Difference between revisions of "Phosgene" - New World Encyclopedia
(imported latest version of article from Wikipedia) |
|||
| Line 1: | Line 1: | ||
| + | {{Claimed}} | ||
{| class="toccolours" border="1" style="float: right; clear: right; margin: 0 0 1em 1em; border-collapse: collapse;" | {| class="toccolours" border="1" style="float: right; clear: right; margin: 0 0 1em 1em; border-collapse: collapse;" | ||
! {{chembox header}} | Phosgene | ! {{chembox header}} | Phosgene | ||
| Line 86: | Line 87: | ||
| [[Carbonic acid]]<br/>[[Urea]]<br/>[[Carbon monoxide]]<br/>[[Chloroformic acid]] | | [[Carbonic acid]]<br/>[[Urea]]<br/>[[Carbon monoxide]]<br/>[[Chloroformic acid]] | ||
|- | |- | ||
| − | | {{chembox header}} | <small>Except where noted otherwise, data are given for<br> materials in their [[standard state|standard state (at 25 °C, 100 kPa)]]<br/>[[wikipedia:Chemical infobox|Infobox disclaimer and references]]</small> | + | | {{chembox header}} | <small>Except where noted otherwise, data are given for<br/> materials in their [[standard state|standard state (at 25 °C, 100 kPa)]]<br/>[[wikipedia:Chemical infobox|Infobox disclaimer and references]]</small> |
|- | |- | ||
|} | |} | ||
| − | '''Phosgene''' is the [[chemical compound]] with the [[chemical formula|formula]] COCl<sub>2</sub>. This highly [[toxic]] gas gained infamy as a [[chemical weapon]] during [[World War I]], but it is also a valuable industrial reagent and building block in [[organic synthesis]]. It is colourless but can appear as a white or yellowish haze when released into air, due to refraction of light. In low concentrations its odor resembles recently cut [[hay]] or green corn ([[maize]]), while at higher concentrations it may be strongly unpleasant. In addition to its industrial production, small amounts occur naturally from the breakdown of chlorinated compounds and the [[combustion]] of [[chlorine]]-containing [[organic chemistry|organic]] | + | '''Phosgene''' is the [[chemical compound]] with the [[chemical formula|formula]] COCl<sub>2</sub>. This highly [[toxic]] gas gained infamy as a [[chemical weapon]] during [[World War I]], but it is also a valuable industrial reagent and building block in [[organic synthesis]]. It is colourless but can appear as a white or yellowish haze when released into air, due to refraction of light. In low concentrations its odor resembles recently cut [[hay]] or green corn ([[maize]]), while at higher concentrations it may be strongly unpleasant. In addition to its industrial production, small amounts occur naturally from the breakdown of chlorinated compounds and the [[combustion]] of [[chlorine]]-containing [[organic chemistry|organic]] compounds. |
==Structure and basic properties== | ==Structure and basic properties== | ||
| Line 101: | Line 102: | ||
==History== | ==History== | ||
| − | Phosgene was synthesized by the chemist [[John Davy (chemist)|John Davy]] (1790-1868) in | + | Phosgene was synthesized by the chemist [[John Davy (chemist)|John Davy]] (1790-1868) in 1812 by exposing a mixture of carbon monoxide and chlorine to [[sunlight]]. He named it in reference to use of light to promote the reaction; from [[Greek language|Greek]], ''phos'' (light) and ''gene'' (born).<ref>{{cite journal |
| title = On a Gaseous Compound of Carbonic Oxide and Chlorine | | title = On a Gaseous Compound of Carbonic Oxide and Chlorine | ||
| author = John Davy | | author = John Davy | ||
| Line 111: | Line 112: | ||
{{further|[[Use of poison gas in World War I]]}} | {{further|[[Use of poison gas in World War I]]}} | ||
| − | Phosgene was stockpiled as part of U.S. military arsenals until well after [[World War II]] in the form of aerial bombs and mortar rounds.<ref name=FM 3-8>FM 3-8 Chemical Reference handbook; US Army; 1967</ref>, the United States began disposing of its stockpiles in | + | Phosgene was stockpiled as part of U.S. military arsenals until well after [[World War II]] in the form of aerial bombs and mortar rounds.<ref name=FM 3-8>FM 3-8 Chemical Reference handbook; US Army; 1967</ref>, the United States began disposing of its stockpiles in 1969. Even before then, the importance of phosgene as a weapon had declined as the more lethal [[nerve agent]]s entered stockpiles. |
==Production== | ==Production== | ||
| Line 141: | Line 142: | ||
==Safety== | ==Safety== | ||
| + | |||
See [[Material_safety_data_sheet|MSDS]]. Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.<ref>{{cite journal | See [[Material_safety_data_sheet|MSDS]]. Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.<ref>{{cite journal | ||
