Difference between revisions of "Histidine" - New World Encyclopedia
(New page: {{Claimed}}) |
|||
| Line 1: | Line 1: | ||
{{Claimed}} | {{Claimed}} | ||
| + | {{NatOrganicBox | ||
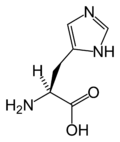
| + | | image=[[Image:L-histidine-skeletal.png|120px|Chemical structure of Histidine]] [[Image:L-histidine-3D-sticks.png|150px|Chemical structure of Histidine]] | ||
| + | | name=2-amino-3-(3H-imidazol-4-yl)propanoic acid | ||
| + | | PubChem = 773 | ||
| + | | CAS = 71-00-1 | ||
| + | | SMILES = N[C@@H](Cc1[nH]cnc1)C(O)=O | ||
| + | | C=6 | H=9 | N=3 | O=2 | ||
| + | | mass=155.16 | ||
| + | }} | ||
| + | [[Image:Succinate Dehygrogenase 1YQ3 Haem group.png|thumb|right|The histidine bound [[heme]] group of [[succinate dehydrogenase]], an [[electron carrier]] in the [[mitochondria]]l [[electron transfer chain]]. The large semi-transparent sphere indicates the location of the [[iron]] [[ion]]. From {{PDB|1YQ3}}.]] | ||
| + | '''Histidine''' (His, H) is one of the 20 most common natural [[amino acid]]s present in [[protein]]s. In the [[Nutrition|nutritional]] sense, in humans, histidine is considered an [[essential amino acid]], but mostly only in children. | ||
| + | |||
| + | ==Chemical properties== | ||
| + | The [[imidazole]] [[side chains]] and the relatively neutral pKa of histidine (ca 6.0) mean that relatively small shifts in cellular [[pH]] will change its charge. For this reason, this amino acid side chain finds its way into considerable use as a co-ordinating [[ligand]] in [[metalloprotein]]s, and also as a [[catalyst|catalytic]] site in certain [[enzyme]]s. The imidazole side chain has two nitrogens with different properties: One is bound to hydrogen and donates its lone pair to the aromatic ring and as such is slighty [[acidic]], whereas the other one donates only one electron pair to the ring so it has a free lone pair and is [[basic (chemistry)|basic]]. These properties are exploited in different ways in proteins. In [[catalytic triad]]s, the basic nitrogen of histidine is used to abstract a proton from [[serine]], [[threonine]] or [[cysteine]] to activate it as a [[nucleophile]]. In a histidine proton shuttle, histidine is used to quickly shuttle protons, it can do this by abstracting a proton with its basic nitrogen to make a positively-charged intermediate and then use another molecule, a buffer, to extract the proton from its acidic nitrogen. In [[carbonic anhydrase]]s, a histidine proton shuttle is utilized to rapidly shuttle protons away from a zinc-bound water molecule to quickly regenerate the active form of the enzyme. | ||
| + | |||
| + | ==Metabolism== | ||
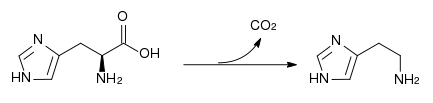
| + | The amino acid is a precursor for [[histamine]] and [[carnosine]] biosynthesis. | ||
| + | |||
| + | [[Image:Histidine decarboxylase.svg|thumb|center|433px|Conversion of [[histidine]] to [[histamine]] by [[histidine decarboxylase]]]] | ||
| + | |||
| + | The enzyme [[histidine ammonia-lyase]] converts histidine into [[ammonia]] and [[urocanic acid]]. A deficiency in this enzyme is present in the rare metabolic disorder [[histidinemia]]. | ||
| + | |||
| + | ==Sources== | ||
| + | Histidine is found in [[fruits]] such as [[bananas]] and [[grapes]], [[meat]] and [[poultry]], and [[milk]] and milk products. It is also found in root [[vegetables]] and all green vegetables, though in lesser quantities. | ||
| + | |||
| + | ==Forms== | ||
| + | There are two [[enantiomers]]: D-histidine and L-histidine. | ||
| + | |||
| + | ==History== | ||
| + | Histidine was first isolated in 1896 by German physician [[Albrecht Kossel]]. | ||
| + | |||
| + | ==Additional images== | ||
| + | <gallery> | ||
| + | Image:Histidine_resonant.png|Histidine | ||
| + | </gallery> | ||
| + | |||
| + | == External links == | ||
| + | *[http://www.chem.qmul.ac.uk/iubmb/enzyme/reaction/AminoAcid/His1.html Histidine biosynthesis (early stages)] | ||
