thousands of years in the Andes and remains practiced by millions in South America today (Cortes 2013). Individuals may suck on wads of the leaves and keep them in their cheeks for hours at a time. Coca leaves are typically mixed with an alkaline substance (such as lime) to help dissolve the alkaloids into the saliva and chewed into a wad that is retained in the mouth between gum and cheek (much in the same as chewing tobacco is chewed) and sucked of its juices. The juices are absorbed slowly by the mucous membrane of the inner cheek and by the gastrointestinal tract when swallowed. While the cocaine in the plant has little effect on the unbroken skin, it does act on the mucous membranes of the mouth (as well as the membranes of the eye, nose, and stomach) (Royal Botanic Gardens 1985). However, since the alkaloid cocaine is only in trace amounts in the leaves, it does not cause the euphoric and psychoactive effects associated with use of the concentrated drug. Concentrations vary by variety and region, but leaves have been reported variously as between 0.25% and 0.77% (Plowman and Rivier 1983), between 0.35% and 0.72% by dry weight (Nathanson et al. 1993), and between 0.3% and 1.5% and averaging 0.8% in fresh leaves (Casale and Klein 1993).
Coca leaves also can be boiled to provide a tea. Although coca leaf chewing is common mainly among the indigenous populations, the consumption of coca tea (Mate de coca) is common among all sectors of society in the Andean countries.
Because cocaine is hydrolyzed and rendered inactive in the acidic stomach, it is not readily absorbed when ingested alone. Only when mixed with a highly alkaline substance (such as lime) can it be absorbed into the bloodstream through the stomach. The efficiency of absorption of orally administered cocaine is limited by two additional factors. First, the drug is partly catabolized by the liver. Second, capillaries in the mouth and esophagus constrict after contact with the drug, reducing the surface area over which the drug can be absorbed. Nevertheless, cocaine metabolites can be detected in the urine of subjects that have sipped even one cup of coca leaf infusion. Therefore, this is an actual additional form of administration of cocaine, albeit an inefficient one.
Other methods. An oral method for the psychoactive drug is to wrap up some cocaine in rolling paper and swallow (parachute) it. Little research has been focused on another method: the suppository (anal or vaginal insertion) method of administration, also known as "plugging." This method of administration is commonly administered using an oral syringe. Cocaine can be dissolved in water and withdrawn into an oral syringe which may then be lubricated and inserted into the anus or vagina before the plunger is pushed. The rectum and the vaginal canal is where the majority of the drug would likely be taken up, through the membranes lining its walls.
Uses
Recreational psychoactive drug
Cocaine is best known worldwide for its illegal use as a recreational psychoactive drug. As noted above, this concentrated form of cocaine particularly is used nasally (nasal insufflation is also known as "snorting," "sniffing," or "blowing"), injected, or smoked. In the United States, the development of "crack" cocaine introduced the substance to a generally poorer inner-city market.
The United Nations Office of Drugs and Crime estimated that in 2009, the US cocaine market was $37 billion (and shrinking over the past ten years) and the West and Central European Cocaine market was US$ 33 billion (and increasing over the past ten years) (USODC 2011). According to a 2007 United Nations report, Spain is the country with the highest rate of cocaine usage (3.0% of adults in the previous year) (UNODC 2007). Other countries where the usage rate meets or exceeds 1.5% are the United States (2.8%), England and Wales (2.4%), Canada (2.3%), Italy (2.1%), Bolivia (1.9%), Chile (1.8%), and Scotland (1.5%) (UNODC 2007).
The production, distribution and sale of cocaine products is restricted and/or illegal in most countries. Internationally, it is regulated by the Single Convention on Narcotic Drugs, and the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances. In the United States, the manufacture, importation, possession, and distribution of cocaine is additionally regulated by the 1970 Controlled Substances Act. Cocaine is generally treated as a 'hard drug', with severe penalties for possession and trafficking.
Medicine
Strictly regulated, cocaine can be applied externally to the skin to numb pain. Cocaine was historically used as a topical anesthetic in eye and nasal surgery. It is now predominantly used for nasal and lacrimal duct surgery. The major disadvantages of this use are cocaine's intense vasoconstrictor activity and potential for cardiovascular toxicity. Cocaine has since been largely replaced in Western medicine by synthetic local anesthetics such as benzocaine, proparacaine, lignocaine/xylocaine/lidocaine, and tetracaine though it remains available for use if specified. If vasoconstriction is desired for a procedure (as it reduces bleeding), the anesthetic is combined with a vasoconstrictor such as phenylephrine or epinephrine.
In Australia, cocaine is currently prescribed for use as a local anesthetic for conditions such as mouth and lung ulcers. Some ENT specialists occasionally use cocaine within the practice when performing procedures such as nasal cauterization. In this scenario dissolved cocaine is soaked into a ball of cotton wool, which is placed in the nostril for the 10–15 minutes immediately prior to the procedure, thus performing the dual role of both numbing the area to be cauterized, and vasoconstriction. Even when used this way, some of the used cocaine may be absorbed through oral or nasal mucosa and give systemic effects.
In the United States, cocaine remains an FDA-approved Schedule C-II drug, which can be prescribed by a healthcare provider, but is strictly regulated. A form of cocaine available by prescription is applied to the skin to numb eye, nose, and throat pain and narrow blood vessels (WebMD 2013b).
Unprocessed coca leaf traditionally has been used for a variety of medical purposes, including as a stimulant to overcome fatigue, hunger, and thirst. Because coca constricts blood vessels, it also serves to oppose bleeding, and coca seeds were used for nosebleeds. Coca leaf also has been used to overcome altitude sickness, and in the Andes tourists have been offered coca tea for this purpose (Cortes 2013). In addition, coca extracts have been used as a muscle and cerebral stimulant to alleviate nausea, vomiting, and stomach pains without upsetting digestion (Botany Central 2013; WebMD 2013b). (See the article coca for these and other uses of the coca leaf.)
In the United States, a Stepan Company plant in Maywood, New Jersey manufactures pure cocaine for medical use and also produces a cocaine-free extract of the coca leaf, which is used as a flavoring ingredient in Coca-Cola. Other companies have registrations with the DEA to import coca leaf according to 2011 Federal Register Notices for Importers (ODC 2011), including Johnson Matthey, Inc, Pharmaceutical Materials; Mallinckrodt Inc; Penick Corporation; and the Research Triangle Institute.
History
- Cocaine was first isolated in 1859 by Albert Niemann of the University of Göttingen; it was he who named coca's chief alkaloid "cocaine" (Inciardi 1992).
