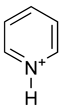
Pyridine
| Pyridine | |
|---|---|
| |
| IUPAC name | Pyridine |
| Other names | Azabenzene Azine py |
| Identifiers | |
| CAS number | [] |
| SMILES | C1=NC=CC=C1 |
| Properties | |
| Molecular formula | C5H5N |
| Appearance | colourless liquid |
| Density | 0.9819 g/cm³, liquid |
| Melting point |
−41.6 °C |
| Boiling point |
115.2 °C |
| Solubility in water | Miscible |
| Viscosity | 0.94 cP at 20 °C |
| Hazards | |
| EU classification | Flammable (F) Harmful (Xn) |
| NFPA 704 |
|
| Flash point | 21 °C |
| Related Compounds | |
| Related amines | Picoline Quinoline |
| Related compounds | Aniline Pyrimidine |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Pyridine is a fundamentally important chemical compound with the formula C5H5N. It is a liquid with a distinctively putrid, fishy odor. Its molecules have a six-membered ring structure that can be found in many compounds, including the nicotinamides.
This compound has numerous applications. It is both a versatile solvent and a building block for a variety of other organic compounds. It is a starting material in the manufacture of insecticides, herbicides, pharmaceuticals, food flavorings, dyes, rubber chemicals, adhesives, paints, explosives, and disinfectants. In addition, it is a denaturant for antifreeze mixtures and is sometimes used as a ligand in coordination chemistry.
Properties
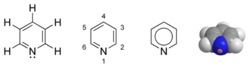
Pyridine can be classified as a heterocyclic aromatic organic compound. Each molecule of pyridine is a six-membered ring related to the structure of benzene—one CH group in the benzene ring is replaced by a nitrogen atom.
Pyridine has a lone pair of electrons at the nitrogen atom. Because this lone pair is not delocalized into the aromatic system of "pi" electrons, pyridine is basic, with chemical properties similar to those of tertiary amines.
Pyridine is protonated by reaction with acids and forms a positively charged aromatic polyatomic ion called a pyridinium cation. This cation is the conjugate acid of pyridine and its pKa has a value of 5.30.
The bond lengths and bond angles in pyridine and the pyridinium ion are almost identical.[1] This is because protonation of pyridine does not affect the aromatic pi system.
The structure of the pyridine molecule makes it polar. It is thus a polar but aprotic solvent. It is fully miscible with a broad range of other solvents, including hexane and water.
Occurrence and synthesis
Pyridine was originally isolated industrially from crude coal tar. There are now many methods in industry and in the laboratory for the synthesis of pyridine and its derivatives.[2]
Currently, pyridine is synthesized from acetaldehyde, formaldehyde, and ammonia, a process that involves acrolein as an intermediate:
- CH2O + NH3 + 2 CH3CHO → C5H5N + 3 H2O
By substituting other aldehydes for acetaldehyde, one can obtain alkyl and aryl substituted pyridines. 26,000 tons were produced worldwide in 1989.[3]
Additional methods of synthesis
- The Hantzsch pyridine synthesis is a multicomponent reaction involving formaldehyde, a keto-ester and a nitrogen donor.
- Other examples of the pyridine class can be formed by the reaction of 1,5-diketones with ammonium acetate in acetic acid followed by oxidation. This reaction is called the "Kröhnke pyridine synthesis."
- Pyridinium salts can be obtained in the Zincke reaction.
- The "Ciamician-Dennstedt Rearrangement" (1881) is the ring-expansion of pyrrole with dichlorocarbene to 3-chloropyridine and HCl[4]
- In the "Chichibabin pyridine synthesis" (Aleksei Chichibabin, 1906) the reactants are three equivalents of a linear aldehyde and ammonia
Organic reactions
In organic reactions, pyridine behaves as a tertiary amine with protonation, alkylation, acylation and N-oxidation at the nitrogen atom. It also behaves as an aromatic compound with nucleophilic substitutions.
- Pyridine is a good nucleophile (with a donor number of 33.1). It is easily attacked by alkylating agents to give N-alkylpyridinium salts.
- Nucleophilic aromatic substitution takes place at C2 and C4 for example in the Chichibabin reaction of pyridine with sodium amide to 2-aminopyridine. In the Emmert reaction (B. Emmert, 1939) pyridine is reacted with a ketone in presence of aluminium or magnesium and mercuric chloride to the carbinol also at C2.[5]
Applications
- Pyridine is widely used as a versatile solvent. Deuterated pyridine, called pyridine-d5, is a common solvent for1H NMR spectroscopy.
- It is important in industrial organic chemistry, both as a fundamental building block and as a solvent and reagent in organic synthesis.[6] It is used as a solvent in Knoevenagel condensations.
- Pyridine-borane, C5H5NBH3 (m.p. 10–11°C), is a mild reducing agent with improved stability compared to sodium borohydride (NaBH4) in protic solvents and improved solubility in aprotic organic solvents.
- Pyridine-sulfur trioxide, C5H5NSO3 (mp 175 °C), is a sulfonation agent used to convert alcohols to sulfonates, which in turn undergo C-O bond scission (break-up) upon reduction with hydride agents.