| + | *[http://www.chem.qmul.ac.uk/iubmb/enzyme/reaction/AminoAcid/His2.html Histidine biosynthesis (later stages)] | ||
| + | *[http://www.chem.qmul.ac.uk/iubmb/enzyme/reaction/AminoAcid/His3.html Histidine catabolism] | ||
| + | *[http://www.compchemwiki.org/index.php?title=Histidine Computational Chemistry Wiki] | ||
| + | |||
| + | {{ChemicalSources}} | ||
| + | |||
| + | {{AminoAcids}} | ||
| + | |||
| + | |||
| + | [[Category:Proteinogenic amino acids]] | ||
| + | [[Category:Basic amino acids]] | ||
| + | [[Category:Essential amino acids]] | ||
| + | [[Category:Imidazoles]] | ||
| + | {{credit|138340299}} | ||
| + | [[Category:Life sciences]] | ||
Revision as of 15:16, 15 June 2007
| |
Histidine | |
| Systematic (IUPAC) name | |
| 2-amino-3-(3H-imidazol-4-yl)propanoic acid | |
| Identifiers | |
| CAS number | 71-00-1 |
| PubChem | 773 |
| Chemical data | |
| Formula | C6H9N3O2 |
| Mol. weight | 155.16 |
| SMILES | N[C@@H](Cc1[nH]cnc1)C(O)=O |
| Complete data | |
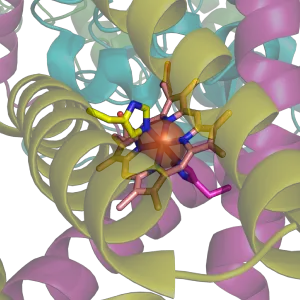
Histidine (His, H) is one of the 20 most common natural amino acids present in proteins. In the nutritional sense, in humans, histidine is considered an essential amino acid, but mostly only in children.
Chemical properties
The imidazole side chains and the relatively neutral pKa of histidine (ca 6.0) mean that relatively small shifts in cellular pH will change its charge. For this reason, this amino acid side chain finds its way into considerable use as a co-ordinating ligand in metalloproteins, and also as a catalytic site in certain enzymes. The imidazole side chain has two nitrogens with different properties: One is bound to hydrogen and donates its lone pair to the aromatic ring and as such is slighty acidic, whereas the other one donates only one electron pair to the ring so it has a free lone pair and is basic. These properties are exploited in different ways in proteins. In catalytic triads, the basic nitrogen of histidine is used to abstract a proton from serine, threonine or cysteine to activate it as a nucleophile. In a histidine proton shuttle, histidine is used to quickly shuttle protons, it can do this by abstracting a proton with its basic nitrogen to make a positively-charged intermediate and then use another molecule, a buffer, to extract the proton from its acidic nitrogen. In carbonic anhydrases, a histidine proton shuttle is utilized to rapidly shuttle protons away from a zinc-bound water molecule to quickly regenerate the active form of the enzyme.
Metabolism
The amino acid is a precursor for histamine and carnosine biosynthesis.
The enzyme histidine ammonia-lyase converts histidine into ammonia and urocanic acid. A deficiency in this enzyme is present in the rare metabolic disorder histidinemia.
Sources
Histidine is found in fruits such as bananas and grapes, meat and poultry, and milk and milk products. It is also found in root vegetables and all green vegetables, though in lesser quantities.
Forms
There are two enantiomers: D-histidine and L-histidine.
History
Histidine was first isolated in 1896 by German physician Albrecht Kossel.
Additional images
External links
- Histidine biosynthesis (early stages)
- Histidine biosynthesis (later stages)
- Histidine catabolism
- Computational Chemistry Wiki
Template:ChemicalSources
| Major families of biochemicals | ||
| Peptides | Amino acids | Nucleic acids | Carbohydrates | Nucleotide sugars | Lipids | Terpenes | Carotenoids | Tetrapyrroles | Enzyme cofactors | Steroids | Flavonoids | Alkaloids | Polyketides | Glycosides | ||
| Analogues of nucleic acids: | The 20 Common Amino Acids | Analogues of nucleic acids: |
| Alanine (dp) | Arginine (dp) | Asparagine (dp) | Aspartic acid (dp) | Cysteine (dp) | Glutamic acid (dp) | Glutamine (dp) | Glycine (dp) | Histidine (dp) | Isoleucine (dp) | Leucine (dp) | Lysine (dp) | Methionine (dp) | Phenylalanine (dp) | Proline (dp) | Serine (dp) | Threonine (dp) | Tryptophan (dp) | Tyrosine (dp) | Valine (dp) | ||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