- Coca wine (of which Vin Mariani was the best-known brand) and other coca-containing preparations were widely sold as patent medicines and tonics, with claims of a wide variety of health benefits. The original version of Coca-Cola was among these, although the amount in Coca-Cola may have been only trace amounts. Products with cocaine became illegal in most countries outside of South America in the early 20th century, after the addictive nature of cocaine was widely recognized.
Discovery

For over a thousand years South American indigenous peoples have chewed the leaves of Erythroxylon coca, a plant that contains vital nutrients as well as numerous alkaloids, including cocaine. The coca leaf was, and still is, chewed almost universally by some indigenous communities. The remains of coca leaves have been found with ancient Peruvian mummies, and pottery from the time period depicts humans with bulged cheeks, indicating the presence of something on which they are chewing.[7] There is also evidence that these cultures used a mixture of coca leaves and saliva as an anesthetic for the performance of trepanation.[8]
When the Spanish arrived in South America, most at first ignored aboriginal claims that the leaf gave them strength and energy, and declared the practice of chewing it the work of the Devil.[citation needed] But after discovering that these claims were true, they legalized and taxed the leaf, taking 10% off the value of each crop.[9] In 1569, Nicolás Monardes described the practice of the natives of chewing a mixture of tobacco and coca leaves to induce "great contentment":
When they wished to make themselves drunk and out of judgment they chewed a mixture of tobacco and coca leaves which make them go as they were out of their wittes.
In 1609, Padre Blas Valera wrote:
Coca protects the body from many ailments, and our doctors use it in powdered form to reduce the swelling of wounds, to strengthen broken bones, to expel cold from the body or prevent it from entering, and to cure rotten wounds or sores that are full of maggots. And if it does so much for outward ailments, will not its singular virtue have even greater effect in the entrails of those who eat it?[citation needed]
Isolation and naming
Although the stimulant and hunger-suppressant properties of coca had been known for many centuries, the isolation of the cocaine alkaloid was not achieved until 1855. Various European scientists had attempted to isolate cocaine, but none had been successful for two reasons: the knowledge of chemistry required was insufficient at the time. [citation needed] Additionally contemporary conditions of sea-shipping from South America could degrade the cocaine in the plant samples available to Europeans. [citation needed]
The cocaine alkaloid was first isolated by the German chemist Friedrich Gaedcke in 1855. Gaedcke named the alkaloid "erythroxyline", and published a description in the journal Archiv der Pharmazie.[11]
In 1856, Friedrich Wöhler asked Dr. Carl Scherzer, a scientist aboard the Novara (an Austrian frigate sent by Emperor Franz Joseph to circle the globe), to bring him a large amount of coca leaves from South America. In 1859, the ship finished its travels and Wöhler received a trunk full of coca. Wöhler passed on the leaves to Albert Niemann, a Ph.D. student at the University of Göttingen in Germany, who then developed an improved purification process.[12]
Niemann described every step he took to isolate cocaine in his dissertation titled Über eine neue organische Base in den Cocablättern (On a New Organic Base in the Coca Leaves), which was published in 1860—it earned him his Ph.D. and is now in the British Library. He wrote of the alkaloid's "colourless transparent prisms" and said that, "Its solutions have an alkaline reaction, a bitter taste, promote the flow of saliva and leave a peculiar numbness, followed by a sense of cold when applied to the tongue." Niemann named the alkaloid "cocaine" from "coca" (from Quechua "cuca") + suffix "ine".[12][13] Because of its use as a local anesthetic, a suffix "-caine" was later extracted and used to form names of synthetic local anesthetics.
The first synthesis and elucidation of the structure of the cocaine molecule was by Richard Willstätter in 1898.[14] The synthesis started from tropinone, a related natural product and took five steps.
Popularization

In 1859, an Italian doctor, Paolo Mantegazza, returned from Peru, where he had witnessed first-hand the use of coca by the natives. He proceeded to experiment on himself and upon his return to Milan he wrote a paper in which he described the effects. In this paper he declared coca and cocaine (at the time they were assumed to be the same) as being useful medicinally, in the treatment of "a furred tongue in the morning, flatulence, and whitening of the teeth."
A chemist named Angelo Mariani who read Mantegazza's paper became immediately intrigued with coca and its economic potential. In 1863, Mariani started marketing a wine called Vin Mariani, which had been treated with coca leaves, to become cocawine. The ethanol in wine acted as a solvent and extracted the cocaine from the coca leaves, altering the drink's effect. It contained 6 mg cocaine per ounce of wine, but Vin Mariani which was to be exported contained 7.2 mg per ounce, to compete with the higher cocaine content of similar drinks in the United States. A "pinch of coca leaves" was included in John Styth Pemberton's original 1886 recipe for Coca-Cola, though the company began using decocainized leaves in 1906 when the Pure Food and Drug Act was passed. The actual amount of cocaine that Coca-Cola contained during the first 20 years of its production is practically impossible to determine.[citation needed]
In 1879 cocaine began to be used to treat morphine addiction. Cocaine was introduced into clinical use as a local anesthetic in Germany in 1884, about the same time as Sigmund Freud published his work Über Coca, in which he wrote that cocaine causes:
Exhilaration and lasting euphoria, which in no way differs from the normal euphoria of the healthy person. You perceive an increase of self-control and possess more vitality and capacity for work. In other words, you are simply normal, and it is soon hard to believe you are under the influence of any drug. Long intensive physical work is performed without any fatigue. This result is enjoyed without any of the unpleasant after-effects that follow exhilaration brought about by alcohol. Absolutely no craving for the further use of cocaine appears after the first, or even after repeated taking of the drug.
In 1885 the U.S. manufacturer Parke-Davis sold cocaine in various forms, including cigarettes, powder, and even a cocaine mixture that could be injected directly into the user's veins with the included needle. The company promised that its cocaine products would "supply the place of food, make the coward brave, the silent eloquent and render the sufferer insensitive to pain."
By the late Victorian era cocaine use had appeared as a vice in literature. For example, it was injected by Arthur Conan Doyle's fictional Sherlock Holmes, generally to offset the boredom he felt when he was not working on a case.
