Hydrogen peroxide
| Hydrogen peroxide | |
|---|---|
 | |
| General | |
| Systematic name | Dihydrogen dioxide |
| Other names | Hydrogen peroxide hydrogen dioxide dioxidane |
| Molecular formula | H2O2 |
| Molar mass | 34.0147 g·mol·−1. |
| Appearance | Very pale blue color; colorless in solution. |
| CAS number | [7722-84-1] [1] |
| Properties | |
| Density and phase | 1.4 g·cm−3, liquid |
| Solubility in water | Miscible. |
| Melting point | -11 °C (262.15 K) |
| Boiling point | 150.2 °C (423.35 K) |
| Acidity (pKa) | 11.65 |
| Viscosity | 1.245 cP at 20 °C |
| Structure | |
| Molecular shape | bent |
| Dipole moment | 2.26 D |
| Hazards | |
| MSDS | 30% hydrogen peroxide msds 60% hydrogen peroxide msds |
| Main hazards | Oxidant, corrosive. |
| NFPA 704 | |
| Flash point | Non-flammable. |
| R/S statement | R: R5, R8, R20, R22,R35 S: (S1), S2, S17, S26,S28, S36, S37, S39, S45 |
| RTECS number | MX0900000 |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other anions | ? |
| Other cations | Sodium peroxide |
| Related compounds | Water ozone hydrazine |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Hydrogen peroxide (H2O2) is a very pale blue liquid which appears colorless in a dilute solution, slightly more viscous than water. It is a weak acid. It has strong oxidizing properties and is therefore a powerful bleaching agent that has found use as a disinfectant, as an oxidizer, and in rocketry (particularly in high concentrations as high-test peroxide (HTP) as a monopropellant), and in bipropellant systems.
History
Hydrogen peroxide was first isolated in 1818 by Louis Jacques Thénard by reacting barium peroxide with nitric acid. An improved version of this process used hydrochloric acid, followed by sulphuric acid to precipitate the barium sulfate byproduct. Thenard's process was used from the end of the nineteenth century until the middle of the twentieth century.[1] Modern production methods are discussed below.
Manufacture
Hydrogen peroxide is manufactured today almost exclusively by the autoxidation of 2-ethyl-9,10-dihydroxyanthracene to 2-ethylanthraquinone and hydrogen peroxide using oxygen from the air. The anthraquinone derivative is then extracted out and reduced back to the dihydroxy compound using hydrogen gas in the presence of a metal catalyst. The overall equation for the process is deceptively simple:
- H2 + O2 → H2O2
However the economics of the process depend on effective recycling of the quinone and extraction solvents, and of the hydrogenation catalyst.
Formerly inorganic processes were used, employing the electrolysis of an aqueous solution of sulfuric acid or acidic ammonium bisulfate (NH4HSO4), followed by hydrolysis of the peroxydisulfate ((SO4)2)2− which is formed.
Storage
Regulations vary, but low concentrations, such as 2.5% are widely available and legal to buy for medical use.
Hydrogen peroxide should be stored in a container made from a material that doesn't react or catalyse the chemical. Numerous materials and processes are available, some stainless steels, many plastics, glasses and some aluminum alloys are compatible.[2]
As peroxide is a strong oxidizer it should be stored away from fuel sources and sources of catalytic contamination (see decomposition section). Apart from obvious fire risks, peroxide vapor can react with hydrocarbons and alcohols to form contact explosives. Because oxygen is formed during natural decomposition of the peroxide, the resultant increase in pressure can cause a container (such as of glass) to shatter.
Peroxide should be kept cool, as peroxide vapor can detonate above 70 °C.
Deaths have occurred from storage in inadequately marked containers due to its apparent similarity to water.
Physical properties
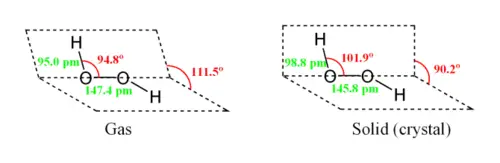
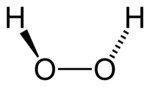
While the anti conformer would minimize steric repulsions, a 90° torsion angle would optimize mixing between the filled p-type orbital of the oxygen (one of the lone pairs) and the LUMO of the vicinal O-H bond.[3] Reflecting a compromise between the two interactions, gaseous and liquid hydrogen peroxide adopts an anticlinal "skewed" shape. This rotational conformation is a compromise between the anti conformer, which would minimize steric repulsion, and between the lone pairs on the oxygen atoms. Despite the fact that the O-O bond is a single bond, the molecule has a remarkably high barrier to complete rotation of 29.45 kJ/mol (compared with 12.5 kJ/mol for the rotational barrier of ethane). The increased barrier is also attributed to lone-pair lone-pair repulsion. The bond angles are affected by hydrogen bonding, which is relevant to the structural difference between gaseous and crystalline forms; indeed a wide range of values is seen in crystals containing molecular H2O2.
