Multiple sclerosis
Multiple sclerosis (MS) is a chronic, inflammatory, noninfectious disease that affects the central nervous system (CNS).
MS causes gradual destruction of myelin (demyelination) and breakdown of neuronal axons throughout the brain and spinal cord. It produces many symptoms, including changes in tactile sensation, visual problems, muscle weakness, depression, difficulties with coordination and speech, severe fatigue, cognitive impairment, problems with balance, and pain. MS may begin with mild symptoms but almost all patients experience a relentless progression toward total loss of mobility. At present,there is no known cause or cure for MS.
The myelin sheath surrounds and protects the neuronal axons and helps them carry electrical signals. The name multiple sclerosis refers to the multiple scars (or scleroses) on the myelin sheaths. This scarring causes diverse symptoms, depending upon which pathways are interrupted.
The total number of people suffering from MS is not known. Estimates for the United States range from 350,000 up to 500,000. The worldwide incidence is estimated to affect 1 to 2.5 million people (Dangond 2006; Porter 2007).
MS affects primarily white adults, with an age of onset typically between 20 and 40 years, and is more common in women than in men by about 2:1 (Dangond 2006; Calabresi 2004). Several subtypes, or patterns of progression, have been described, and several types of therapy have proven to be helpful.
Although there is no known cause (idiopathic) or cure for multiple sclerosis, there has been a documented, miraculous, total recovery of one sufferer, the nun Rita Klaus, after more than 20 years of MS. All of the "permanent" damage to her central nervous system reverted to normal. None of her physicians could offer any explanation. It is attributed to an unknown "spiritual" or mental mechanism, having come one day after hearing a spiritual voice (Dossey 1999). This would seem to suggest that the medical profession still has a lot to learn about the role of the metaphysical realm in healing diseases.
Signs and symptoms
Among the variety of symptoms caused by MS are a decrease in sensation, muscle weakness, muscle spasms, difficulty in moving, difficulties with coordination and balance, problems in speech or swallowing, visual problems (nystagmus, optic neuritis, or diplopia), fatigue, acute or chronic pain syndromes, bladder and bowel difficulties, cognitive impairment, and emotional disturbances (mainly depression). The main clinical measure of the progression of disability and severity of the symptoms is the Expanded Disability Status Scale or EDSS (Kurtzke 1983).
The initial attacks are often transient and mild (or asymptomatic). They often do not prompt a health care visit and sometimes are only identified in retrospect once the diagnosis has been made based on further attacks. The most common initial symptoms reported are:
- Changes in sensation in the arms, legs ,or face (33 percent)
- Complete or partial vision loss (optic neuritis) (16 percent)
- Weakness (13 percent)
- Double vision (7 percent)
- Unsteadiness when walking (5 percent)
- Balance problems (3 percent)
Many rare initial symptoms have been reported, such as aphasia or psychosis (Navarro et al. 2005; Jongen 2006). Fifteen percent of individuals have multiple symptoms when they first seek medical attention (Paty et al. 1994 ) For some people the initial MS attack is preceded by infection, trauma, or strenuous physical effort.
Diagnosis
Multiple sclerosis is difficult to diagnose in its early stages. In fact, definite diagnosis of MS cannot be made until there is evidence of at least two anatomically separate demyelinating events occurring at least thirty days apart.
Historically, different diagnostic criteria have been used, such as the Schumacher criteria and the Poser criteria. Currently, the McDonald criteria represent the best international efforts to standardize the diagnosis of MS (McDonald et al. 2001).
Clinical data alone may be sufficient for a diagnosis of MS. If an individual has suffered two separate episodes of neurologic symptoms characteristic of MS, and the individual also has consistent abnormalities on physical examination, a diagnosis of MS can be made with no further testing. Since some people with MS seek medical attention after only one attack, other testing may hasten the diagnosis and allow earlier initiation of therapy.
Magnetic resonance imaging (MRI) of the brain and spine is often used to evaluate individuals with suspected MS. MRI shows areas of demyelination as bright lesions on T2-weighted images or FLAIR (fluid attenuated inversion recovery) sequences. Gadolinium contrast is used to demonstrate active plaques on T1-weighted images. Because MRI can reveal lesions that occurred previously but produced no clinical symptoms, it can provide the evidence of chronicity needed for a definite diagnosis of MS.
Testing of cerebrospinal fluid (CSF) can provide evidence of chronic inflammation of the central nervous system. The CSF is tested for oligoclonal bands, which are immunoglobulins found in 85 to 95 percent of people with definite MS (but also found in people with other diseases) (Rudick et al. 1987). Combined with MRI and clinical data, the presence of oligoclonal bands can help make a definite diagnosis of MS.
