Hydrazine
| Hydrazine | |
|---|---|
| |
| |
| |
| IUPAC name | Hydrazine |
| Identifiers | |
| CAS number | [] |
| RTECS number | MU7175000 |
| Properties | |
| Molecular formula | N2H4 |
| Molar mass | 32.05 g/mol |
| Appearance | Colourless liquid |
| Density | 1.01 g/mL (liquid) |
| Melting point |
1 °C (274 K) |
| Boiling point |
114 °C (387 K) |
| Solubility in water | miscible |
| Viscosity | 0.9 cP at 25°C[1] |
| Structure | |
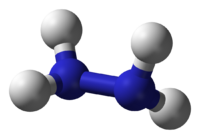
| Molecular shape | pyramidal at N |
| Dipole moment | 1.85 D[1] |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | Toxic, causes burns |
| NFPA 704 |
|
| R-phrases | 45-10-23/24/25-34-43-50/53 |
| S-phrases | 53-45-60-61 |
| Flash point | 37.78°C (closed cup) |
| Related Compounds | |
| Related hydrides | hydrogen peroxide |
| Related compounds | ammonia monomethylhydrazine dimethylhydrazine phenylhydrazine |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
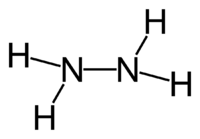
Hydrazine is the chemical compound with formula N2H4. It is widely used in chemical synthesis and is a component in some rocket fuels. With an ammonia-like odor, hydrazine has a liquid range and density similar to water.
Molecular structure and properties
Conceptually, hydrazine arises via coupling a pair of ammonia molecules by removal of one hydrogen per molecule. Each H2N-N subunit is pyramidal. The N-N distance is 1.45 Å, and the molecule adopts a gauche conformation.[2] The rotational barrier is twice that of ethane. These structural properties resemble that of gaseous hydrogen peroxide, which adopts a "skewed" anticlinal conformation, and also experiences a strong rotational barrier.
It has basic properties comparable to ammonia but 15 times weaker.
- N2H4 + H+ → [N2H5]+ K = 8.5 x 10-7
(for ammonia K = 1.78 x 10-5) It can be diprotonated only with difficulty:[3]
- [N2H5]+ + H+ → [N2H6]2+ K = 8.4 x 10-16
Synthesis
Theodor Curtius synthesized free hydrazine for the first time in 1889 via a circuitous route.[4]
Hydrazine is produced in the Olin Raschig process from sodium hypochlorite and ammonia, a process announced in 1907. This method relies on the reaction of chloramine with ammonia.[5]
In the Atofina-PCUK cycle, hydrazine is produced in several steps from acetone, ammonia, and hydrogen peroxide. Acetone and ammonia first react to give the imine followed by oxidation with hydrogen peroxide to the oxaziridine, a three-membered ring containing carbon, oxygen, and nitrogen, followed by ammonolysis to the hydrazone, a process that couples two nitrogen atoms. This hydrazone reacts with one more equivalent of acetone, and the resulting azine is hydrolyzed to give hydrazine, regenerating acetone. Unlike the Raschig process, this process does not produce salt. The PCUK stands for Produits Chimiques Ugine Kuhlmann, a French chemical manufacturer.[6]
Hydrazine can also be produced via the so-called ketazine and peroxide processes.
In 2001, Microbiologist Marc Strous from the University of Nijmegen in the Netherlands discovered that hydrazine is produced from yeast bacteria and the open ocean bacterium anammox (Brocadia anammoxidans). They are the only discovered organisms to naturally produce hydrazine.[7]
Hydrazine derivatives
Many substituted hydrazines are known, and several occur naturally. Some examples:
- gyromitrin and agaritine are phenylhydrazines found in the commercially produced mushroom species Agaricus bisporus. Gyromitrin is metabolized into monomethyl hydrazine.
- iproniazid, hydralazine and phenelzine are hydrazine-containing medications.
- 1,1-dimethylhydrazine and 1,2-dimethylhydrazine are hydrazines where two hydrogen atoms are replaced by methyl groups.
- 2,4-dinitrophenylhydrazine (2,4-DNP) is commonly used to test for ketones and aldehydes in organic chemistry.
- phenylhydrazine, C6H5NHNH2, the first hydrazine to be discovered.
