Difference between revisions of "Lactic acid" - New World Encyclopedia
({{Contracted}}) |
Rick Swarts (talk | contribs) |
||
| Line 33: | Line 33: | ||
''For the production of milk by mammals, see [[Lactation]].'' | ''For the production of milk by mammals, see [[Lactation]].'' | ||
| + | '''Lactic acid''' (IUPAC systematic name'''2-hydroxypropanoic acid'''), also known as '''milk acid''', is a colorless, water-soluable, liquid organic acid that plays a role in several [[biochemistry|biochemical]] processes. It is produced in muscles through anaerobic glucose metabolism, and is the bitter-tasking fermentation product of bacteria interacting with lactose (milk sugar), occuring naturally in sour milk, yourt, and cheese. cream, buttermilk, most common acid constituent of fermented milk products. Also present in various fruits and wines and in soil.. Commercially produced lactic acid is sued for flavoring, imparting a tart flavor, an as preservative, dyeing, and pharmaceuticals. | ||
| + | leather tanning, textile dyeing, making plastics, etc. | ||
| − | + | Lactic acid is a carboxylic acid—an organic (carbon-containing) acid characterized by the presence of a carboxyl group, which has the formula -C(=O)OH, usually written -COOH or -CO<sub>2</sub>H. Lactic acid has the chemical formula of [[Carbon|C]]<sub>3</sub>[[Hydrogen|H]]<sub>6</sub>[[Oxygen|O]]<sub>3</sub>. | |
| − | Lactic acid | + | ==Overview and chemistry== |
| + | Lactic acid was first isolated in 1780 by a Swedish chemist, Carl Wilhelm Scheele. It has an hydroxyl (OH) group adjacent to the carboxyl group, making it an alpha hydroxy acid (AHA). In solution, it can lose a [[proton]] from the acidic group, producing the '''lactate''' [[ion]] CH<sub>3</sub>CH(OH)COO<sup>−</sup>. It is miscible with water or ethanol, and is hygroscopic (ability of a substance to attract [[water]] molecules). | ||
| − | + | Lactic acid is chiral (cannot be superimposed on its mirror image, like the right and left hand of a person) and has two optical [[isomer]]s. One is known as <small>L</small>-(+)-lactic acid or (''S'')-lactic acid and the other, its mirror image, is <small>D</small>-(-)-lactic acid or (''R'')-lactic acid. <small>L</small>-(+)-Lactic acid is the biologically important isomer. | |
| − | + | In animals, <small>L</small>-lactate is constantly produced from [[pyruvate]] via the [[enzyme]] lactate dehydrogenase (LDH) in a process of [[fermentation (biochemistry)|fermentation]] during normal [[metabolism]] and exercise. It does not increase in concentration until the rate of lactate production exceeds the rate of lactate removal, which is governed by a number of factors including monocarboxylate transporters, concentration and isoform of LDH, and oxidative capacity of [[tissue]]s. The concentration of [[blood]] lactate is usually 1-2 mmol/L at rest, but can rise to over 20 mmol/L during intense exertion. | |
| − | In [[medicine]], lactate is one of the main components of | + | Lactic acid fermentation is also performed by ''Lactobacillus'' [[bacteria]]. These convert [[lactose]] and other [[sugar]]s to lactic acid. They are common and usually benign. In humans, they are present in the [[vagina]] and the [[gastrointestinal tract]], where they are [[symbiotic]] and make up a small portion of the gut flora. Many species are prominent in decaying plant material. The production of lactic acid makes its environment acidic which inhibits the growth of some harmful bacteria. Some ''Lactobacillus'' species are used industrially for the production of yogurt, [[cheese]], [[sauerkraut]], [[pickle]]s, [[beer]], [[wine]], [[cider]], [[kimchi]], and other fermented foods, as well as animal feeds, Although considered beneficial, these bacteria also can operate in the [[mouth]]; the [[acid]] they produce is responsible for the [[tooth]] decay known as caries. |
| + | |||
| + | In [[medicine]], lactate is one of the main components of Ringer's lactate or lactated Ringer's solution. This intravenous fluid consists of [[sodium]] and [[potassium]] [[cation]]s, with lactate and [[chloride]] [[anion]]s, in solution with distilled [[water]] in concentration so as to be isotonic compared to [[human]] [[blood]]. It is most commonly used for fluid resuscitation after blood loss due to trauma, [[surgery]] or a burn injury. | ||
== Exercise and lactate == | == Exercise and lactate == | ||
| Line 77: | Line 82: | ||
[[Category:Life sciences]] | [[Category:Life sciences]] | ||
| − | {{credit|118876263}} | + | {{credit|Lactic_acid|118876263|Lactobacillus|120796459}} |
Revision as of 21:25, 8 April 2007
| Lactic acid | |
|---|---|
| |
| Chemical name | 2-hydroxypropanoic acid |
| Chemical formula | C3H6O3 |
| Molecular mass | 90.08 g/mol |
| CAS number | [50-21-5] L: [79-33-4] D: [10326-41-7] D/L: [598-82-3] |
| Melting point | L: 53 °C D: 53 °C D/L: 16.8 °C |
| Boiling point | 122 °C @ 12 mmHg |
| SMILES | CC(O)C(=O)O |
| Disclaimer and references | |
For the production of milk by mammals, see Lactation.