The brain of a person with MS often responds less to stimulation of the optic nerve and sensory nerves. These brain responses can be examined using visual evoked potentials (VEPs) and somatosensory evoked potentials (SEPs). Decreased activity on either test can reveal demyelination. Along with other data, these exams can help find the widespread nerve involvement required for a definite diagnosis of MS (Gronseth et al. 2000).
Another test that may become important in the future is measurement of antibodies against myelin proteins, such as myelin oligodendrocyte glycoprotein (MOG) and myelin basic protein (MBP). As of 2007, however, there is no established role for these tests in diagnosing MS.
The signs and symptoms of MS can be similar to other medical problems, such as neuromyelitis optica, stroke, brain inflammation, infections such as Lyme disease, tumors, and other autoimmune problems, such as lupus. Lyme disease can produce identical MRI lesions and CSF abnormalities (Garcia-Monco et al. 1990; Hansen et al. 1990; Schluesener et al. 1989; Kohler et al. 1988). Additional testing may be needed to help distinguish MS from these other problems.
Disease course and clinical subtypes
The course of MS is difficult to predict, and the disease may at times either lie dormant or progress steadily.
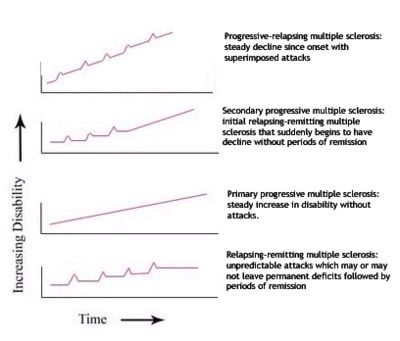
Several subtypes, or patterns of progression, have been described. Subtypes use the past course of the disease in an attempt to predict the future course. Subtypes are important not only for prognosis but also for therapeutic decisions. In 1996, the United States National Multiple Sclerosis Society standardized the following four subtype definitions: (Lublin et al. 1996):
Relapsing-remitting
Relapsing-remitting describes the initial course of 85 percent to 90 percent of individuals with MS. This subtype is characterized by unpredictable attacks (relapses) followed by periods of months to years of relative quiet (remission) with no new signs of disease activity. Deficits suffered during the attacks may either resolve or may be permanent. When deficits always resolve between attacks, this is referred to as "benign" MS.
Secondary progressive
Secondary progressive describes around 80 percent of those with initial relapsing-remitting MS, who then begin to have neurologic decline between their acute attacks without any definite periods of remission. This decline may include new neurologic symptoms, worsening cognitive function, or other deficits. Secondary progressive is the most common type of MS and causes the greatest amount of disability.
Primary progressive
Primary progressive describes the approximately 10 percent of individuals who never have remission after their initial MS symptoms. Decline occurs continuously without clear attacks. The primary progressive subtype tends to affect people who are older at disease onset.
Progressive relapsing
Progressive relapsing describes those individuals who, from the onset of their MS, have a steady neurologic decline but also suffer superimposed attacks; and is the least common of all subtypes
Special cases of the disease with non-standard behavior have also been described, although many researchers believe they are different diseases. These cases are sometimes referred to as borderline forms of multiple sclerosis and are Neuromyelitis optica (NMO), Balo concentric sclerosis, Schilder's diffuse sclerosis, and Marburg multiple sclerosis (Fontaine 2001).
Factors triggering a relapse
MS relapses are often unpredictable. Some attacks, however, are preceded by common triggers. In general, relapses occur more frequently during spring and summer than during autumn and winter. Infections, such as the common cold, influenza, and gastroenteritis, increase the risk for a relapse (Confavreux 2002).
Emotional or physical stress may also trigger an attack (Buljevac et al. 2003; Brown et al. 2006a, 2006b) as well as severe illness of any kind. Statistically, there is not good evidence that either trauma or surgery trigger relapses.
People with MS can participate in sports, but they should probably avoid extremely strenuous exertion, such as marathon running. Heat can transiently increase symptoms, which is known as Uhthoff's phenomenon. This is why some people with MS avoid saunas or even hot showers. However, heat is not an established trigger of relapses (Tataru et al. 2006).
Pregnancy can directly affect the susceptibility for relapse. The last three months of pregnancy offer a natural protection against relapses. However, during the first few months after delivery, the risk for a relapse is increased 20 to 40 percent. Pregnancy does not seem to influence long-term disability. Children born to mothers with MS are not at increased risk for birth defects or other problems (Worthington et al. 1994).