Chemical properties
H2O2 is one of the most powerful oxidizers known—stronger than chlorine, chlorine dioxide, and potassium permanganate. And through catalysis, H2O2 can be converted into hydroxyl radicals (.OH) with reactivity second only to fluorine.
| Oxidant | Oxidation potential, V |
|---|---|
| Fluorine | 3.0 |
| Hydroxyl radical | 2.8 |
| Ozone | 2.1 |
| Hydrogen peroxide | 1.8 |
| Potassium permanganate | 1.7 |
| Chlorine dioxide | 1.5 |
| Chlorine | 1.4 |
Hydrogen peroxide can decompose spontaneously into water and oxygen. It usually acts as an oxidizing agent, but there are many reactions where it acts as a reducing agent, releasing oxygen as a by-product.
It also readily forms both inorganic and organic peroxides.
Decomposition
Hydrogen peroxide always decomposes (disproportionates) exothermically into water and oxygen gas spontaneously:
- 2 H2O2 → 2 H2O + O2
This process is very favorable; it has a ΔHo of −98.2 kJ·mol−1 and a ΔGo of −119.2 kJ·mol−1 and a ΔS of 70.5 J·mol−1·K−1. The rate of decomposition is dependent on the temperature and concentration of the peroxide, as well as the pH and the presence of impurities and stabilizers. Hydrogen peroxide is incompatible with many substances that catalyse its decomposition, including most of the transition metals and their compounds. Common catalysts include manganese dioxide, and silver. The same reaction is catalyzed by the enzyme catalase, found in the liver, whose main function in the body is the removal of toxic byproducts of metabolism and the reduction of oxidative stress. The decomposition occurs more rapidly in alkali, so acid is often added as a stabilizer.
The liberation of oxygen and energy in the decomposition has dangerous side effects. Spilling high concentration peroxide on a flammable substance can cause an immediate fire, which is further fueled by the oxygen released by the decomposing hydrogen peroxide.
In the presence of certain catalysts, such as Fe2+ or Ti3+, the decomposition may take a different path, with free radicals such as HO· (hydroxyl) and HOO· being formed. A combination of H2O2 and Fe2+ is known as Fenton's reagent.
A common concentration for hydrogen peroxide is "20 volume," which means that when 1 volume of hydrogen peroxide is decomposed, it produces 20 volumes of oxygen. This is equivalent to about 6% or 1.7M.
The hydrogen peroxide you buy at the drug store is a three percent solution. In such small amounts, it is less stable, decomposing faster, but it is stabilized with acetanilide, a substance that has toxic side effects in significant amounts.
Redox reactions
In aqueous solution, hydrogen peroxide can oxidize or reduce a variety of inorganic ions. When it acts as a reducing agent, oxygen gas is also produced. In acid solution Fe2+ is oxidized to Fe3+,
- [[2 Fe2+]](aq) + H2O2 + 2 H+(aq) → 2 [[Fe3+]](aq) + 2H2O(l)
and sulfite (SO32−) is oxidized to sulfate (SO42−). However, potassium permanganate is reduced to Mn2+ by acidic H2O2. Under alkaline conditions, however, some of these reactions reverse; for example, Mn2+ is oxidized to Mn4+ (as MnO2).
Another example of hydrogen peroxide acting as a reducing agent is the reaction with Sodium hypochlorite, this is a convenient method for preparing oxygen in the laboratory.
NaOCl + H2O2 → O2 + NaCl + H2O
Hydrogen peroxide is frequently used as an oxidizing agent in organic chemistry. For example, methyl phenyl sulfide was oxidized to methyl phenyl sulfoxide in 99% yield in methanol in 18 hours (or 20 minutes using a TiCl3 catalyst):
- Ph-S-CH3 + H2O2 → Ph-S(O)-CH3 + H2O
Alkaline hydrogen peroxide is used for epoxidation of electron-deficient alkenes such as acrylic acids, and also for oxidation of alkylboranes to alcohols, the second step of hydroboration-oxidation.