In 1879, Vassili von Anrep, of the University of Würzburg, devised an experiment to demonstrate the analgesic properties of the newly discovered alkaloid. He prepared two separate jars, one containing a cocaine-salt solution, with the other containing merely salt water. He then submerged a frog's legs into the two jars, one leg in the treatment and one in the control solution, and proceeded to stimulate the legs in several different ways. The leg that had been immersed in the cocaine solution reacted very differently from the leg that had been immersed in salt water.[16]
Karl Koller (a close associate of Sigmund Freud, who would write about cocaine later) experimented with cocaine for ophthalmic usage. In an infamous experiment in 1884, he experimented upon himself by applying a cocaine solution to his own eye and then pricking it with pins. His findings were presented to the Heidelberg Ophthalmological Society. Also in 1884, Jellinek demonstrated the effects of cocaine as a respiratory system anesthetic. In 1885, William Halsted demonstrated nerve-block anesthesia,[17] and James Leonard Corning demonstrated peridural anesthesia.[18] 1898 saw Heinrich Quincke use cocaine for spinal anesthesia.
In early 20th-century Memphis, Tennessee, cocaine was sold in neighborhood drugstores on Beale Street, costing five or ten cents for a small boxful. Stevedores along the Mississippi River used the drug as a stimulant, and white employers encouraged its use by black laborers.[19]
In 1909, Ernest Shackleton took "Forced March" brand cocaine tablets to Antarctica, as did Captain Scott a year later on his ill-fated journey to the South Pole.[20]
During the mid-1940s, amidst WWII, cocaine was considered for inclusion as an ingredient of a future generation of 'pep pills' for the German military code named D-IX.[21]
Prohibition
Template:Globalize/US
Prohibition of cocaine in the United States
Calls for prohibition began long before the Harrison Act was passed by Congress in 1914 – a law requiring cocaine and narcotics to be dispensed only with a doctor's order.[22] Before this, various factors and groups acted on primarily a state level influencing a move towards prohibition and away from a laissez-faire attitude.[23]
By 1903 cocaine consumption had grown to about five times that of 1890. Non-medical users accounted for almost the entire increase as cocaine-users extended outside the middle-aged, white, professional class. Cocaine became associated with laborers, youths, blacks and the urban underworld.[24]
The popularization of cocaine first began with laborers who used cocaine as a stimulant to increase productivity.[24] Cocaine was often supplied by employers.[25] Cocaine was often supplied to African American workers, who many employers believed were better physical workers; cocaine was thought to provide added strength and constitution and according to the Medical News, made blacks “impervious to the extremes of heat and cold.”[24] However, users of cocaine quickly acquired a reputation as dangerous and in 1897, the first state bill of control for cocaine sales came from a mining county in Colorado.Spillane, pp. 92-93</ref>
The popularization of cocaine use was not confined to African Americans or simple laborers. In Northern cities, cocaine use increased amongst poorer people – in fact, cocaine was often cheaper than alcohol.[25] In the Northeast in particular, cocaine became popular amongst workers in factories, textile mills and on rail roads.[26] In some instances, cocaine use supplemented or replaced caffeine as the drug-of-choice to keep workers awake and working overtime.[26]
This period of increasing cocaine use followed with increasing fears that young children were being preyed upon and forced into cocaine addiction.[27] Indeed, it was even thought that cocaine was used to seduce young girls away from their homes and cause them to be addicted and dependent upon the substance and therefore fall prey to an inescapable cycle of prostitution.[28] Fears of the corruption of the youth by cocaine were popular and widespread but there is little evidence to support their veracity.[27]
Mainstream media reported cocaine epidemics as early as 1894 in Dallas, Texas. Reports of the cocaine epidemic would foreshadow a familiar theme in later so-called epidemics, namely that cocaine presented a social threat more dangerous than simple health effects and had insidious results when used by blacks and members of the lower class. Similar anxiety-ridden reports appeared throughout cities in the South leading some to declare that “the cocaine habit has assumed the proportions of an epidemic among the colored people.” In 1900, state legislatures in Alabama, Georgia and Tennessee considered anti-cocaine bills for the first time.[29]
Hyperbolic reports of the effect of cocaine on African Americans went hand-in-hand with this hysteria. In 1901, the Atlanta Constitution reported that “Use of the drug [cocaine] among negroes is growing to an alarming extent.”[30] The New York Times reported that under the influence of cocaine, “sexual desires are increased and perverted … peaceful negroes become quarrelsome, and timid negroes develop a degree of 'Dutch courage' that is sometimes almost incredible.”[31] A medical doctor even wrote “cocaine is often the direct incentive to the crime of rape by the negroes.”[31] To complete the characterization, a judge in Mississippi declared that supplying a “negro” with cocaine was more dangerous than injecting a dog with rabies.[32]
These attitudes not only influenced drug law and policy but also led to increased violence against African Americans. In 1906, a major race riot led by whites erupted; it was sparked by reports of crimes committed by black ‘cocaine fiends.’[30] Indeed, white-led, race riots spawning from reports of blacks under the influence of cocaine were not uncommon.[33] Police in the South widely adopted the use of a heavier caliber handguns so as to better stop a cocaine-crazed black person – believed to be empowered with super-human strength.[34] Another dangerous myth perpetuated amongst police was that cocaine imbued African Americans with tremendous accuracy with firearms and therefore police were better advised to shoot first in questionable circumstances.[35]
Ultimately public opinion rested against the cocaine user. Criminality was commonly believed to be a natural result of cocaine use.[36] Much of the influence for these kind of perceptions came from the widespread publicity given to notorious cases.[23] While the historical reality of cocaine’s effect on violence and crime is difficult to disentangle from inflamed perceptions, it does appear that public opinion was swayed by the image of the violent, cocaine-crazed fiend and pushed over the edge by a few violent episodes.[36] It was an image of the cocaine-user that carried acute racial overtones.[23]
Before any substantive federal regulation of cocaine, state and local municipalities evoked their own means to regulate cocaine. Because of the initial lack of targeted legislation, on both federal and state level, the most typical strategy by law enforcement was the application of nuisance laws pertaining to vagrancy and disturbing the peace.[37] Subsequent legislative actions aimed at controlling the distribution of cocaine rather than its manufacture.[38] Reformers took this approach in part because of legal precedents which made it easier to control distributors such as pharmacies; state and local boards of hearth or boards of pharmacy often took the place of regulatory bodies for controlling the distribution of cocaine.[38]