Many potential triggers have been examined and found not to influence relapse rates in MS. Influenza vaccination is safe. There is also no evidence that hepatitis B, varicella, tetanus, or Bacille Calmette-Guerin (BCG-immunization for tuberculosis) increases the risk for relapse (Confavreux et al. 2001).
Pathophysiology
Multiple sclerosis is a disease in which the myelin substance that covers the axons of nerve cells degenerates. Most researchers believe that certain lymphocytes, known as T cells, play a key role in the development of MS.
According to a strictly immunological explanation of MS, the inflammatory processes triggered by the T cells create leaks in the blood-brain barrier. This allows infiltration of monocytes, macrophages, and lymphocytes into the connective tissue (parenchyma) of the CNS (Dangond 2006). These leaks cause a number of other damaging effects such as swelling, activation of macrophages, and more activation of cytokines and other destructive proteins, such as matrix metalloproteinases. A deficiency of uric acid has also been implicated in this process (Rentzos et al. 2006).
In a person with MS, these lymphocytes recognize myelin as foreign and attack it as if it were an invading virus. This triggers inflammatory processes, stimulating other immune cells and soluble factors like cytokines and antibodies.
It is known that a repair process, called remyelination, takes place in early phases of the disease, but the oligodendrocytes that originally formed a myelin sheath cannot completely rebuild it. The newly-formed myelin sheaths are thinner and often not as effective as the original ones. Repeated attacks lead to successively fewer effective remyelinations, until a scar-like plaque is built up around the damaged axons, according to four different damage patterns (Lucchinetti et al. 2000). The CNS should be able to recruit oligodendrocyte stem cells capable of turning into mature myelinating oligodendrocytes, but it is suspected that something inhibits these stem cells in affected areas.
Not only myelin, but also the axons are damaged by the attacks (Pascual et al. 2007). Often, the brain is able to compensate for some of this damage, due to the phenomenon of neuroplasticity. MS symptoms develop as the cumulative result of multiple lesions in the brain and spinal cord. This is why symptoms can vary greatly between different individuals, depending on where the lesions occur.
Causes
Although many risk factors for multiple sclerosis have been identified, no definitive cause has been found. MS may occur as a result of some combination of both environmental and genetic factors. Various theories try to combine the known data into plausible explanations. Although most accept an autoimmune explanation, several theories suggest that MS is an appropriate immune response to an underlying condition. The need for alternative theories is supported by the poor results of present therapies that are based on the the autoimmune theory (Behan et al. 2002; Chaudhuri et al. 2004; Altmann 2005).
Environmental
The most popular hypothesis is that a viral infection or retroviral reactivation primes a susceptible immune system for an abnormal reaction later in life. On a molecular level, this might occur if there is a structural similarity between the infectious virus and some component of the CNS which would lead to eventual confusion in the immune system.
Since MS seems to be more common in people who live farther from the equator, another theory proposes that decreased sunlight exposure (van der Mei et al. 2003 ) and possibly decreased vitamin D production may help cause MS. This theory is bolstered by recent research into the biochemistry of vitamin D, which has shown that it is an important immune system regulator. There is evidence of a link between Vitamin D deficiency and the onset of multiple sclerosis (Munger et al. 2006). Other data shows that sun exposure during childhood reduces the risk of MS (Islam et al. 2007).
Since MS is less common in children with siblings, it has been proposed that less exposure to illness in childhood leads to an immune system that is not primed to fight infection and is thus more likely to attack the body. One explanation for this would be an imbalance between the Th1 type of helper T-cells, which fight infection, and the Th2 type, which are more active in allergy and more likely to attack the body.
Other theories describe MS as an immune response to a chronic infection. The association of MS with the Epstein-Barr virus suggests a potential viral contribution in at least some individuals (Levin et al. 2005). Still others believe that MS may sometimes result from a chronic infection with spirochetal bacteria, a hypothesis supported by research in which cystic forms of spirochetes were isolated from the CSF of all MS patients in a small study (Brorson et al. 2001). When the cysts were cultured, propagating spirochetes emerged. Another bacterium that has been implicated in MS is Chlamydophila pneumoniae. It or its DNA has been found in the CSF of MS patients by several research laboratories, with one study finding that the oligoclonal bands of 14 of the 17 MS patients studied consisted largely of antibodies to Chlamydophila antigens (Yao et al. 2001).
Severe stress may also be a factor—a large study in Denmark found that parents who had lost a child unexpectedly were 50 percent more likely to develop MS than parents who had not (Li et al. 2004). Smoking has also been shown to be an independent risk factor for developing MS (Franklin et al. 2003).
Genetic
MS is not considered a hereditary disease. However, increasing scientific evidence suggests that genetics may play a role in determining a person's susceptibility to MS.