Uses in chemistry
Hydrazines are part of many organic syntheses, often those of practical significance in pharmaceuticals, such as antituberculants, as well as in textile dyes and in photography.
Hydrazone formation
Illustrative of the condensation of hydrazine with a simple carbonyl is its reaction with acetone to give the azine. This azine reacts further with hydrazine to afford the hydrazone:[8]
- 2 (CH3)2CO + N2H4 → 2 H2O + [(CH3)2C=N]2
- [(CH3)2C=N]2 + N2H4 → 2 (CH3)2C=NNH2
The acetone azine is an intermediate in the Atofina-PCUK synthesis. Direct alkylation of hydrazines with alkyl halides in the presence of base affords alkyl-substituted hydrazines, but the reaction is typically inefficient due to poor control on level of substitution (same as in ordinary amines). The reduction of hydrazones to hydrazines present a clean way to produce 1,1-dialkylated hydrazines.
In a related reaction 2-cyanopyridines react with hydrazine to form amide hydrazides which can be converted using 1,2-diketones into triazines.
Wolff-Kishner reduction
Hydrazine is used in the Wolff-Kishner reduction, a reaction that transforms the carbonyl group of a ketone or aldehyde into a methylene (or methyl) group via a hydrazone intermediate. The production of the highly stable dinitrogen from the hydrazine derivative helps to drive the reaction.
Heterocyclic chemistry
Being bifunctional, with two amines, hydrazine is a key building block for the preparation of many heterocyclic compounds via condensation with a range of difunctional electrophiles. With 2,4-pentanedione, it condenses to give the 3,5-dimethylpyrazole.[9] In the Einhorn-Brunner reaction hydrazines react with imides to give triazoles.
Sulfonation
Being a good nucleophile, N2H4 is susceptible to attack by sulfonyl halides and acyl halides.[10] The tosylhydrazine also forms hydrazones upon treatment with carbonyls.
Deprotection of phthalimides
Hydrazine is used to cleave N-alkylated phthalimide derivatives. This scission reaction allows phthalimide anion to be used as amine precursor in the Gabriel synthesis.[11]
Reducing agent
Hydrazine is a convenient reductant because the by-products are typically nitrogen gas and water. Thus, it is used as an antioxidant, an oxygen scavenger, and a corrosion inhibitor in water boilers and heating systems. It is also used to reduce metal salts and oxides to the pure metals in electroless nickel plating and plutonium extraction from nuclear reactor waste.
Hydrazinium salts
Hydrazine is converted to solid salts by treatment with mineral acids. A common salt is hydrazine hydrogen sulfate, [N2H5]HSO4, which probably should be called hydrazinium bisulfate. Hydrazine bisulfate is used as an alternative treatment of cancer-induced cachexia. The salt of hydrazine and hydrazoic acid N5H5 was of scientific interest, because of the high nitrogen content and the explosive properties.
Other industrial uses
Hydrazine is used in many processes including: production of spandex fibers, as a polymerization catalyst; a blowing agent; in fuel cells, solder fluxes; and photographic developers, as a chain extender in urethane polymerizations, and heat stabilizers. In addition, a semiconductor deposition technique using hydrazine has recently been demonstrated, with possible application to the manufacture of thin-film transistors used in liquid crystal displays. Hydrazine in a 70% hydrazine, 30% water solution is used to power the EPU (emergency power unit) on the F-16 fighter plane. The explosive Astrolite is made by combining hydrazine with ammonium nitrate.
Rocket fuel
Hydrazine was first used as a rocket fuel during World War II for the Messerschmitt Me 163B (the first rocket-powered fighter plane), under the name B-Stoff (hydrazine hydrate) and in a mixture with methanol (M-Stoff) and hydrogen peroxide called C-Stoff.
Hydrazine is also used as a low-power monopropellant for the maneuvering thrusters of spacecraft, and the Space Shuttle's Auxiliary Power Units. In addition, monopropellant hydrazine-fueled rocket engines are often used in terminal descent of spacecraft. A collection of such engines were used in both Viking landers as well as the Phoenix lander launched in August 2007.