Some states took the position of outright banning of all forms of cocaine sale; Georgia was the first to do this in 1902.[39] A New Orleans ordinance banned cocaine sales as well but left an ill-defined exception for therapeutic uses.[38] A more common requirement was to restrict the sale of cocaine or impose labeling requirements. A 1907 California law limiting sale of cocaine to only those with a physician’s prescription resulted in the arrest of over 50 store owners and clerks in the first year.[38] A 1913 New York state law limited druggists’ cocaine stocks to under 5 ounces. Labeling requirements initially operated on a state level with some states even going so far as to require that cocaine and cocaine-containing products be labeled as poison.[40]
Eventually the federal government stepped in and instituted a national labeling requirement for cocaine and cocaine-containing products through the Food and Drug Act of 1906.[40] The next impactful federal regulation was the Harrison Narcotics Tax Act of 1914. While this act is often seen as the start of prohibition, the act itself was not actually a prohibition on cocaine, but instead setup a regulatory and licensing regime.[22] The Harrison Act did not recognize addiction as a treatable condition and therefore the therapeutic use of cocaine, heroin or morphine to such individuals was outlawed – leading the Journal of American Medicine to remark, “[the addict] is denied the medical care he urgently needs, open, above-board sources from which he formerly obtained his drug supply are closed to him, and he is driven to the underworld where he can get his drug, but of course, surreptitiously and in violation of the law.”[41] The Harrison Act left manufacturers of cocaine untouched so long as they met certain purity and labeling standards.[42] Despite that cocaine was typically illegal to sell and legal outlets were more rare, the quantities of legal cocaine produced declined very little.[42] Legal cocaine quantities did not decrease until the Jones-Miller Act of 1922 put serious restrictions on cocaine manufactures.[42]
In 1995 the World Health Organization (WHO) and the United Nations Interregional Crime and Justice Research Institute (UNICRI) announced in a press release the publication of the results of the largest global study on cocaine use ever undertaken. However, a decision by an American representative in the World Health Assembly banned the publication of the study, because it seemed to make a case for the positive uses of cocaine. An excerpt of the report strongly conflicted with accepted paradigms, for example "that occasional cocaine use does not typically lead to severe or even minor physical or social problems."[43] In the sixth meeting of the B committee the US representative threatened that "If WHO activities relating to drugs failed to reinforce proven drug control approaches, funds for the relevant programs should be curtailed". This led to the decision to discontinue publication. A part of the study has been recuperated. Available are profiles of cocaine use in 20 countries.
It was reported in October 2010 that the use of cocaine in Australia has doubled since monitoring began in 2003.[44]
A problem with illegal cocaine use, especially in the higher volumes used to combat fatigue (rather than increase euphoria) by long-term users, is the risk of ill effects or damage caused by the compounds used in adulteration. Cutting or "stepping on" the drug is commonplace, using compounds which simulate ingestion effects, such as Novocain (procaine) producing temporary anesthaesia as many users believe a strong numbing effect is the result of strong and/or pure cocaine, ephedrine or similar stimulants that are to produce an increased heart rate. The normal adulterants for profit are inactive sugars, usually mannitol, creatine or glucose, so introducing active adulterants gives the illusion of purity and to 'stretch' or make it so a dealer can sell more product than without the adulterants.[citation needed] The adulterant of sugars therefore allows the dealer to sell the product for a higher price because of the illusion of purity and allows to sell more of the product at that higher price, enabling dealers to make a lot of revenue with little cost of the adulterants. Cocaine trading carries large penalties in most jurisdictions, so user deception about purity and consequent high profits for dealers are the norm. A study by the European Monitoring Centre for Drugs and Drug Addiction in 2007 showed that the purity levels for street purchased cocaine was often under 5% and on average under 50% pure.[45]
Detection in biological fluids
Cocaine and its major metabolites may be quantitated in blood, plasma or urine to monitor for abuse, confirm a diagnosis of poisoning or assist in the forensic investigation of a traffic or other criminal violation or a sudden death. Most commercial cocaine immunoassay screening tests cross-react appreciably with the major cocaine metabolites, but chromatographic techniques can easily distinguish and separately measure each of these substances. When interpreting the results of a test, it is important to consider the cocaine usage history of the individual, since a chronic user can develop tolerance to doses that would incapacitate a cocaine-naive individual, and the chronic user often has high baseline values of the metabolites in his system. Cautious interpretation of testing results may allow a distinction between passive or active usage, and between smoking versus other routes of administration.[46] In 2011, researchers at John Jay College of Criminal Justice reported that dietary zinc supplements can mask the presence of cocaine and other drugs in urine. Similar claims have been made in web forums on that topic.[47]
Society and culture
Legal status
The production, distribution and sale of cocaine products is restricted (and illegal in most contexts) in most countries as regulated by the Single Convention on Narcotic Drugs, and the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances. In the United States the manufacture, importation, possession, and distribution of cocaine is additionally regulated by the 1970 Controlled Substances Act.
Some countries, such as Peru and Bolivia permit the cultivation of coca leaf for traditional consumption by the local indigenous population, but nevertheless prohibit the production, sale and consumption of cocaine. In addition, some parts of Europe and Australia allow processed cocaine for medicinal uses only.
Interdiction
In 2004, according to the United Nations, 589 tonnes of cocaine were seized globally by law enforcement authorities. Colombia seized 188 t, the United States 166 t, Europe 79 t, Peru 14 t, Bolivia 9 t, and the rest of the world 133 t.[48]
Illicit trade
Because of the extensive processing it undergoes during preparation, cocaine is generally treated as a 'hard drug', with severe penalties for possession and trafficking. Demand remains high, and consequently black market cocaine is quite expensive. Unprocessed cocaine, such as coca leaves, are occasionally purchased and sold, but this is exceedingly rare as it is much easier and more profitable to conceal and smuggle it in powdered form. The scale of the market is immense: 770 tonnes times $100 per gram retail = up to $77 billion.[citation needed]
Production
Until 2012, Colombia was the world's leading producer of cocaine.[49][50] Three-quarters of the world's annual yield of cocaine has been produced in Colombia, both from cocaine base imported from Peru (primarily the Huallaga Valley) and Bolivia, and from locally grown coca. There was a 28% increase from the amount of potentially harvestable coca plants which were grown in Colombia in 1998. This, combined with crop reductions in Bolivia and Peru, made Colombia the nation with the largest area of coca under cultivation after the mid-1990s. Coca grown for traditional purposes by indigenous communities, a use which is still present and is permitted by Colombian laws, only makes up a small fragment of total coca production, most of which is used for the illegal drug trade.