Some populations, such as the Roma, Inuit, and Bantus, rarely, if ever, get MS. The indigenous peoples of the Americas and Asians have very low incidence rates.
In the population at large, the chance of developing MS is less than a tenth of one percent. However, if one person in a family has MS, that person's first-degree relatives—parents, children, and siblings—have a one to three percent chance of getting the disease.
For identical twins, the likelihood that the second twin may develop MS if the first twin does is about 30 percent; for fraternal twins the likelihood is closer to that for non-twin siblings, or about 4 percent. The fact that the rate for identical twins both developing MS is significantly less than 100 percent suggests that the disease is not entirely genetically controlled. Some (but definitely not all) of this effect may be due to shared exposure to something in the environment, or to the fact that some people with MS lesions remain essentially asymptomatic throughout their lives.
Further indications that more than one gene is involved in MS susceptibility come from studies of families in which more than one member has MS. Several research teams found that people with MS inherit certain regions on individual genes more frequently than people without MS. Of particular interest is the human leukocyte antigen (HLA) or major histocompatibility complex region on chromosome 6. HLAs are genetically determined proteins that influence the immune system. However, there are other genes in this region that are not related to the immune system.
The HLA patterns of MS patients tend to be different from those of people without the disease (Dangond 2006). Investigations in northern Europe and America have detected three HLAs that are more prevalent in people with MS than in the general population. Studies of American MS patients have shown that people with MS also tend to exhibit these HLAs in combination—that is, they have more than one of the three HLAs—more frequently than the rest of the population. Furthermore, there is evidence that different combinations of the HLAs may correspond to variations in disease severity and progression.
Studies of families with multiple cases of MS suggest that another area related to MS susceptibility may be located on chromosome 5. Other regions on chromosomes 2, 3, 7, 11, 17, 19, and X have also been identified as possibly containing genes involved in the development of MS.
These studies strengthen the theory that MS is the result of a number of factors rather than a single gene or other agent. Development of MS is likely to be influenced by the interactions of a number of genes, each of which (individually) has only a modest effect. Additional studies are needed to specifically pinpoint which genes are involved, determine their function, and learn how each gene's interactions with other genes and with the environment make an individual susceptible to MS.
Treatments
There is no known definitive cure for multiple sclerosis. However, several types of therapy have proven to be helpful. Different therapies are used for patients with different forms of MS. Treatment is aimed at returning function after an attack, preventing new attacks, and preventing disability.
Various disease-modifying treatments have been approved by the U.S. Food and Drug Administration (FDA) as well as in other countries. More treatments are being studied and undergoing the approval process.
- Interferons: (trade name Betaseron)
- Interferon beta-1a is a medication derived from human cytokines which help regulate the immune system and has been approved by the FDA for relapsing forms of secondary progressive MS.
- Glatiramer acetate: (trade name Copaxone)
- A synthetic medication made of four amino acids that are found in myelin. This drug stimulates T cells in the body's immune system to change from harmful, pro-inflammatory agents to beneficial, anti-inflammatory agents that work to reduce inflammation at lesion sites.
- MIitoxantrone: (trade name Novantrone)
- This medication is effective, but is limited by cardiac toxicity. Novantrone has been approved by the U.S.'s FDA for secondary progressive, progressive-relapsing, and worsening relapsing-remitting MS.
- Natalizumab: (trade name Tysabri)
- This medication is effective and safe alone but in combination with other immunotherapies can lead to progressive multifocal leukoencephalopathy.
Relapsing-remitting symptomatic attacks can be treated. Patients are typically given high doses of intravenous corticosteroids, such as methylprednisolone, to end the attack sooner and leave fewer lasting deficits. Patients' self-reporting indicates that many find benefit from a number of other medicines.
Currently there are no approved treatments for primary progressive multiple sclerosis, though several medications are being studied.
There are many MS patients who have benefited from nontraditional therapies. In a nationwide survey of 3,140 adult Americans with MS (Ko 2003), they stated that the most effective alternative medicine was prayer. The second most effective alternative was exercise. Vitamins and herbs were also used frequently to alleviate symptoms.
Bee sting therapy, in which bees are allowed to sting someone with MS, has many advocates, who claim that bee venom gives more relief from their symptoms than their doctors can provide.
Extracts of the kalawalla plant (Polypodium leucotomos) are also reported by some MS sufferers to reduce their symptoms to a greater degree than conventional drugs.