In all hydrazine monopropellant engines the hydrazine is passed by a catalyst such as iridium metal supported by high-surface-area alumina (aluminium oxide) or carbon nanofibers,[12] or more recently molybdenum nitride on alumina,[13] which causes it to decompose into ammonia, nitrogen gas, and hydrogen gas according to the following reactions:
- 3 N2H4 → 4 NH3 + N2
- N2H4 → N2 + 2 H2
- 4 NH3 + N2H4 → 3 N2 + 8 H2
These reactions are extremely exothermic (the catalyst chamber can reach 800 °C in a matter of milliseconds[12]), and they produce large volumes of hot gas from a small volume of liquid hydrazine,[13] making it an efficient thruster propellant.
Other variants of Hydrazine that are used as rocket fuel are monomethylhydrazine, CH3NHNH2 (also known as MMH) and unsymmetrical dimethylhydrazine, (CH3)2NNH2 (also known as UDMH). These are used as two component rocket fuel, often together with dinitrogen tetroxide, N2O4.
Safety
Hydrazine is highly toxic and dangerously unstable, especially in the anhydrous form. Symptoms of acute exposure to high levels of hydrazine may include irritation of the eyes, nose, and throat, dizziness, headache, nausea, pulmonary edema, seizures, and coma in humans. Acute exposure can also damage the liver, kidneys, and central nervous system in humans. The liquid is corrosive and may produce dermatitis from skin contact in humans and animals. Effects to the lungs, liver, spleen, and thyroid have been reported in animals chronically exposed to hydrazine via inhalation. Increased incidences of lung, nasal cavity, and liver tumors have been observed in rodents exposed to hydrazine.
ReferencesISBN links support NWE through referral fees
- ↑ 1.0 1.1 "Chemistry of the Elements", 2nd ed., Greenwood, N. N. and Earnshaw, A., Butterworth-Heinemann, Oxford (1997).
- ↑ Miessler, Gary L. and Tarr, Donald A. Inorganic Chemistry, Third Edition. Pearson Prentice Hall (2004). ISBN 0-13-035471-6.
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ↑ Curtius, J. Prakt. Chem. 1889, 39, 107-39.
- ↑ Adams, R.; Brown, B. K. (1941). "Hydrazine Sulfate". Org. Synth.; Coll. Vol. 1: 309.
- ↑ Riegel, Emil Raymond. "Hydrazine" Riegel's Handbook of Industrial Chemistry p. 192 (1992).
- ↑ Brian Handwerk. "Bacteria Eat Human Sewage, Produce Rocket Fuel", National Geographic, 9 Nov 2005. Retrieved 2007-11-12.
- ↑ Day, A. C.; Whiting, M. C.. "Acetone Hydrazone". Org. Synth.; Coll. Vol. 6: 10.
- ↑ Wiley, R. H.; Hexner, P. E.. "3,5-Dimethylpyrazole". Org. Synth.; Coll. Vol. 4: 351.
- ↑ Friedman, L; Litle, R. L.; Reichle, W. R.. "p-Toluenesulfonyl Hydrazide". Org. Synth.; Coll. Vol. 5: 1055.
- ↑ Weinshenker, N. M.; Shen, C. M.; Wong, J. Y. (1988). "Polymeric carbodiimide". Org. Synth.; Coll. Vol. 6: 951.
- ↑ 12.0 12.1 Vieira, R. and C. Pham-Huu, N. Keller and M. J. Ledoux (2002). New carbon nanofiber/graphite felt composite for use as a catalyst support for hydrazine catalytic decomposition. Chemical Communications (9): 954—955.
- ↑ 13.0 13.1 Chen, Xiaowei and et al. (April 2002). Catalytic Decomposition of Hydrazine over Supported Molybdenum Nitride Catalysts in a Monopropellant Thruster. Catalysis Letters 79: 21–25.
See also
- List of Stoffs
External links
- The Late Show with Rob! Tonight’s Special Guest: Hydrazine (PDF) — Robert Matunas
cs:Hydrazin de:Hydrazin el:Υδραζίνη es:Hidracina fa:هیدرازین fr:Hydrazine ko:하이드라진 it:Idrazina he:הידרזין lv:Hidrazīns lb:Hydrazin nl:Hydrazine ja:ヒドラジン pl:Hydrazyna pt:Hidrazina ru:Гидразин fi:Hydratsiini sv:Hydrazin uk:Гідразин zh:肼