An interview with a coca farmer published in 2003 described a mode of production by acid-base extraction that has changed little since 1905. Roughly 625 pounds of leaves were harvested per hectare, six times per year. The leaves were dried for half a day, then chopped into small pieces with a strimmer and sprinkled with a small amount of powdered cement (replacing sodium carbonate from former times). Several hundred pounds of this mixture was soaked in 50 US gallons (Template:Convert/L) of gasoline for a day, then the gasoline was removed and the leaves were pressed for remaining liquid, after which they could be discarded. Then battery acid (weak sulfuric acid) was used, one bucket per 25 kilograms of leaves, to create a phase separation in which the cocaine free base in the gasoline was acidified and extracted into a few buckets of "murky-looking smelly liquid". Once powdered caustic soda was added to this, the cocaine precipitated and could be removed by filtration through a cloth. The resulting material, when dried, was termed pasta and sold by the farmer. The 3750 pound yearly harvest of leaves from a hectare produced 2.5 kg (6 lb) of pasta, approximately 40–60% cocaine. Repeated recrystallization from solvents, producing pasta lavada and eventually crystalline cocaine, were performed at specialized laboratories after the sale.[51]
Attempts to eradicate coca fields through the use of defoliants have devastated part of the farming economy in some coca growing regions of Colombia, and strains appear to have been developed that are more resistant or immune to their use. Whether these strains are natural mutations or the product of human tampering is unclear. These strains have also shown to be more potent than those previously grown, increasing profits for the drug cartels responsible for the exporting of cocaine. Although production fell temporarily, coca crops rebounded in numerous smaller fields in Colombia, rather than the larger plantations.
The cultivation of coca has become an attractive, and in some cases even necessary, economic decision on the part of many growers due to the combination of several factors, including the persistence of worldwide demand, the lack of other employment alternatives, the lower profitability of alternative crops in official crop substitution programs, the eradication-related damages to non-drug farms, and the spread of new strains of the coca plant.
| 2000 | 2001 | 2002 | 2003 | 2004 | |
|---|---|---|---|---|---|
| Net cultivation (km2) | 1875 | 2218 | 2007.5 | 1663 | 1662 |
| Potential pure cocaine production (tonnes) | 770 | 925 | 830 | 680 | 645 |
The latest estimate provided by the U.S. authorities on the annual production of cocaine in Colombia refers to 290 metric tons. As of the end of 2011, the seizure operations of Colombian cocaine carried out in different countries have totaled 351.8 metric tons of cocaine, i.e. 121.3% of Colombia’s annual production according to the U.S. Department of State’s estimates. [53][54]
Synthesis
Synthetic cocaine would be highly desirable to the illegal drug industry, as it would eliminate the high visibility and low reliability of offshore sources and international smuggling, replacing them with clandestine domestic laboratories, as are common for illicit methamphetamine. However, natural cocaine remains the lowest cost and highest quality supply of cocaine. Actual full synthesis of cocaine is rarely done. Formation of inactive enantiomers (cocaine has 4 chiral centres – 1R,2R,3S,5S – hence a total potential of 16 possible enantiomers and diastereoisomers) plus synthetic by-products limits the yield and purity. Names like "synthetic cocaine" and "new cocaine" have been misapplied to phencyclidine (PCP) and various designer drugs.
Trafficking and distribution
Organized criminal gangs operating on a large scale dominate the cocaine trade. Most cocaine is grown and processed in South America, particularly in Colombia, Bolivia, Peru, and smuggled into the United States and Europe, the United States being the world's largest consumer of cocaine,[55] where it is sold at huge markups; usually in the US at $80–$120 for 1 gram, and $250–300 for 3.5 grams (1/8 of an ounce, or an "eight ball").
Caribbean and Mexican routes
Cocaine shipments from South America transported through Mexico or Central America are generally moved over land or by air to staging sites in northern Mexico. The cocaine is then broken down into smaller loads for smuggling across the U.S.–Mexico border. The primary cocaine importation points in the United States are in Arizona, southern California, southern Florida, and Texas. Typically, land vehicles are driven across the U.S.-Mexico border. Sixty five percent of cocaine enters the United States through Mexico, and the vast majority of the rest enters through Florida.[56]
Cocaine traffickers from Colombia, and recently Mexico, have also established a labyrinth of smuggling routes throughout the Caribbean, the Bahama Island chain, and South Florida. They often hire traffickers from Mexico or the Dominican Republic to transport the drug. The traffickers use a variety of smuggling techniques to transfer their drug to U.S. markets. These include airdrops of 500–700 kg in the Bahama Islands or off the coast of Puerto Rico, mid-ocean boat-to-boat transfers of 500–2,000 kg, and the commercial shipment of tonnes of cocaine through the port of Miami.
Chilean route
Another route of cocaine traffic goes through Chile, this route is primarily used for cocaine produced in Bolivia since the nearest seaports lie in northern Chile. The arid Bolivia-Chile border is easily crossed by 4x4 vehicles that then head to the seaports of Iquique and Antofagasta. While the price of cocaine is higher in Chile than in Peru and Bolivia, the final destination is usually Europe, especially Spain where drug dealing networks exist among South American immigrants.
Techniques
Cocaine is also carried in small, concealed, kilogram quantities across the border by couriers known as “mules” (or “mulas”), who cross a border either legally, for example, through a port or airport, or illegally elsewhere. The drugs may be strapped to the waist or legs or hidden in bags, or hidden in the body. If the mule gets through without being caught, the gangs will reap most of the profits. If he or she is caught however, gangs will sever all links and the mule will usually stand trial for trafficking alone.
Bulk cargo ships are also used to smuggle cocaine to staging sites in the western Caribbean–Gulf of Mexico area. These vessels are typically 150–250-foot (50–80 m) coastal freighters that carry an average cocaine load of approximately 2.5 tonnes. Commercial fishing vessels are also used for smuggling operations. In areas with a high volume of recreational traffic, smugglers use the same types of vessels, such as go-fast boats, as those used by the local populations.
Sophisticated drug subs are the latest tool drug runners are using to bring cocaine north from Colombia, it was reported on March 20, 2008. Although the vessels were once viewed as a quirky sideshow in the drug war, they are becoming faster, more seaworthy, and capable of carrying bigger loads of drugs than earlier models, according to those charged with catching them.[57]
Sales to consumers
Cocaine is readily available in all major countries' metropolitan areas. According to the Summer 1998 Pulse Check, published by the U.S. Office of National Drug Control Policy, cocaine use had stabilized across the country, with a few increases reported in San Diego, Bridgeport, Miami, and Boston. In the West, cocaine usage was lower, which was thought to be due to a switch to methamphetamine among some users; methamphetamine is cheaper, three and a half times more powerful, and lasts 12 to 24 times longer with each dose.[58][59] Nevertheless, the number of cocaine users remain high, with a large concentration among urban youth.