Prognosis
The probable outcome for a person with MS depends on the subtype of the disease, the individual's sex, race, age, initial symptoms, and the degree of disability the person experiences. The life expectancy of people with MS is now nearly the same as that of unaffected people. This is due mainly to improved methods of limiting disability, such as physical therapy, occupational therapy, and speech therapy, along with more successful treatment of common complications of disability, such as pneumonia and urinary tract infections (Weinshenker 1994). Nevertheless, half of the deaths in people with MS are directly related to the consequences of the disease, while 15 percent are due to suicide (Stern 2005).
Individuals with progressive subtypes of MS, particularly the primary progressive subtype, have a more rapid decline in function. In the primary progressive subtype, supportive equipment (such as a wheelchair or standing frame) is often needed after six to seven years. However, when the initial disease course is the relapsing-remitting subtype, the average time until such equipment is needed is twenty years. This means that many individuals with MS will never need a wheelchair. There is also more cognitive impairment in the progressive forms than in the relapsing-remitting course.
The earlier in life that MS occurs, the slower the disability progresses. Individuals who are older than fifty when diagnosed are more likely to experience a chronic progressive course, with more rapid progression of disability. Those diagnosed before age 35 have the best prognosis. Females generally have a better prognosis than males. Although individuals of African descent tend to develop MS less frequently, they are often older at the time of onset and may have a worse prognosis.
Initial MS symptoms of visual loss or sensory problems, such as numbness or tingling, are markers for a relatively good prognosis, whereas difficulty walking and weakness are markers for a relatively poor prognosis. Better outcomes are also associated with the presence of only a single symptom at onset, the rapid development of initial symptoms and their rapid regression.
The degree of disability varies among individuals with MS. In general, one of three individuals will still be able to work after 15–20 years. Fifteen percent of people diagnosed with MS never have a second relapse, and these people have minimal or no disability after ten years (Pittock et al. 2004). Two-thirds of people with MS with low disability after five years will not get much worse during the next ten years. It should be noted that most of these outcomes were observed before the use of medications such as interferon, which can delay disease progression for several years.
Currently there are no clinically established laboratory investigations available that can predict prognosis or response to treatment. However, several promising approaches have been proposed. These include measurement of the two antibodies: (1) Anti-myelin oligodendrocyte glycoprotein and (2) anti-myelin basic protein, and measurement of a ligand called TRAIL (TNF-related apoptosis-inducing ligand) (Berger et al. 2003).
One well documented exception to the standard prognosis is Rita Klaus, a nun, who developed MS at age 20. As the years passed, her symptoms worsened and her doctors told her that she had suffered irreversible nerve damage and would be confined to a wheelchair. Then one day at the age of 44 she heard a "spiritual" voice inviting her to become healed. The next day she experienced itching and heat in her legs and she was able to wiggle her toes. She removed her leg braces and climbed 13 stairs. This miraculous cure was attested to by several of her doctors. One of her doctors said it was impossible to recover from MS. This was an unusual case in that all the "permanent" damage to her CNS had reverted to normal. None of her physicians could offer any explanation (Dossey 1999).
Many people on the internet claim that they have overcome all or most of the symptoms of MS by means of special exercises or diet. But their credibility is weakened when they offer to sell their method.
Epidemiology
In northern Europe, continental North America, and Australasia, about one of every 1000 citizens suffers from multiple sclerosis, whereas in the Arabian peninsula, Asia, and continental South America, the frequency is much lower. In sub-Saharan Africa, MS is extremely rare. With important exceptions, there is a north-to-south gradient in the northern hemisphere and a south-to-north gradient in the southern hemisphere, with MS being much less common in people living near the equator (Kurtzke 2003).
Climate, diet, geomagnetism, toxins, sunlight exposure, genetic factors, and infectious diseases have all been discussed as possible reasons for these regional differences. Environmental factors during childhood may play an important role in the development of MS later in life. This idea is based on several studies of migrants showing that if migration occurs before the age of fifteen, the migrant acquires the new region's susceptibility to MS. If migration takes place after age fifteen, the migrant keeps the susceptibility of his home country (Marrie 2004).
MS occurs mainly in Caucasians. It is twenty-fold lower in the Inuit people of Canada than in other Canadians living in the same region. It is also rare in the Native American tribes of North America, Australian Aborigines, and the Māori of New Zealand. Scotland appears to have the highest rate of MS in the world (Rothwell et al. 1998). The reasons for this are unknown. These few examples point out that either genetic background or lifestyle or cultural factors may play an role in the development of MS.
As observed in many autoimmune disorders, MS is more common in females than males; the mean sex ratio is about two females for every male. In children (who rarely develop MS), the sex ratio may reach three females for each male. In people over age fifty, MS affects males and females equally. Onset of symptoms usually occurs between fifteen to forty years of age, rarely before age fifteen or after age sixty.