Formation of peroxide compounds
Hydrogen peroxide is a weak acid, and it can form hydroperoxide or peroxide salts or derivatives of many metals.
For example, on addition to an aqueous solution of chromic acid (CrO3) or acidic solutions of dichromate salts, it will form an unstable blue peroxide CrO(O2)2. In aqueous solution it rapidly decomposes to form oxygen gas and chromium salts.
It can also produce peroxoanions by reaction with anions; for example, reaction with borax leads to sodium perborate, a bleach used in laundry detergents:
- Na2B4O7 + 4 H2O2 + 2 NaOH → 2 Na2B2O4(OH)4 + H2O
H2O2 converts carboxylic acids (RCOOH) into peroxy acids (RCOOOH), which are themselves used as oxidizing agents. Hydrogen peroxide reacts with acetone to form acetone peroxide, and it interacts with ozone to form hydrogen trioxide. Reaction with urea produces carbamide peroxide, used for whitening teeth. An acid-base adduct with triphenylphosphine oxide is a useful "carrier" for H2O2 in some reactions.
Hydrogen peroxide reacts with ozone to form trioxidane.
Alkalinity
Hydrogen peroxide is a much weaker base than water, but it can still form adducts with very strong acids. The superacid HF/SbF5 forms unstable compounds containing the [H3O2]+ ion.
Uses
Industrial applications
About 50 percent of the world's production of hydrogen peroxide in 1994 was used for pulp- and paper-bleaching. Other bleaching applications are becoming more important as hydrogen peroxide is seen as an environmentally benign alternative to chlorine-based bleaches.
Other major industrial applications for hydrogen peroxide include the manufacture of sodium percarbonate and sodium perborate, used as mild bleaches in laundry detergents. It is used in the production of certain organic peroxides (such as dibenzoyl peroxide), which in turn are used in polymerizations and other chemical processes. Hydrogen peroxide is also used in the production of epoxides such as propylene oxide. Its reactions with carboxylic acids produce the corresponding "per-acids." For example, its reaction with acetic acid generates peracetic acid, and its reaction with meta-chlorobenzoic acid produces meta-chloroperoxybenzoic acid (mCPBA). The latter is commonly reacted with alkenes to give the corresponding epoxides.
Domestic uses
Diluted H2O2 (around 30 percent) is used to bleach human hair, hence the phrases peroxide blonde and bottle blonde. It can absorb into skin upon contact and create a local skin capillary embolism that appears as a temporary whitening of the skin. It also whitens skeletons that are to be put on display.
Commercial peroxide, as bought at the drugstore in a 2.5-3% solution, can be used to remove bloodstains from carpets and clothing. If a few tablespoons of peroxide are poured onto the stain, they will bubble up in the area of the blood. After a few minutes the excess liquid can be wiped up with a cloth or paper towel and the stain will be gone. Care should be taken, however, as hydrogen peroxide will bleach or discolor many fabrics.
Hydrogen peroxide, being an oxidizing agent, is used in glow sticks. It reacts with phenyl oxalate ester to form an unstable CO2 dimer which in turn causes an added dye to reach an excited state, the latter relaxing to release photons of light.
Treatment of wastewater
Hydrogen peroxide is a strong oxidizer, effective in controlling sulfide and organic related odors in wastewater collection and treatment systems. It is typically applied to a wastewater system most frequently where there is a retention time of less than five hours and at least 30 minutes prior to the point where the hydrogen sulfide is released. Hydrogen peroxide will oxidize the hydrogen sulfide present and in addition promote bio-oxidation of organic odors. Hydrogen peroxide decomposes to oxygen and water adding dissolved oxygen to the system thereby reducing Biological Oxygen Demand (BOD).
Treatment of plants
Some gardeners and hydroponics implementers have professed the value of hydrogen peroxide in their watering solutions. They claim its spontaneous decomposition releases oxygen that can enhance a plant's root development and also help treat root rot, which is cellular root death due to lack of oxygen.