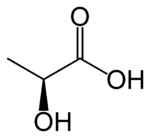
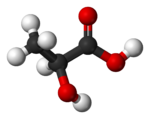
Lactic acid (IUPAC systematic name2-hydroxypropanoic acid), also known as milk acid, is a colorless, water-soluable, liquid organic acid that plays a role in several biochemical processes. It is produced in muscles through anaerobic glucose metabolism, and is the bitter-tasking fermentation product of bacteria interacting with lactose (milk sugar), occuring naturally in sour milk, yourt, and cheese. cream, buttermilk, most common acid constituent of fermented milk products. Also present in various fruits and wines and in soil.. Commercially produced lactic acid is sued for flavoring, imparting a tart flavor, an as preservative, dyeing, and pharmaceuticals. leather tanning, textile dyeing, making plastics, etc.
Lactic acid is a carboxylic acid—an organic (carbon-containing) acid characterized by the presence of a carboxyl group, which has the formula -C(=O)OH, usually written -COOH or -CO2H. Lactic acid has the chemical formula of C3H6O3.
Overview and chemistry
Lactic acid was first isolated in 1780 by a Swedish chemist, Carl Wilhelm Scheele. It has an hydroxyl (OH) group adjacent to the carboxyl group, making it an alpha hydroxy acid (AHA). In solution, it can lose a proton from the acidic group, producing the lactate ion CH3CH(OH)COO−. It is miscible with water or ethanol, and is hygroscopic (ability of a substance to attract water molecules).
Lactic acid is chiral (cannot be superimposed on its mirror image, like the right and left hand of a person) and has two optical isomers. One is known as L-(+)-lactic acid or (S)-lactic acid and the other, its mirror image, is D-(-)-lactic acid or (R)-lactic acid. L-(+)-Lactic acid is the biologically important isomer.
In animals, L-lactate is constantly produced from pyruvate via the enzyme lactate dehydrogenase (LDH) in a process of fermentation during normal metabolism and exercise. It does not increase in concentration until the rate of lactate production exceeds the rate of lactate removal, which is governed by a number of factors including monocarboxylate transporters, concentration and isoform of LDH, and oxidative capacity of tissues. The concentration of blood lactate is usually 1-2 mmol/L at rest, but can rise to over 20 mmol/L during intense exertion.
Lactic acid fermentation is also performed by Lactobacillus bacteria. These convert lactose and other sugars to lactic acid. They are common and usually benign. In humans, they are present in the vagina and the gastrointestinal tract, where they are symbiotic and make up a small portion of the gut flora. Many species are prominent in decaying plant material. The production of lactic acid makes its environment acidic which inhibits the growth of some harmful bacteria. Some Lactobacillus species are used industrially for the production of yogurt, cheese, sauerkraut, pickles, beer, wine, cider, kimchi, and other fermented foods, as well as animal feeds, Although considered beneficial, these bacteria also can operate in the mouth; the acid they produce is responsible for the tooth decay known as caries.
In medicine, lactate is one of the main components of Ringer's lactate or lactated Ringer's solution. This intravenous fluid consists of sodium and potassium cations, with lactate and chloride anions, in solution with distilled water in concentration so as to be isotonic compared to human blood. It is most commonly used for fluid resuscitation after blood loss due to trauma, surgery or a burn injury.
