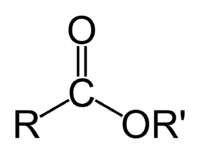
Ester
In chemistry, esters are organic compounds in which an organic group (symbolized by R' in this article) replaces the hydrogen atom of a hydroxyl group in an oxoacid. A molecule of an oxoacid has an -OH (OH) group from which the hydrogen (H) can dissociate as an H+ ion. Many esters have distinctive odors and flavors, leading to their use as artificial flavorings and fragrances.
The most common esters are the carboxylate esters, where the acid in question is a carboxylic acid. For example, if the acid is acetic acid, the ester is called an acetate. Stable esters such as carbamates, RO(CO)NHR', and dialkyl carbonates, RO(CO)OR, can be formed from unstable carbamic acid or carbonic acid, respectively. Esters may also be formed with inorganic acids; for example, dimethyl sulfate is an ester, and sometimes called "sulfuric acid, dimethyl ester."
Esters are named similarly to salts; although they are not composed of cations and anions as salts are, the terminology used to refer to them follows the same pattern: a more electropositive part followed by a more electronegative part.
An ester can be thought of as a product of a condensation reaction of an acid (usually an organic acid) and an alcohol (or phenol compound), although there are other ways to form esters. Condensation is a type of chemical reaction in which two molecules are joined together and eliminate a small molecule, in this case two-OH groups are joined eliminating a water molecule. A condensation reaction to form an ester is called esterification. Esterification can be catalysed by the presence of H+ ions. Sulfuric acid is often used as a catalyst for this reaction. The name ester is derived from the German Essig-Äther, an old name for acetic acid ethyl ester (ethyl acetate).
Nomenclature
Esters can be produced by an equilibrium reaction between an alcohol and a carboxylic acid. The ester is named according to the alkyl group (the part from the alcohol) and then the alkanoate (the part from the carboxylic acid) which make it up.[1] For example, the reaction between methanol and butyric acid yields the ester methyl butyrate C3H7-COO-CH3 (as well as water). The simplest ester is methyl formate (H-COO-CH3, also called methyl methanoate).
For esters derived from the simplest carboxylic acids, the traditional names are recommended by the International Union of Pure and Applied Chemistry (IUPAC),[2]. Examples of names (from lower molecular-weight acids) are: formate (from formic acid), acetate (acetic acid), propionate (from propionic acid), butyrate (butyric acid), and so forth. For esters from higher molecular-weight acids, the alkane name with an -oate ending is generally preferred—for example, hexanoate from hexanoic acid. Common esters of aromatic acids include benzoates such as methyl benzoate, and phthalates, with substitution allowed in the name.
Physical properties
Esters participate in hydrogen bonds as hydrogen-bond acceptors, but cannot act as hydrogen-bond donors, unlike their parent alcohols. This ability to participate in hydrogen bonding makes them more water-soluble than their parent hydrocarbons. However, the limitations on their hydrogen bonding also make them more hydrophobic than either their parent alcohols or parent acids.
Their lack of hydrogen-bond-donating ability means that ester molecules cannot hydrogen-bond to each other, which makes esters generally more volatile than a carboxylic acid of similar molecular weight. This property makes them very useful in organic analytical chemistry: unknown organic acids with low volatility can often be esterified into a volatile ester, which can then be analyzed using gas chromatography, gas liquid chromatography, or mass spectrometry.
Odors and flavors
Examples of esters with distinctive odors and flavors are given below.