In addition to the amounts previously mentioned, cocaine can be sold in "bill sizes": for example, $10 might purchase a "dime bag," a very small amount (0.1–0.15 g) of cocaine. Twenty dollars might purchase 0.15–0.3 g. However, in lower Texas, it is sold cheaper due to it being easier to receive: a dime for $10 is 0.4g, a 20 is 0.8–1.0 gram and a 8-ball (3.5g) is sold for $60 to $80, depending on the quality and dealer. These amounts and prices are very popular among young people because they are inexpensive and easily concealed on one's body. Quality and price can vary dramatically depending on supply and demand, and on geographic region.[60]
The European Monitoring Centre for Drugs and Drug Addiction reports that the typical retail price of cocaine varied between €50 and €75 per gram in most European countries, although Cyprus, Romania, Sweden and Turkey reported much higher values.[61]
Notes
- ↑ 1.0 1.1 K. Fattinger, N. L. Benowitz, R. T. Jones, and D. Verotta, "Nasal Mucosal Versus Gastrointestinal Absorption of Nasally Administered Cocaine," Eur. J. Clin. Pharmacol. 56, iss. 4(2000): 305–10. PMID 10954344.
- ↑ G. Barnett, R. Hawks, and R. Resnick, "Cocaine Pharmacokinetics in Humans," J Ethnopharmacol 3, issue 2–3(1981): 353–66. PMID 7242115.
- ↑ A. R. Jeffcoat, M. Perez-Reyes, J. M. Hill, B. M. Sadler, and C. E. Cook, "Cocaine Disposition in Humans After Intravenous Injection, Nasal Insufflation (Snorting), or Smoking," Drug Metab. Dispos. 17, iss. 2(1989): 153–9. PMID 2565204.
- ↑ P. Wilkinson, C. Van Dyke, P. Jatlow, P. Barash, and R. Byck, "Intranasal and Oral Cocaine Kinetics," Clin. Pharmacol. Ther. 27, iss. 3(1980): 386–94. PMID 7357795.
- ↑ D. Nutt, L. A. King, W. Saulsbury, and C. Blakemore, "Development of a Rational Scale to Assess the Harm of Drugs of Potential Misuse". The Lancet 369, iss. 9566 (2007): 1047–1053. Retrieved September 3, 2013
- ↑ Pagliaro, Louis and Ann Marie Pagliaro (2004). Pagliaros’ Comprehensive Guide to Drugs and Substances of Abuse. Washington, D.C.: American Pharmacists Association. ISBN 978-1-58212-066-9.
- ↑ Altman AJ, Albert DM, Fournier GA (1985). Cocaine's use in ophthalmology: our 100-year heritage. Surv Ophthalmol 29 (4): 300–6.
- ↑ Gay GR, Inaba DS, Sheppard CW, Newmeyer JA (1975). Cocaine: history, epidemiology, human pharmacology, and treatment. a perspective on a new debut for an old girl. Clin. Toxicol. 8 (2): 149–78.
- ↑ "Drug that spans the ages: The history of cocaine", The Independent (UK), 2006. Retrieved 2010-04-30.
- ↑ Monardes, Nicholas and Translated into English by J. Frampton (1925). Joyfull Newes out of the Newe Founde Worlde. New York, NY: Alfred Knopf.
- ↑ Gaedcke, F. (1855). Ueber das Erythroxylin, dargestellt aus den Blättern des in Südamerika cultivirten Strauches Erythroxylon Coca. Archiv der Pharmazie 132 (2): 141–150.
- ↑ 12.0 12.1 Albert Niemann (1860). Ueber eine neue organische Base in den Cocablättern. Archiv der Pharmazie 153 (2): 129–256.
- ↑ Harper, Douglas. Cocaine. Online Etymology Dictionary.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedHumphrey2001 - ↑ Experience Vin Mariani today | Grupo Mariani S.A. Cocanaturally.com. Retrieved 2011-01-15.
- ↑ Yentis SM, Vlassakov KV (1999). Vassily von Anrep, forgotten pioneer of regional anesthesia. Anesthesiology 90 (3): 890–5.
- ↑ Halsted W (1885). Practical comments on the use and abuse of cocaine. New York Medical Journal 42: 294–295.
- ↑ Corning JL (1885). An experimental study. New York Medical Journal 42.
- ↑ Barlow, William. "Looking Up At Down": The Emergence of Blues Culture. Temple University Press (1989), p. 207. ISBN 978-0-87722-583-6.
- ↑ Streatfeild, Dominic (2003). Cocaine: An Unauthorized Biography. Picador. ISBN 978-0-312-42226-4.
- ↑ Jeevan Vasagar: cocaine-based "wonder drug" tested on concentration camp inmates. Amphetamines.com (2002-11-19). Retrieved 2011-01-15.
- ↑ 22.0 22.1 Madge, p. 106
- ↑ 23.0 23.1 23.2 Spillane, p. 121
- ↑ 24.0 24.1 24.2 Spillane, p. 91
- ↑ 25.0 25.1 Madge, p. 84
- ↑ 26.0 26.1 Spillane, p. 93
- ↑ 27.0 27.1 Gootenberg|Gootenberg, p. 33
- ↑ Madge, p. 102
- ↑ Spillane, p. 94
- ↑ 30.0 30.1 Madge, p. 85
- ↑ 31.0 31.1 Madge, p. 89
- ↑ Madge, p. 88
- ↑ Spillane, p. 120
- ↑ Madge, p. 90
- ↑ Madge, p. 91
- ↑ 36.0 36.1 Spillane, p. 119
- ↑ Spillane, p. 111
- ↑ 38.0 38.1 38.2 38.3 Gootenberg|Gootenberg, p. 35
- ↑ Madge, p. 82
- ↑ 40.0 40.1 Gootenberg|Gootenberg, p. 37
- ↑ Madge, p. 107
- ↑ 42.0 42.1 42.2 Gootenberg|Gootenberg, p. 40
- ↑ WHO/UNICRI (1995). WHO Cocaine Project. Retrieved 2012-06-08.
- ↑ "Cocaine use doubles in a decade", Sydney Morning Herald, 15 October 2010. Retrieved 19 October 2010.
- ↑ EMCDDA (2007). EMCDDA Retail Cocaine Purity Study.
- ↑ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 9th edition, Biomedical Publications, Seal Beach, CA, 2011, pp. 390–394.