Exercise and lactate
During intense exercise, such as sprinting type activities, when the rate of demand for energy is high, lactate is produced faster than the ability of the tissues to remove it and lactate concentration begins to rise. This is a beneficial process since the regeneration of NAD+ ensures that energy production is maintained and exercise can continue. The increased lactate produced can be removed in a number of ways including: oxidation to pyruvate by well-oxygenated muscle cells which is then directly used to fuel the citric acid cycle and conversion to glucose via the Cori cycle in the liver through the process of gluconeogenesis.
Contrary to popular belief, this increased concentration of lactate does not directly cause acidosis, nor is it responsible for delayed onset muscle soreness.[1] This is because lactate itself is not capable of releasing a proton, and secondly, the acidic form of lactate, lactic acid, cannot be formed under normal circumstances in human tissues. Analysis of the glycolytic pathway in humans indicates that there are not enough hydrogen ions present in the glycolytic intermediates to produce lactic or any other acid.
The acidosis that is associated with increases in lactate concentration during heavy exercise arises from a separate reaction. When ATP is hydrolysed, a hydrogen ion is released. ATP-derived hydrogen ions are primarily responsible for the decrease in pH. During intense exercise, aerobic metabolism cannot produce ATP quickly enough to supply the demands of the muscle. As a result, anaerobic metabolism becomes the dominant energy producing pathway as it can form ATP at high rates. Due to the large amounts of ATP being produced and hydrolysed in a short period of time, the buffering systems of the tissues are overcome, causing pH to fall and creating a state of acidosis. This may be one factor, among many, that contributes to the acute muscular discomfort experienced shortly after intense exercise.
The effect of lactate on acidosis has been the topic of many recent conferences in the field of exercise physiology. Robergs et al. have accurately chased the proton movement that occurs during glycolysis. However, in doing so, they have suggested that [H+] is an independent variable that determines its own concentration. A recent review by Lindinger et al. has been written to rebut the stoichiometric approach used by Robergs et al. In using this stoichiometric process, Robergs et al. have ignored the causitive factors (independent variables) of [H+]. These factors are strong ion difference [SID], PCO2, and weak acid buffers. Lactate is a strong anion, and causes a reduction in [SID] which causes and increase in [H+] to maintain electroneutrality. PCO2 also causes an increase in [H+]. During exercise, intramuscular [lactate] and PCO2 increase, causing an increase in [H+], and thus a decrease in pH. It is unclear as to why Robergs et al. have ignored these independent variables in their review. [SID], PCO2, and [weak acid buffer] have been measured during exercise by Jones et al. The reader interested in acid-base balance during exercise is recommended to consult work by Peter Stewart.
Lactic acid in food
Lactic acid is primarily found in sour milk products, such as: koumiss, leban, yogurt, kefir and some cottage cheeses. The casein in fermented milk is coagulated (curdled) by lactic acid.
Although it can be fermented from lactose (milk sugar), most commercially used lactic acid is derived by using bacteria such as Bacillus acidilacti, Lactobacillus delbueckii or Lactobacillus bulgaricus to ferment carbohydrates from nondairy sources such as cornstarch, potatoes and molasses. Thus, although it is commonly known as "milk acid", products claiming to be vegan do sometimes feature lactic acid as an ingredient.
Lactic acid may also be found in various processed foods, usually either as a pH adjusting ingredient, or as a preservative (either as antioxidant or for control of pathogenic micro-organisms). It may also be used as a fermentation booster in rye and sourdough breads.[2]
Lactic acid as a polymer precursor
Two molecules of lactic acid can be dehydrated to lactide, a cyclic lactone. A variety of catalysts can polymerise lactide to either heterotactic or syndiotactic polylactide, which as biodegradable polyesters with valuable (inter alia) medical properties are currently attracting much attention.
ReferencesISBN links support NWE through referral fees
- ↑ Robergs R, Ghiasvand F, Parker D (2004). Biochemistry of exercise-induced metabolic acidosis.. Am J Physiol Regul Integr Comp Physiol 287 (3): R502-16. PMID 15308499.
- ↑ "Food Applications". Galactic Div. of Finasucre. 2006. http://www.lactic.com/
See also
- Cori cycle
- Alanine cycle
External links
- Lactic acid and running: myths, legends and reality
- Biochemistry of exercise-induced metabolic acidosis.
Template:ChemicalSources
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.