| chemical name | molar mass (g/mol) |
♦ | ♦ | ♦ | image | odor |
|---|---|---|---|---|---|---|
| allyl hexanoate | pineapple | |||||
| benzyl acetate | 150.18 | 1 | 1 | 0 | pear, strawberry, jasmine | |
| bornyl acetate | pine tree flavor | |||||
| butyl butyrate | 144.21 | 2 | 2 | 0 | pineapple | |
| ethyl acetate | 88.12 | 1 | 3 | 0 | nail polish remover, model paint, model airplane glue | |
| ethyl butyrate | 116.16 | banana, pineapple, strawberry | ||||
| ethyl hexanoate | strawberry | |||||
| ethyl cinnamate | 176.21 | cinnamon | ||||
| ethyl formate | 74.08 | lemon, rum, strawberry | ||||
| ethyl heptanoate | 158.27 | apricot, cherry, grape, raspberry | ||||
| ethyl isovalerate | apple | |||||
| ethyl lactate | 118.13 | 1 | 1 | 0 | butter cream | |
| ethyl nonanoate | grape | |||||
| ethyl valerate | 130.18 | 1 | 3 | 0 | apple | |
| geranyl acetate | 196.29 | 0 | 1 | 0 | geranium | |
| geranyl butyrate | cherry | |||||
| geranyl pentanoate | apple | |||||
| isobutyl acetate | 116.16 | 1 | 3 | 0 | cherry, raspberry, strawberry | |
| isobutyl formate | raspberries | |||||
| isoamyl acetate | 130.19 | pear, banana (flavoring in Pear Drops) | ||||
| isopropyl acetate | 102.1 | 1 | 3 | 0 | fruity | |
| linalyl acetate | lavender, sage | |||||
| linalyl butyrate | peach | |||||
| linalyl formate | apple, peach | |||||
| methyl acetate | 74.08 | 1 | 3 | 0 | peppermint | |
| methyl anthranilate | 151.165 | grape, jasmine | ||||
| methyl benzoate | 136.15 | fruity, ylang ylang, feijoa fruit | ||||
| methyl benzyl acetate | cherry | |||||
| methyl butyrate | 102.13 | pineapple, apple | ||||
| methyl cinnamate | strawberry | |||||
| methyl pentanoate | 116.16 | flowery | ||||
| methyl phenyl acetate | honey | |||||
| methyl salicylate (oil of wintergreen) | 152.1494 | root beer, wintergreen, Germolene™ and Ralgex™ ointments (UK) | ||||
| nonyl caprylate | orange | |||||
| octyl acetate | 172.27 | fruity-orange | ||||
| octyl butyrate | parsnip | |||||
| amyl acetate (pentyl acetate) | 130.19 | apple, banana | ||||
| pentyl butyrate (amyl butyrate) | 158.24 | apricot, pear, pineapple | ||||
| pentyl hexanoate (amyl caproate) | apple, pineapple | |||||
| pentyl pentanoate (amyl valerate) | 172.15 | apple | ||||
| propyl ethanoate | pear | |||||
| propyl isobutyrate | rum | |||||
| terpenyl butyrate | cherry |
Ester synthesis
Esters can be prepared in the laboratory in several ways:
- by esterification of carboxylic acid derivatives and alcohols
- by transesterifications between other esters
- by Dieckmann condensation or Claisen condensation of esters carrying acidic α-protons
- by Favorskii rearrangement of α-haloketones in presence of base
- by nucleophilic displacement of alkyl halides with carboxylic acid salts
- by Baeyer-Villiger oxidation of ketones with peroxides
- by Pinner reaction of nitriles with an alcohol
Ester reactions
Esters react in a number of ways:
- Esters may undergo hydrolysis - the breakdown of an ester by water. This process can be catalyzed both by acids and bases. The base-catalyzed process is called saponification. The hydrolysis yields an alcohol and a carboxylic acid or its carboxylate salt.
- Esters also react if heated with primary or secondary amines, producing amides.
- Phenyl esters react to hydroxyarylketones in the Fries rearrangement.
- Di-esters such as diethyl malonate react as nucleophile with alkyl halides in the malonic ester synthesis after deprotonation.
- Specific esters are functionalized with an α-hydroxyl group in the Chan rearrangement
- Esters are converted to isocyanates through intermediate hydroxamic acids in the Lossen rearrangement.
See also
Notes
- ↑ Specific Classes of Compounds: Salts and Esters. (IUPAC naming of esters.) Retrieved May 24, 2007.
- ↑ Carboxylic acids and related groups: Unsubstituted parent structures. (IUPAC parent groups using traditional names.) Retrieved May 24, 2007.
ReferencesISBN links support NWE through referral fees
- McMurry, John. Organic Chemistry, 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052.
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry, 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692.
- Solomons, T.W. Graham, and Fryhle, Craig B. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998.
External links
All links retrieved March 20, 2024.
- Simon Cotton Ethyl acetate. Uppingham School, Rutland, UK.
- Project shum Making an Ester Homepage.
- IUPAC Compendium of Chemical Terminology - the Gold Book. International Union of Pure and Applied Chemistry.
| Functional groups |
|---|
| Chemical class: Alcohol • Aldehyde • Alkane • Alkene • Alkyne • Amide • Amine • Azo compound • Benzene derivative • Carboxylic acid • Cyanate • Ester • Ether • Haloalkane • Imine • Isocyanide • Isocyanate • Ketone • Nitrile • Nitro compound • Nitroso compound • Peroxide • Phosphoric acid • Pyridine derivative • Sulfone • Sulfonic acid • Sulfoxide • Thioether • Thiol • Toluene derivative |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.