- ↑ Venkatratnam, Abhishek and Nathan H. Lents (July 2011). Zinc Reduces the Detection of Cocaine, Methamphetamine, and THC by ELISA Urine Testing. Journal of Analytical Toxicology 35 (6): 333–340.
- ↑ (2006) "Cocaine: Seizures, 1998–2003", World Drug Report 2006 (PDF), New York: United Nations.
- ↑ Colombia. CIA World Factbook
- ↑ Peru Overtakes Colombia as Top Cocaine Producer. NBC News (31 July 2012)
- ↑ Streatfeild, Dominic (2003). Cocaine: An Unauthorized Biography. Macmillan. ISBN 978-0-312-42226-4.
- ↑ NDIC (2006). National Drug Threat Assessment 2006.
- ↑ Cocaine Seized Worldwide Highest Ever in 2011. Narcoleaks
- ↑ Colombia. State.gov. Retrieved 2013-03-26.
- ↑ Field Listing – Illicit drugs (by country). Cia.gov. Retrieved 2011-01-15.
- ↑ Jacobson, Robert (2005) Illegal Drugs: America's Anguish. Farmington Hills, MI: Thomson Gale, ISBN 1414404190.
- ↑ "Coast Guard hunts drug-running semi-subs", CNN, 2008-03-20. Retrieved 2008-03-20.
- ↑ Meth Info. methproject.org
- ↑ Drugs of Abuse. City of Denison Iowa. Retrieved 2011-11-13.
- ↑ "Drugs", The Economist, June 28, 2007, Prices: USA around $110/g, Israel/ Germany/ Britain around $46/g, Colombia $2/g, New Zealand recordbreaking $714.30/g.
- ↑ European Monitoring Centre for Drugs and Drug Addiction (2008). Annual report: the state of the drugs problem in Europe. Luxembourg: Office for Official Publications of the European Communities. ISBN 978-92-9168-324-6.
ReferencesISBN links support NWE through referral fees
- Botany Central. 2013. Erythroxylum coca: The coca plant. Botany Central. Retrieved August 6, 2013.
- Boucher, D. H. 1991. Cocaine and the coca plant. BioScience 41(2): 72-76.
- Casale, J. F., and R. F. X. Klein. 1993. Illicit production of cocaine. Forensic Science Review 5: 95-107. Retrieved August 14, 2013.
- Cortes, R. 2013. The condemned coca leaf. NY Daily News January 13, 2013. Retrieved August 11, 2013.
- Casale, J. F., and R. F. X. Klein. 1993. Illicit production of cocaine. Forensic Science Review 5: 95-107. Retrieved August 14, 2013.
- Boucher, D. H. 1991. Cocaine and the coca plant. BioScience 41(2): 72-76.
- Casale, J. F., and R. F. X. Klein. 1993. Illicit production of cocaine. Forensic Science Review 5: 95-107. Retrieved August 14, 2013.
- Cortes, R. 2013. The condemned coca leaf. NY Daily News January 13, 2013. Retrieved August 11, 2013.
- Inciardi,J. A. 1992. The War on Drugs II: The Continuing Epic of Heroin, Cocaine, Crack, Crime, AIDS, and Public Policy. Mayfield. ISBN 1559340169.
- Nathanson, J. A., E. J. Hunnicutt, L. Kantham, and C. Scavone. 1993. Cocaine as a naturally occurring insecticide. Proc. Nat. Acad. Sci. 90: 9645-9648.
- Plowman, T, and L. Rivier. 1983. Cocaine and Cinnamoylcocaine content of thirty-one species of Erythroxylum (Erythroxylaceae)". Annals of Botany 51: 641–659.
- Royal Botanic Gardens, Kew. 1985. Entry for Erythroxylum coca Lam. [family ERYTHROXYLACEAE]. JSTOR. Retrieved August 6, 2013.
.[8]
- Nathanson, J. A., E. J. Hunnicutt, L. Kantham, and C. Scavone. 1993. Cocaine as a naturally occurring insecticide. Proc. Nat. Acad. Sci. 90: 9645-9648.
- Office of Diversion Control (ODC). 2011. Importers Notice of Registration - 2011. Drug Enforcement Agency, U.S. Department of Justice. Retrieved August 14, 2013.
Cocaine-related sudden death not so rare
January 13, 2010 Lisa Nainggolan
inShare
http://www.theheart.org/article/1039143.do
National Institutes of Health/National Institute On Drug Abuse (NIH/NIDA) (2003, May 6). Sigma Receptors Play Role In Cocaine-induced Suppression Of Immune System. ScienceDaily. Retrieved September 5, 2013, from http://www.sciencedaily.com /releases/2003/05/030506073758.htm
D. Nutt, L. A. King, W. Saulsbury, and C. Blakemore, "Development of a Rational Scale to Assess the Harm of Drugs of Potential Misuse". The Lancet 369, iss. 9566 (2007): 1047–1053. Retrieved September 3, 2013
- Plowman, T, and L. Rivier. 1983. Cocaine and Cinnamoylcocaine content of thirty-one species of Erythroxylum (Erythroxylaceae)". Annals of Botany 51: 641–659.
Cocaine misuse doubles both the risks of hemorrhagic and ischemic strokes,[15] as well as increases the risk of other infarctions, such as myocardial infarction.[16]
[17][18]Cite error: Closing </ref> missing for <ref> tag
unodc 2007. [19]
- United Nations Office of Drugs and Crime (USODC). 2011. The transatlantic cocaine market: Research paper. United Nations. Retrieved August 13, 2013.
[20][21]
- WebMD. 2013a. Cocaine use and its effects. WebMD. Retrieved August 12, 2013.
- WebMD. 2013b. Find a vitamin or supplement: Coca. WebMD. Retrieved August 13, 2013.
[22] which can be life-threatening.[22]
[27] because of methylecgonidine's effects on lung tissue[28] and liver tissue.[29]
Bibliography
- Gootenberg, Paul, Ed. (1999). Cocaine: Global Histories. London: Routledge. ISBN 0203026462.
- Madge, Tim (2001). White Mischief: A Cultural History of Cocaine. Edinburgh: Mainstream Publishing Company.
- Spillane, Joseph F. (2000). Cocaine: From Medical Marvel to Modern Menace in the United States, 1884–1920. Baltimore and London: The John Hopkins University Press. ISBN 0801862302.
Further reading
- Feiling, Tom (2009). The Candy Machine: How Cocaine Took Over the World. London: Penguin. ISBN 978-0-14-103446-1.