As previously discussed, there is a genetic component to MS. On average, one of every 25 siblings of individuals with MS will also develop MS. Almost half of the identical twins of MS-affected individuals will develop MS, but only one of twenty fraternal twins. If one parent is affected by MS, each child has a risk of only about one in forty of developing MS later in life (Sadovnick et al. 1996).
Finally, it is important to remark that advances in the study of related diseases have shown that some cases formerly considered MS are not MS at all. All the studies before 2004 can be affected by an inability to distinguish MS and Devic's disease (NMO) before this date. The error can be considerable in some areas, and is considered to be 30 percent in Japan (Weinshenker 2005).
History
In 1868, the neurologist Jean-Martin Charcot recognized multiple sclerosis as a distinct, separate disease. Summarizing previous reports and adding his own observations on clinical and pathological data, Charcot called the disease sclerose en plaques. The three signs of MS now known as Charcot's triad are dysarthria (problems with speech), ataxia (problems with coordination), and tremor. Charcot also observed cognition changes in MS since he described his patients as having a "marked enfeeblement of the memory" and "with conceptions that formed slowly" (Charcot 1868).
Prior to Charcot, Robert Hooper a pathologist and practicing physician, Robert Carswell, a professor of pathology, and Jean Cruveilhier, a professor of pathologic anatomy, had described and illustrated many of the disease's clinical details.
After this, several people, such as Eugène Devic, Jozsef Balo, Paul Ferdinand Schilder, and Otto Marburg found special cases of the disease that some authors consider different diseases and now are called the borderline forms of multiple sclerosis.
There are several historical accounts of people who may have had MS. Saint Lidwina of Schiedam (1380-1433), a Dutch nun, may have been one of the first MS patients. From the age of sixteen until her death at age 53, she suffered intermittent pain, weakness of the legs, and vision loss—symptoms typical of MS. There is an earlier story from Iceland of a young woman called Halla who may have had MS. She was blind and deaf for two weeks, sometime between 1293 and 1323. Her loss was very sudden but after praying to the saints, she recovered them seven days later (Poser 1994).
Augustus Frederick d'Este, an illegitimate grandson of King George III of Great Britain, quite possibly suffered from MS. D'Este left a detailed diary describing his 22 years living with the disease. He began his diary in 1822 and made his last entry in 1846. This diary was not discovered until 1948. His symptoms began at age 28 with a sudden transient visual loss after the funeral of a friend. During the course of his disease he developed weakness of the legs, clumsiness of the hands, numbness, dizziness, bladder disturbances, and erectile dysfunction. In 1844, he began to use a wheelchair. Despite his illness, he kept an optimistic view of life (Firth 1948).
Another early account of a possible MS victim was kept by the British diarist W.N.P. Barbellion, who maintained a detailed log of his diagnosis and struggle with MS. His diary was published in 1919, as The Journal of a Disappointed Man.
ReferencesISBN links support NWE through referral fees
- Altmann, D. 2005. Evaluating the evidence for multiple sclerosis as an autoimmune disease. Arch. Neurol. 62(4): 688-689.
- Behan, P., A. Chaudhuri, and B.O. Roep. 2002. [The pathogenesis of multiple sclerosis revisited. J. R. Coll. Physicians Edinb. 32: 244–265. Retrieved October 19, 2007.
- Berger, T., P. Rubner, F. Schautzer, R. Egg, H. Ulmer, I. Mayringer, E. Dilitz, F. Deisenhammer, and M. Reindl. 2003. Antimyelin antibodies as a predictor of clinically definite multiple sclerosis after a first demyelinating event. New England J. Med. 349(2): 139-145.
- Brorson, O., S. H. Brorson, T. H. Henriksen, P. R. Skogen, and R. Schøyen. 2001. Association between multiple sclerosis and cystic structures in cerebrospinal fluid. Infection 29(6): 315-319.
- Brown, R. F., C. C. Tennant, M. Sharrock, S. Hodgkinson, S. M. Dunn, and J. D. Pollard. 2006a. Relationship between stress and relapse in multiple sclerosis: Part I. Important features. Mult. Scler. 12(4): 453-464.
- Brown, R. F., C. C. Tennant, M. Sharrock, S. Hodgkinson, S. M. Dunn, and J. D. Pollard. 2006b. Relationship between stress and relapse in multiple sclerosis: Part II. Direct and indirect relationships. Mult. Scler. 12(4): 465-475.
- Buljevac, D., W. C. Hop, W. Reedeker, et al.. 2003. Self reported stressful life events and exacerbations in multiple sclerosis: prospective study. B.M.J. 327(7416): 646.