Use in aquaculture
The Food and Drug Administration (FDA) has classified hydrogen peroxide as a Low Regulatory Priority (LRP) drug for use in controlling fungal growth on fish and fish eggs.[4] In addition, recent laboratory tests performed by fish culturists have shown that common household hydrogen peroxide can be used safely to provide oxygen for small fish.[5] When added to water, hydrogen peroxide decomposes and releases oxygen.
Use as a propellant
H2O2 can be used either as a monopropellant (not mixed with fuel) or as the oxidizer component of a bipropellant rocket. Use as a monopropellant takes advantage of the decomposition of 70–98+% concentration hydrogen peroxide into steam and oxygen. The propellant is pumped into a reaction chamber where a catalyst (usually a silver or platinum screen) triggers decomposition, and the hot (>600 °C) oxygen/steam produced is used directly for thrust. H2O2 monopropellant produces a maximum specific impulse (Isp) of 161 s (1.6 kN·s/kg), which makes it a low-performance monopropellant. Compared to hydrazine, peroxide is less toxic, but it is also much less powerful. The famous Bell Rocket Belt used hydrogen peroxide monopropellant.
As a bipropellant, H2O2 is decomposed to burn a fuel as an oxidizer. Specific impulses as high as 350 s (3.5 kN·s/kg) can be achieved, depending on the fuel. Peroxide used as an oxidizer gives a somewhat lower Isp than liquid oxygen, but is dense, storable, noncryogenic and can be more easily used to drive gas turbines to give high pressures. It also can be used for regenerative cooling of rocket engines. Peroxide was used very successfully as an oxidizer for early World-War-II era German rockets, and for the low-cost British launchers, Black Knight and Black Arrow.
In the 1940s and 1950s, the Walter turbine used hydrogen peroxide for use in submarines while submerged; it was found to be too noisy and maintenance-demanding compared to the conventional diesel-electric power system. Some torpedoes used hydrogen peroxide as oxidizer or propellant, but this use has been discontinued by most navies for safety reasons. Hydrogen peroxide leaks were blamed for the sinkings of HMS Sidon and the Russian submarine Kursk. It was discovered, for example, by the Japanese Navy in torpedo trials, that the concentration of H2O2 in right-angle bends in HTP pipework can often lead to explosions in submarines and torpedoes. Hydrogen peroxide is still used on Soyuz for driving gas turbines to power turbopumps, however. SAAB Underwater Systems is manufacturing the Torpedo 2000. This torpedo is used by the Swedish navy. The torpedo is powered by a piston engine, propelled by HTP as an oxidizer and kerosene as a fuel in a bipropellant system.[6]
While its application as a monopropellant for large engines has waned, small thrusters for attitude control that run on hydrogen peroxide are still in use on some satellites. Such thrusters also provide benefits on spacecraft, making them easier to throttle and resulting in safer loading and handling of fuel before launch (as compared to hydrazine monopropellant). However, hydrazine is a more popular monopropellent in spacecraft because of its higher specific impulse and lower rate of decomposition.
- Concentration for propellant use
Hydrogen peroxide works best as a propellant in extremely high concentrations: roughly over 70 percent. Although any concentration of peroxide will generate some hot gas (oxygen plus some steam), at concentrations above approximately 67 percent, the heat of decomposing hydrogen peroxide becomes large enough to completely vaporize all the liquid at standard temperature. This represents a safety and utilization turning point, since decomposition of any concentration above this amount is capable of transforming the liquid entirely to heated gas (the higher the concentration, the hotter the resulting gas). This very hot steam/oxygen mixture can then be used to generate maximal thrust, power, or work, but it also makes explosive decomposition of the material far more hazardous.
Normal propellant grade concentrations therefore vary from 70 to 98 percent, with common grades of 70, 85, 90, and 98 percent. Many of these grades and variations are described in detail in the United States propellant specification number MIL-P-16005 Revision F, which is currently available. The available suppliers of high concentration propellant grade hydrogen peroxide are generally one of the large commercial companies which make other grades of hydrogen peroxide; including Solvay Interox, FMC, and Degussa. Other companies which have made propellant grade hydrogen peroxide in the recent past include Air Liquide and DuPont. DuPont recently sold its hydrogen peroxide manufacturing business to Degussa.
