Glucagon
Glucagon is a catabolic (part of metabolism that breaks down larger molecules) protein hormone that works together with its anabolic counterpart, insulin, to regulate the levels of glucose in the bloodstream. It is produced by the alpha cells of the islets of Langerhans in the pancreas of vertebrates. Working to opposite effect from insulin, glucagon acts to increase blood sugar levels by stimulating the breakdown of liver glycogen to glucose (glycogenolysis), increasing release of glucose from the liver into the blood, preventing the liver from storing glucose, and increasing glucose formation in the liver from dietary protein (amino acids) and fats (Bender and Bender 2005; MedNet 2001). Glucagon also transiently paralyzes the muscles of the intestines.
Glycogen is the principal storage form of glucose in animal cells. It is a large, branched polymer of linked glucose residues (portions of larger molecules) that can be readily mobilized as an energy source, increasing the amount of glucose immediately available to the organism between meals and during muscular activity. Since the brain relies on glucose as its preferred fuel, the ability to maintain a steady supply of glucose, which is the major sugar circulating in the blood of higher animals, is crucial to survival.
The catabolic/anabolic pair of glucagon and insulin, both of them hormones secreted from the islets of Langerhans, circulate in the blood at the constantly fluctuating levels required to maintain proper levels of glucose in the blood even as the body's nutritional intake and energy expenditure pass through several highs and lows each day. The system involving these two hormones in maintaining the glucose balance shares significant similarities with the Taoist model of the opposite yet complementary and interdependent forces of yin-yang, which are both together integral to the natural order of things. Similarly, glucagon and insulin are necessary to create overall harmony and balance in the living organism.
Structure
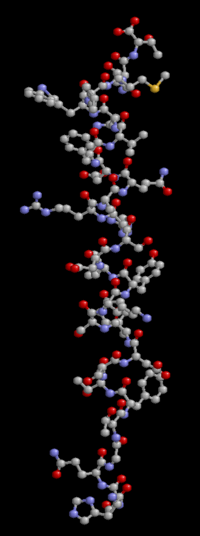
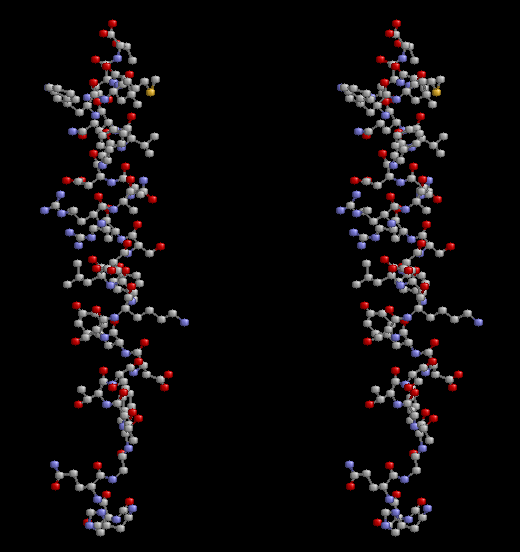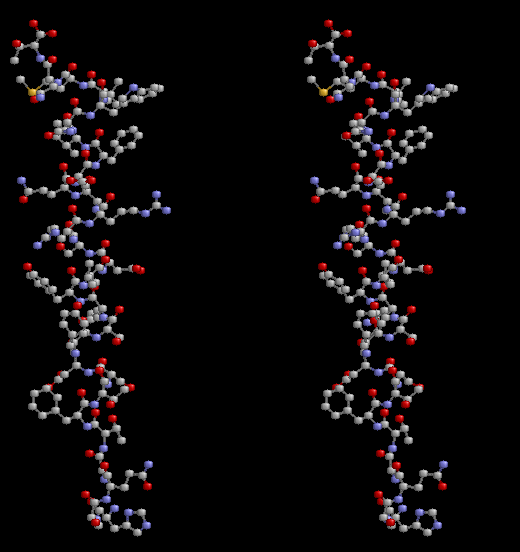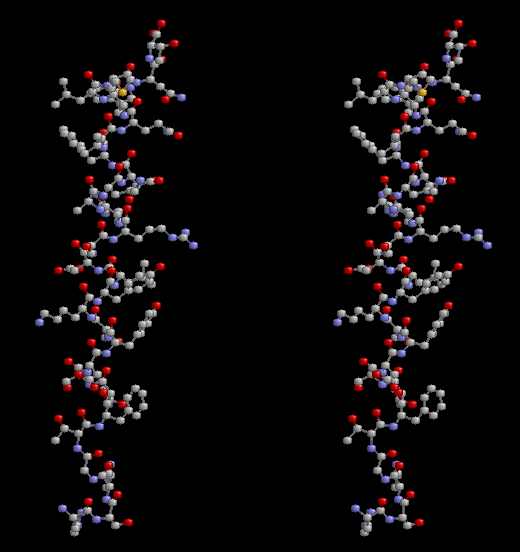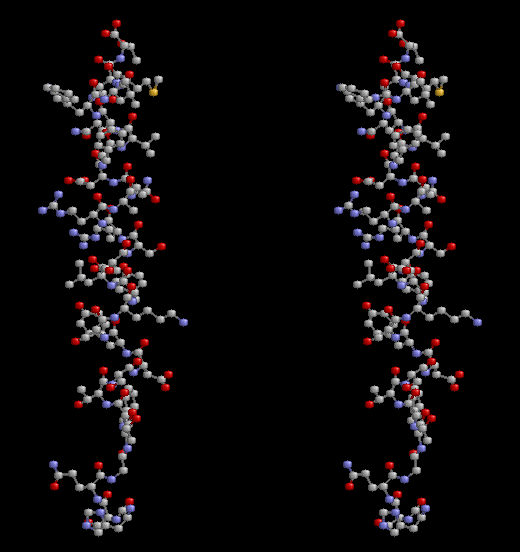