- Calabresi, P. A. 2004. Diagnosis and management of multiple sclerosis. Am Fam Physician 70(10): 1935-1944. Retrieved October 20, 2007.
- Charcot, J. 1868. Histologie de la sclerose en plaques. Gazette des hopitaux, Paris 41: 554–555.
- Chaudhuri, A., and P. Behan. 2004. Multiple sclerosis is not an autoimmune disease. Arch. Neurol. 61(10): 1610–1612.
- Confavreux, C . 2002. Infections and the risk of relapse in multiple sclerosis. Brain 125(Pt. 5): 933-934.
- Confavreux, C., S. Suissa, P. Saddier, V. Bourdès, and S. Vukusic. 2001. Vaccinations and the risk of relapse in multiple sclerosis. Vaccines in Multiple Sclerosis Study Group. New England J. Med. 344(5): 319-326.
- Dangond, F. 2006. Multiple sclerosis. eMedicine Neurology. Retrieved October 20, 2007.
- Dossey, L. 1999. Reinventing Medicine: Beyond Mind-Body to a New Era of Healing. New York: HarperCollins Publishers Inc. ISBN 0062516221.
- Firth, D. 1948. The Case of August D`Esté. Cambridge: Cambridge University Press.
- Fontaine, B. 2001. [1] Borderline forms of MS]. Rev . Neurol. (Paris) 157(8-9 Pt. 2): 929-934. Retrieved October 20, 2007.
- Franklin, G. M. and L. Nelson. 2003. Environmental risk factors in multiple sclerosis: causes, triggers, and patient autonomy. Neurology 61(8): 1032-1034.
- Garcia-Monco, J.C., J. Miro Jornet, B. Fernandez Villar, J.L. Benach, A. Guerrero Espejo, and J. A. Berciano. 1990. Multiple sclerosis or Lyme disease? a diagnosis problem of exclusion. Med. Clin. (Barc) 94(18):685-688.
- Gronseth, G. S. and E. J. Ashman. 2000. Practice parameter: the usefulness of evoked potentials in identifying clinically silent lesions in patients with suspected multiple sclerosis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 54(9): 1720–1725.
- Hansen, K., M. Cruz, and H. Link. 1990. Oligoclonal Borrelia burgdorferi-specific IgG antibodies in cerebrospinal fluid in Lyme neuroborreliosis. J. Infect. Dis. 161(6): 1194-1202.
- Islam, T., W. J. Gauderman, W. Cozen, and T. M. Mack. 2007. Childhood sun exposure influences risk of multiple sclerosis in monozygotic twins. Neurology 69: 381-388. Retrieved October 20, 2007.
- Jongen, P. 2006. Psychiatric onset of multiple sclerosis. J. Neurol. Sci. 245(1-2): 59–62.
- Ko, D. 2003. Prayer appears to benefit individuals with multiple sclerosis—Letter to the Editor. Townsend letter for Doctors and Patients. p. 123-125.
- Kohler, J., U. Kern, J. Kasper, B. Rhese-Kupper, and U. Thoden. 1988. Chronic central nervous system involvement in Lyme borreliosis. Neurology 38(6): 863-867.
- Kurtzke, J. F. 1983. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 33(11): 1444-1452.
- Kurtzke, J. F. 2003. Epidemiology and multiple sclerosis. a personal review. Cuadernos de Esclerosis Multiple 16.
- Levin, L.I., K.L. Munger, M.V. Rubertone, C.A. Peck, E.T. Lennette, D. Spiegelman, and A. Ascherio. 2005. Temporal relationship between elevation of epstein-barr virus antibody titers and initial onset of neurological symptoms in multiple sclerosis. JAMA 293(20): 2496-2500.
- Li, J., C. Johansen, H. Bronnum-Hansen, E. Stenager, N. Koch-Henriksen, and J. Olsen. 2004. The risk of multiple sclerosis in bereaved parents: A nationwide cohort study in Denmark. Neurology 62(5): 726-729.
- Lublin, F. D. and S. C. Reingold. 1996. Defining the clinical course of multiple sclerosis: Results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology 46 (4): 907-911.
- Lucchinetti, C., W. Bruck, J. Parisi, B. Scherhauer, M. Rodriguez, and H. Lassmann. 2000. Heterogeneity of multiple sclerosis lesions: Implications for the pathogenesis of demyelination. Ann . Neurol. 47(6): 707-17.
- Marrie, R. A. 2004. Environmental risk factors in multiple sclerosis aetiology. Lancet Neurol. 3(12): 709-718. Review.