Propellant-grade hydrogen peroxide is available to qualified buyers. Typically this chemical is only sold to commercial companies or government institutions which have the ability to properly handle and utilize the material. Non-professionals have purchased 70 percent or lower concentration hydrogen peroxide (the remaining 30 percent is water with traces of impurities and stabilizing materials, such as tin salts, phosphates, nitrates, and other chemical additives), and increased its concentration themselves. Amateurs try distillation, but this is extremely dangerous with hydrogen peroxide; peroxide vapor can ignite or detonate depending on specific combinations of temperature and pressure. In general any boiling mass of high concentration hydrogen peroxide at ambient pressure will produce vapor phase hydrogen peroxide which can detonate. This hazard is mitigated, but not entirely eliminated with vacuum distillation. Other approaches for concentrating hydrogen peroxide are sparging and fractional crystallization.
High concentration hydrogen peroxide is readily available in 70, 90, and 98 percent concentrations in sizes of one gallon, 30 gallon, and bulk tanker truck volumes. Propellant grade hydrogen peroxide is being used on current military systems and is in numerous defense and aerospace research and development programs. Many privately funded rocket companies are using hydrogen peroxide, notably Blue Origin, and some amateur groups have expressed interest in manufacturing their own peroxide, for their use and for sale in small quantities to others.
Therapeutic uses
Hydrogen peroxide has been used as an antiseptic and anti-bacterial agent for many years. While its use has decreased in recent years with the popularity of better-smelling and more readily available over-the-counter products, it is still used by many hospitals, doctors and dentists in sterilizing, cleaning and treating everything from floors to root canal procedures.
About three percent H2O2 is used medically for cleaning wounds, removing dead tissue, or as an oral debriding agent. However, most over-the-counter peroxide solutions are not suitable for ingestion.
Recently, alternative medical practitioners have advocated administering doses of hydrogen peroxide intravenously in extremely low (less than one percent) concentrations for hydrogen peroxide therapy — a controversial alternative medical treatment for cancer. However, according to the American Cancer Society, "there is no scientific evidence that hydrogen peroxide is a safe, effective or useful cancer treatment." They advise cancer patients to "remain in the care of qualified doctors who use proven methods of treatment and approved clinical trials of promising new treatments." [7] Another controversial alternative medical treatment is inhaling hydrogen peroxide of a concentration around 1%. Internal use of hydrogen peroxide has a history of causing fatal blood disorders, and its recent use as a therapeutic treatment has been linked to several deaths.[8][9]
Hydrogen peroxide is Generally Recognised As Safe (GRAS) as an antimicrobial agent, an oxidizing agent and more by the US Food and Drug Administration.[10] Hydrogen peroxide can also be used as a toothpaste when mixed with correct quantities of baking soda and salt.[11] Like benzoyl peroxide, hydrogen peroxide is also sometimes used in the treatment of acne.
Hydrogen peroxide is also used as an emetic in veterinary practice.[12]
Hazards
Hydrogen peroxide, either in pure or diluted form, can pose several risks:
- Above roughly 70 percent concentrations, hydrogen peroxide can give off vapor that can detonate above 70 °C (158 °F) at normal atmospheric pressure. The remaining liquid can then undergo what is called a boiling liquid expanding vapor explosion (BLEVE). Distillation of hydrogen peroxide at normal pressures is thus highly dangerous.
- Hydrogen peroxide vapors can form sensitive contact explosives with hydrocarbons such as greases. Hazardous reactions ranging from ignition to explosion have been reported with alcohols, ketones, carboxylic acids (particularly acetic acid), amines and phosphorus. It has been said that "peroxides kill chemists."
- Hydrogen peroxide, if spilled on clothing (or other flammable materials), will preferentially evaporate water until the concentration reaches sufficient strength, then clothing will spontaneously ignite. Leather generally contains metal ions from the tanning process and often catches fire almost immediately.[13]
- Concentrated hydrogen peroxide (at concentrations exceeding 50 percent) is corrosive, and even domestic-strength solutions can cause irritation to the eyes, mucous membranes and skin.[14] Swallowing hydrogen peroxide solutions is particularly dangerous, as decomposition in the stomach releases large quantities of gas (10 times the volume of a 3 percent solution) leading to internal bleeding. Inhaling over 10 percent can cause severe pulmonary irritation.
Hydrogen peroxide is naturally produced as a byproduct of oxygen metabolism, and virtually all organisms possess enzymes known as peroxidases, which apparently harmlessly catalytically decomposes low concentrations of hydrogen peroxide to water and oxygen (see Decomposition above).