Glucagon is a 29-amino acid polypeptide (chains of amino acids linked together via peptide bonds and having a defined sequence). Its primary structure in humans is: NH2-His-Ser-Gln-Gly-Thr-Phe- Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Ser- Arg-Arg-Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu- Met-Asn-Thr-COOH.
The polypeptide has a molecular weight of 3485 daltons.
Physiology
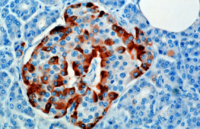
Glucagon is synthesized and secreted from alpha cells (α-cells) of the islets of Langerhans, which are located in the endocrine portion of the pancreas.
The pancreas, found in vertebrates near the stomach and small intestine, is a gland that has both an endocrine function and an exocrine function. The majority of pancreatic cells are exocrine and secrete one to three liters of digestive fluid per day. In humans, the endocrine portion of this organ consists of about one million islets of Langerhans scattered throughout the organ and amounting to only one to three percent of the organ weight. The islets are a compact collection of endocrine cells arranged in clusters and cords crisscrossed by a dense network of capillaries. The capillaries are lined by layers of endocrine cells in direct contact with them by either cytoplasmic processes or by direct apposition. The cells release their hormones, without ducts, directly into the capillaries.
In the islet of Langerhans, there are four main types of cells, with the alpha cells making up about 15 to 20 percent of inlet cells. The alpha cells are located in the outer rim of the islets.
Regulatory mechanism
Increased secretion of glucagon is caused by:
- Decreased plasma glucose
- Increased catecholamines—norepinephrine and epinephrine
- Increased plasma amino acids (to protect from hypoglycemia (low blood sugar) if an all protein meal is consumed)
- Sympathetic nervous system
- Acetylcholine
- Cholecystokinin
Decreased secretion of glucagon (inhibition) is caused by:
- Somatostatin
- Insulin
Mechanism of action
Glucagon binds to the glucagon receptor, a G protein-coupled receptor located in the plasma membrane. The conformation change in the receptor activates G proteins, a heterotrimeric protein with alpha, beta, and gamma subunits. The subunits breakup under GTP hydrolysis and the alpha subunit specifically activates the next enzyme in the cascade, adenylate cyclase.