External links
- A look at the Evidence for Cocaine in Mummies
- EMCDDA drugs profile: Cocaine(2007)
- Erowid -> Cocaine Information — A collection of data about cocaine including dose, effects, chemistry, legal status, images and more.
- Slang Dictionary for Cocaine.
- Cocaine content of plants
- Cocaine – The History and the Risks at h2g2
- Cocaine Frequently Asked Questions
- U.S. National Library of Medicine: Drug Information Portal – Cocaine
- Cocaine Market Data and Value-Havocscope Black Markets Data on cocaine trafficking worldwide.
- Cocaine bound to proteins in the PDB
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
- ↑ Aggrawal, Anil. Narcotic Drugs. National Book Trust, India (1995), p. 52-3. ISBN 978-81-237-1383-0.
- ↑ Baigent, Michael (2003). Physical complications of substance abuse: what the psychiatrist needs to know. Curr Opin Psychiatry 16 (3): 291–296.
- ↑ (1981). Cocaine pharmacokinetics in humans. Journal of Ethnopharmacology 3 (2–3): 353–66.; Jones, supra note 19; Wilkinson et al., Van Dyke et al.
- ↑ Bonkovsky HL, Mehta S (2001). Hepatitis C: a review and update. J. Am. Acad. Dermatol. 44 (2): 159–82.
- ↑ (2002) Biological Psychiatry By H. A. H. D'haenen, Johan A. den Boer, Paul Willner. Books.google.com. ISBN 978-0-471-49198-9. Retrieved 2010-03-09.
- ↑ (2009). Alteration of blood-brain barrier function by methamphetamine and cocaine.. Cell and tissue research 336 (3): 385–392.
- ↑ Lowinson, Joyce, H and Ruiz, Pedro. Millman, Robert B. (2004). Substance abuse: a comprehensive textbook. Lippincott Williams & Wilkins; Fourth edition. ISBN 978-0-7817-3474-5. Retrieved May 9, 2011.
- ↑ Kolbrich EA, Barnes AJ, Gorelick DA, Boyd SJ, Cone EJ, Huestis MA (2006). Major and minor metabolites of cocaine in human plasma following controlled subcutaneous cocaine administration. J Anal Toxicol 30 (8): 501–10.
- ↑ Lowinson, Joyce, H and Ruiz, Pedro. Millman, Robert B. (2004). Substance abuse: a comprehensive textbook. Lippincott Williams & Wilkins; Fourth edition. ISBN 978-0-7817-3474-5. Retrieved May 9, 2011.
- ↑ Lluch J, Rodríguez-Arias M, Aguilar MA, Miñarro J (2005). Role of dopamine and glutamate receptors in cocaine-induced social effects in isolated and grouped male OF1 mice. Pharmacol. Biochem. Behav. 82 (3): 478–87.
- ↑ George, Nelson. "Hip Hop America". 1998. Viking Penguin.(Page 40)
- ↑ L. Pagliaro and A. M. Pagliaro, Pagliaros’ Comprehensive Guide to Drugs and Substances of Abuse (Washington, D.C.: American Pharmacists Association, 2004). ISBN 1582120668.
- ↑ Spanagel R, Weiss F (1999). The dopamine hypothesis of reward: past and current status. Trends Neurosci. 22 (11): 521–7.
- ↑ Margaret Reist. "A rose by another name: crack pipe", January 16, 2005. Retrieved 2009-08-21.
- ↑ Susan Jeffrey and Charles Vega (Released: 04/16/2007; Reviewed and Renewed: 04/17/2008). Stimulant Abuse May Increase Stroke Among Young Adults. Retrieved 2011-02-06.
- ↑ Vasica G, Tennant CC (2002). Cocaine use and cardiovascular complications. Med. J. Aust. 177 (5): 260–2.
- ↑ Trozak D, Gould W (1984). Cocaine abuse and connective tissue disease. J Am Acad Dermatol 10 (3).
- ↑ Ramón Peces (1999). Antiglomerular Basement Membrane Antibody-Mediated Glomerulonephritis after Intranasal Cocaine Use. Nephron 81 (4): 434–438.
- ↑ (2007) World Drug Report 2007 (PDF), New York: United Nations.
- ↑ Jared A. Jaffe (2006). Chronic Nephropathies of Cocaine and Heroin Abuse: A Critical Review. Clinical Journal of the American Society of Nephrology 1 (4): 655–67.
- ↑ Fokko J. van der Woude (2000). Cocaine use and kidney damage. Nephrology Dialysis Transplantation 15 (3): 299–301.
- ↑ 22.0 22.1 Zhao, Wei (2008). Mechanisms Mediating Sex Differences in the Effects of Cocaine. ProQuest. ISBN 0549994580. Retrieved September 25, 2012.
- ↑ World Health Organization (2004). Neuroscience of psychoactive substance use and dependence
- ↑ World Health Organization (2007). International medical guide for ships
- ↑ Filip M, Bubar MJ, Cunningham KA (2004). Contribution of serotonin (5-hydroxytryptamine; 5-HT) 5-HT2 receptor subtypes to the hyperlocomotor effects of cocaine: acute and chronic pharmacological analyses. J. Pharmacol. Exp. Ther. 310 (3): 1246–54.
- ↑ Carta M, Allan AM, Partridge LD, Valenzuela CF (2003). Cocaine inhibits 5-HT3 receptor function in neurons from transgenic mice overexpressing the receptor. Eur. J. Pharmacol. 459 (2–3): 167–9.
- ↑ (2003-10-15) Pharmacokinetics and Pharmacodynamics of Methylecgonidine, a Crack Cocaine Pyrolyzate. Journal of Pharmacology and Experimental Therapeutics 307 (3): 1179–87.
- ↑ Yang Y, Ke Q, Cai J, Xiao YF, Morgan JP (2001). Evidence for cocaine and methylecgonidine stimulation of M2 muscarinic receptors in cultured human embryonic lung cells. Br. J. Pharmacol. 132 (2): 451–60.
- ↑ Fandiño AS, Toennes SW, Kauert GF (2002). Studies on hydrolytic and oxidative metabolic pathways of anhydroecgonine methyl ester (methylecgonidine) using microsomal preparations from rat organs. Chem. Res. Toxicol. 15 (12): 1543–8.
- ↑ (2009) Principles of addiction medicine. Lippincott Williams & Wilkins. ISBN 0781774772. Retrieved December 29, 2012.
- ↑ Nora D. Volkow (2000). Effects of route of administration on cocaine induced dopamine transporter blockade in the human brain. Life Sciences 67 (12): 1507–1515.