In one incident, several people were injured after a hydrogen peroxide spill on board an aircraft because the liquid was mistaken for water.[15]
See also
Notes
- ↑ Craig W. Jones. 1999. Applications of Hydrogen Peroxide and Derivatives. RSC Clean Technology Monographs. (Cambridge, UK: Royal Society of Chemistry.)
- ↑ Material Compatibility with Hydrogen Peroxide. OzoneLab. Retrieved December 7, 2007.
- ↑ Eric V. Anslyn, and Dennis A. Dougherty. 2004. Modern Physical Organic Chemistry. (Sausalito, CA: University Science), 122.
- ↑ Guide to Drug, Vaccine, and Pesticide Use in Aquaculture; Table 2. Midwest Tribal Aquaculture Network.
- ↑ Oxygen for Minnows. Great Lakes Sport Fishing Council. Retrieved December 7, 2007.
- ↑ Richard Scott, Homing Instincts. Retrieved December 7, 2007.
- ↑ CA Cancer J Clin. 1993 Jan-Feb;43(1):47-56. "Questionable methods of cancer management: hydrogen peroxide and other 'hyperoxygenation' therapies." PMID 8422605
- ↑ CBS News: 60 Minutes A Prescription for Death? Did Alternative Treatment Lead To Patient's Death? CBS News (Jan 12, 2005).
- ↑ Hydrogen Peroxide. Snopes.com. Retrieved December 7, 2007.
- ↑ Steven Shepherd, M.P.H. Brushing Up on Gum Disease GPO
- ↑ F.D.A.FDA.gov Retrieved January 1, 2008.
- ↑ Drugs to Control or Stimulate Vomiting Merck Veterinary manual.Retrieved January 1, 2008.
- ↑ Material Tests with HTP Armadilloaerospace. (Video demonstration.) Retrieved December 7, 2007.
- ↑ For example, see an MSDS: Hydrogen Peroxide Solution 3%. J.T. Baker. Retrieved December 7, 2007.
- ↑ Hazardous Materials Incident Brief DCA-99-MZ-001, "Spill of undeclared shipment of hazardous materials in cargo compartment of aircraft." National Transportation Safety Board. October 28, 1998; adopted May 17, 2000. Retrieved December 7, 2007.
ReferencesISBN links support NWE through referral fees
- Anslyn, Eric V., and Dennis A. Dougherty. 2004. Modern Physical Organic Chemistry. Sausalito, CA: University Science. ISBN 1891389319.
- Brown Jr., Theodore L., H. Eugene LeMay, Bruce Edward Bursten, and Julia R. Burdge. 2002. Chemistry: The Central Science, 9th ed. Upper Saddle River, NJ: Prentice Hall. ISBN 0130669970.
- Chang, Raymond. 2006. Chemistry, 9th ed. New York, NY: McGraw-Hill Science/Engineering/Math. ISBN 0073221031.
- Cotton, F. Albert, and Geoffrey Wilkinson. 1980. Advanced Inorganic Chemistry, 4th ed. New York, NY: Wiley. ISBN 0471027758.
- Drabowicz, J., et al. 1994. In The Syntheses of Sulphones, Sulphoxides and Cyclic Sulphides, Eds. G. Capozzi et al. Chichester, UK: John Wiley. ISBN 0471939706.
- Greenwood, N. N., and A. Earnshaw. 1998. Chemistry of the Elements, 2nd ed. Oxford, UK; Burlington, MA: Butterworth-Heinemann, Elsevier Science. ISBN 0750633654. Online version available here. Retrieved August 11, 2007.
- Hess, W. T. 1995. "Hydrogen Peroxide." In Kirk-Othmer Encyclopedia of Chemical Technology, Vol. 13., 4th ed. New York: Wiley, 961-995.
- Jones, Craig W. 1999. Applications of Hydrogen Peroxide and Derivatives. RSC Clean Technology Monographs. Cambridge, UK: Royal Society of Chemistry. ISBN 0854045368
- March, J. 1992. Advanced Organic Chemistry, 4th ed. New York: Wiley, 723.
External links
All links retrieved November 28, 2024.
- NIOSH Pocket Guide to Chemical Hazards
- Oxygenation Therapy: Unproven Treatments for Cancer and AIDS
- Explosion of a lorry carrying hydrogen peroxide closes M25 motorway.
- Hydrogen Peroxide in the Human Body.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.