Adenylate cyclase manufactures cAMP (cyclical AMP), which activates protein kinase A (cAMP-dependent protein kinase). This enzyme in turn activates phosphorylase B kinase, which in turn, phosphorylates phosphorylase B. Phosphorylase B is the enzyme responsible for the release of glucose-1-phosphate from glycogen polymers.
Function
Glucagon helps maintain the level of glucose in the blood. It does this by binding to glucagon receptors on hepatocytes (cells making up about the bulk of the cytoplasmic mass of the liver), causing the liver to release glucose—stored in the form of glycogen—through a process known as glycogenolysis. As these stores become depleted, glucagon then encourages the liver to synthesize additional glucose by gluconeogenesis. This glucose is released into the bloodstream. Both of these mechanisms lead to glucose release by the liver, preventing the development of hypoglycemia (lower than normal level of sugar—glucose—in the blood). Glucagon also works against storage of glucose in the liver.
Glucagon increases the free fatty acids and ketoacids into the blood and increases urea production. It may also be involved in other functions as well.
History
In the 1920s, Kimball and Murlin studied pancreatic extracts and found an additional substance with hyperglycemic properties. They described glucagon in 1923 (Kimball and Murlin 1923). The amino acid sequence of glucagon was described in the late-1950s (Bromer et al. 1957). A more complete understanding of its role in physiology and disease was not established until the 1970s, when a specific radioimmunoassay was developed.
Medical uses and pathology
Commercially produced glucagon (synthetic, i.e., manufactured by humans) is made using the bacteria Escherichia coli that have been genetically engineered (MedNet 2001).
An injectable form of glucagon is vital first aid in cases of severe hypoglycemia when the victim is unconscious or for other reasons cannot take sugar orally. Symptoms of hypoglycemia include confusion, nervousness, cool skin, headache, convulsions, or coma (MedNet 2001). In these cases, it is important to raise the glucose level in the blood. The dose for an adult is typically one milligram, and the glucagon is given by intramuscular injection, and quickly raises blood glucose levels. Glucagon can also be administered intravenously.
Glucagon acts very quickly. Common side effects include headache and nausea.
Anecdotal evidence suggests a benefit of higher doses of glucagon in the treatment of overdose with beta blockers (class of drugs used particularly for the management of cardiac arrhythmias and cardioprotection after myocardial infarction). The likely mechanism of action is the increase of cAMP in the myocardium, effectively bypassing the inhibitory action of the β-adrenergic second messenger system (White 1999).
Glucogon temporarily paralyzes the muscles of the intestines and has been used during testing that requires the intestines to be flaccid (MedNet 2001).
Abnormally-elevated levels of glucagon may be caused by pancreatic tumors such as glucagonoma, symptoms of which include necrolytic migratory erythema (NME), elevated amino acids, and hyperglycemia. It may occur alone or in the context of multiple endocrine neoplasia type 1.
Media
|
|
ReferencesISBN links support NWE through referral fees
- Bender, D. A., and A. E. Bender. A Dictionary of Food and Nutrition. New York: Oxford University Press, 2005. ISBN 0198609612
- Bromer, W., L. Winn, and O. Behrens. “The amino acid sequence of glucagon V. Location of amide groups, acid degradation studies and summary of sequential evidence.” J Am Chem Soc, 79: 2807-2810, 1957.
- Kimball C., and J. Murlin. Aqueous extracts of pancreas III. Some precipitation reactions of insulin J Biol Chem, 58: 337-348, 1923. Retrieved September 16, 2007.
- MedinceNet.com (MedNet). Glucagon MedicineNet.com, 2001. Retrieved September 16, 2007.
- White, C. M. “A review of potential cardiovascular uses of intravenous glucagon administration.” J Clin Pharmacol, 39: 442-447, 1999. PMID 10234590 Retrieved September 16, 2007.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.