- It is a starting material in the synthesis of compounds used as intermediates in making insecticides, herbicides, pharmaceuticals, food flavorings, dyes, rubber chemicals, adhesives, paints, explosives, and disinfectants.
- It is used as a denaturant for antifreeze mixtures.
- It is sometimes used as a ligand in coordination chemistry.
Safety and environmental issues
Pyridine is toxic. (Its oral lethal dose LD50 in rats was found to be 891 mg kg–1). It is volatile and can be absorbed through the skin. Available data indicate that "exposure to pyridine in drinking-water led to reduction of sperm motility at all dose levels in mice and increased estrous cycle length at the highest dose level in rats".[7]
Currently, its evaluations as a possible carcinogenic agent showed there is inadequate evidence for the carcinogenicity of pyridine in humans, albeit there is limited evidence of carcinogenic effects on animals.
Effects of acute pyridine intoxication include dizziness, headache, nausea, and anorexia. Further symptoms include abdominal pain and pulmonary congestion. Though resistant to oxidation, pyridine is readily degraded by bacteria, releasing ammonium and carbon dioxide as terminal degradation products.[8]
Related compounds
Structurally or chemically related compounds are:
- DMAP, short for 4-dimethylaminopyridine
- Bipyridine and viologen are simple polypyridine compounds consisting of two pyridine molecules joined by a single bond
- Terpyridine, a molecule of three pyridine rings connected together by two single bonds.
- Quinoline and Isoquinoline have pyridine and a benzene ring fused together.
- Aniline is a benzene derivative with an attached NH2 group and not a pyridine
- Diazines are compounds with one more carbon replaced by nitrogen such as Pyrazine and Pyramidine
- Triazines are compounds with two more carbons replaced by nitrogen and a tetrazine has four nitrogen atoms
- 2,6-Lutidine is a trivial name for 2,6-dimethylpyridine.
- Collidine is the trivial name for 2,4,6-trimethylpyridine.
- Pyridinium p-toluenesulfonate (PPTS) is a salt formed by proton exchange between pyridine and p-toluenesulfonic acid
- 2-Chloropyridine is a toxic environmentally significant component of the breakdown of the pesticide imidacloprid.
Notes
- ↑ T.M. Krygowski, H. Szatyowicz, and J.E. Zachara, How H-bonding Modifies Molecular Structure and -Electron Delocalization in the Ring of Pyridine/Pyridinium Derivatives Involved in H-Bond Complexation, J. Org. Chem (70:22:8859-8865). Retrieved November 28, 2007.
- ↑ T.L. Gilchrist, Heterocyclic Chemistry (Hoboken, NJ: Wiley, 1997). ISBN 0470204818
- ↑ Shinkichi Shimizu, Nanao Watanabe, Toshiaki Kataoka, Takayuki Shoji, Nobuyuki Abe, Sinji Morishita, Hisao Ichimura, Ullmann's Encyclopedia of Industrial Chemistry: Pyridine and Pyridine Derivatives (Hoboken, NJ: John Wiley & Sons, 1993). ISBN 3527303855
- ↑ drugfuture.com, Ciamician-Dennstedt Rearrangement. Retrieved November 2, 2007.
- ↑ Charles H. Tilford, Robert S. Shelton, and M. G. van Campen, Histamine Antagonists. Basically Substituted Pyridine Derivatives, J. Am. Chem (70:12:4001-4009, 1948).
- ↑ A.R. Sherman, Encyclopedia of Reagents for Organic Synthesis: Pyridine (Hoboken, NJ: J. Wiley & Sons, 2004). ISBN 0471936235
- ↑ International Agency for Research on Cancer (IARC), Pyridine Summary & Evaluation. IPCS INCHEM. Retrieved November 2, 2007.
- ↑ G.K. Sims and E.J. O'Loughlin. Degradation of pyridines in the environment, CRC Critical Reviews in Environmental Control (19:4:309-340, 1989).
ReferencesISBN links support NWE through referral fees
- McMurry, John. 2004. Organic Chemistry. Belmont, CA: Brooks/Cole. ISBN 0534420052
- Morrison, Robert T. and Robert N. Boyd. 1992. Organic Chemistry. Englewood Cliffs, NJ: Prentice Hall. ISBN 0-13-643669-2
- Solomons, T.W. Graham and Craig B. Fryhle. 2004. Organic Chemistry. Hoboken, NJ: John Wiley. ISBN 0471417998
External links
All links retrieved December 6, 2022.
| Functional groups |
|---|
| Chemical class: Alcohol • Aldehyde • Alkane • Alkene • Alkyne • Amide • Amine • Azo compound • Benzene derivative • Carboxylic acid • Cyanate • Ester • Ether • Haloalkane • Imine • Isocyanide • Isocyanate • Ketone • Nitrile • Nitro compound • Nitroso compound • Peroxide • Phosphoric acid • Pyridine derivative • Sulfone • Sulfonic acid • Sulfoxide • Thioether • Thiol • Toluene derivative |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.