- McDonald, W. I., A. Compston, G. Edan, D. Goodkin, H. P. Hartung, F. D.Lublin, H.F. McFarland, D.W. Paty, C.H. Polman, S. C. Reingold, M. Sandberg-Wollheim, W. Sibley, A. Thompson, S. van den Noort, B. Y. Weinshenker, and J. S. Wolinsky. 2001. Recommended diagnostic criteria for multiple sclerosis: Guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann. Neurol. 50(1): 121-127.
- Munger, K. L., L. I. Levin, B. W. Hollis, N. S. Howard, and A. Ascherio. 2006. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 296(23): 2832-2838.
- Navarro, S., B. Mondéjar-Marín, A. Pedrosa-Guerrero, I. Pérez-Molina, J. Garrido-Robres, and A. Alvarez-Tejerina. 2005. Aphasia and parietal syndrome as the presenting symptoms of a demyelinating disease with pseudotumoral lesions. Rev. Neurol. 41(10): 601-603.
- Pascual, A. M., M. C. Martínez-Bisbal, I. Boscá, C. Valero, F. Coret, B. Martínez-Granados, L. Marti-Bonmati, A. Mir, B. Celda, and B. Casanova. 2007. Axonal loss is progressive and partly dissociated from lesion load in early multiple sclerosis. Neurology 69(1): 63-67.
- Paty, D., D. Studney, K. Redekop, and F. Lublin. 1994. MS COSTAR: A computerized patient record adapted for clinical research purposes. Ann Neurol 36(Suppl): S134-135.
- Pittock, S. J., R. L. McClelland, W. T. Mayr, N. W. Jorgensen, B. G. Weinshenker, J. Noseworthy, and M. Rodriguez. 2004. Clinical implications of benign multiple sclerosis: A 20-year population-based follow-up study. Ann Neurol 56(2): 303-306.
- Porter, R. S. (ed.). 2007. Multiple Sclerosis. Merck Manual Home Edition. Retrieved October 20, 2007.
- Poser, C. M. 1994. The dissemination of multiple sclerosis: A Viking saga? A historical essay. Ann. Neurol. 36 (Suppl 2):S231-243.
- Rentzos, M., C. Nikolaou, M. Anagnostouli, A. Rombos, K.Tsakanikas, M. Economou, A. Dimitrakopoulos, M. Karouli, and D. Vassilopoulos. 2006 . Serum uric acid and multiple sclerosis. Clinical Neurology and Neurosurgery 108(6 ): 527-531.
- Rothwell, P. M. and D. Charlton. 1998. High incidence and prevalence of multiple sclerosis in south east Scotland: evidence of a genetic predisposition. J. Neurol. Neurosurg. Psychiatr. 64(6): 730- 735.
- Rudick, R. A. and J. N. Whitaker. 1987. Cerebrospinal fluid tests for multiple sclerosis. In P. Scheinberg, ed., Neurology/neurosurgery update series, Vol. 7, CPEC.
- Sadovnick, A. D., G. C. Ebers, D. A. Dyment, and N. J. Risch. 1996. Evidence for genetic basis of multiple sclerosis. The Canadian Collaborative Study Group. Lancet 347: 1728.
- Schluesener, H. J., R. Martin, and V. Sticht-Groh. 1989. Autoimmunity in Lyme disease: Molecular cloning of antigens recognized by antibodies in the cerebrospinal fluid. Autoimmunity 2(4): 323-330.
- Stern, M. 2005. Aging with multiple sclerosis. Physical medicine and rehabilitation clinics of North America 16(1): 219-234.
- Tataru, N., C. Vidal, P. Decavel, E. Berger, and L. Rumbach. 2006. Limited impact of the summer heat wave in France (2003) on hospital admissions and relapses for multiple sclerosis. Neuroepidemiology 27(1): 28-32.
- van der Mei, I. A., A. L. Ponsonby, T. Dwyer, L. Blizzard, R. Simmons, B. V. Taylor, H. Butzkueven, and T. Kilpatrick. 2003. Past exposure to sun, skin phenotype, and risk of multiple sclerosis: Case-control study. BMJ 327(7410 ): 316.
- Weinshenker, B.G. 1994. Natural history of multiple sclerosis. Ann .Neurol. 36 Suppl:S6–11.
- Weinshenker, B. 2005. Western vs optic-spinal MS: two diseases, one treatment? Neurology 64(4): 594-595.
- Worthington, J., R. Jones, M. Crawford, and A. Forti. 1994. Pregnancy and multiple sclerosis: A 3-year prospective study. J. Neurol. 241(4 : 228-233.
- Yao, S. Y., C. W. Stratton, W. M. Mitchell, and S. Sriram. 2001. CSF oligoclonal bands in MS include antibodies against Chlamydophila antigens. Neurology 56(9): 1168-1176.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.