| title = Phosgene exposure: mechanisms of injury and treatment strategies | | title = Phosgene exposure: mechanisms of injury and treatment strategies | ||
| Line 152: | Line 154: | ||
}}</ref> | }}</ref> | ||
Like many reactive chlorides, phosgene combines with water in the tissues of the respiratory tract to form [[hydrochloric acid]]. Phosgene is stable when stored in dry steel containers.<ref name=FM 3-8">'''FM 3-8 Chemical Reference handbook; US Army; 1967</ref>. Phosgene is a member of a class of organic chemicals known as [[alkylating agent]]s.{{Fact|date=April 2007}} These agents can react with both DNA and with enzymes ([[polymerase]]s) that are responsible for replication of DNA in cells. | Like many reactive chlorides, phosgene combines with water in the tissues of the respiratory tract to form [[hydrochloric acid]]. Phosgene is stable when stored in dry steel containers.<ref name=FM 3-8">'''FM 3-8 Chemical Reference handbook; US Army; 1967</ref>. Phosgene is a member of a class of organic chemicals known as [[alkylating agent]]s.{{Fact|date=April 2007}} These agents can react with both DNA and with enzymes ([[polymerase]]s) that are responsible for replication of DNA in cells. | ||
| + | |||
| + | == Notes == | ||
| + | <references/> | ||
==References== | ==References== | ||
| − | < | + | <<Need 3 refs>> |
==External links== | ==External links== | ||
| + | |||
*[http://web.lemoyne.edu/~giunta/jdavy.html Davy's account of his discovery of phosgene] | *[http://web.lemoyne.edu/~giunta/jdavy.html Davy's account of his discovery of phosgene] | ||
*[http://www.ilo.org/public/english/protection/safework/cis/products/icsc/dtasht/_icsc00/icsc0007.htm International Chemical Safety Card 0007] | *[http://www.ilo.org/public/english/protection/safework/cis/products/icsc/dtasht/_icsc00/icsc0007.htm International Chemical Safety Card 0007] | ||
| Line 171: | Line 177: | ||
{{chemical warfare}} | {{chemical warfare}} | ||
| − | + | [[Category:Physical sciences]] | |
| + | [[Category:Military technology]] | ||
[[Category:Chemical weapons]] | [[Category:Chemical weapons]] | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | {{credit|140548673}} | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
Revision as of 04:23, 2 July 2007
| Phosgene | |
|---|---|
| |
| General | |
| Systematic name | Carbonyl chloride |
| Other names | Phosgene CG Carbonic acid dichloride Carbon dichloride oxide Carbon oxychloride Carbonyl dichloride Chloroformyl chloride Dichloroformaldehyde |
| Molecular formula | CCl2O |
| SMILES | O=C(Cl)Cl |
| Molar mass | 98.9 g mol-1 |
| Appearance | colorless gas |
| CAS number | [75-44-5] |
| Properties | |
| Density and phase | 4.248 g dm-3, gas (15 °C) |
| Solubility in water | hydrolysis |
| Other solvents | chlorocarbons |
| Melting point | −118 °C (155 K) |
| Boiling point | 8 °C (281 K) |
| Structure | |
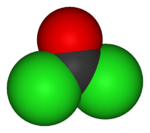
| Molecular shape | Planar |
| Dipole moment | 1.17 D |
| Hazards | |
| MSDS | http://www.vngas.com/pdf/g67.pdf |
| EU classification | Very toxic (T+) |
| NFPA 704 | |
| R-phrases | R26, R34 |
| S-phrases | S1/2, S9, S26, S36/37/39, S45 |
| Flash point | non-flammable |
| RTECS number | SY5600000 |
| Related compounds | |
| Other anions | Carbonyl fluoride |
| Other cations | Nitrosyl chloride |
| Related compounds | Carbonic acid Urea Carbon monoxide Chloroformic acid |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
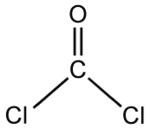
Phosgene is the chemical compound with the formula COCl2. This highly toxic gas gained infamy as a chemical weapon during World War I, but it is also a valuable industrial reagent and building block in organic synthesis. It is colourless but can appear as a white or yellowish haze when released into air, due to refraction of light. In low concentrations its odor resembles recently cut hay or green corn (maize), while at higher concentrations it may be strongly unpleasant. In addition to its industrial production, small amounts occur naturally from the breakdown of chlorinated compounds and the combustion of chlorine-containing organic compounds.
Structure and basic properties
Phosgene is a planar molecule as predicted by VSEPR theory. The C=O distance is 1.18 Å, the C---CL distance is 1.74 Å and the Cl---C---Cl angle is 111.8°.[1]
Phosgene is the simplest and one of the most electrophilic acid chlorides. This high electrophilicity is manifested in the tendency of phosgene to react with water, that is, hydrolyze. This hydrolysis reaction releases hydrogen chloride and carbon dioxide:
- COCl2 + H2O → CO2 + 2 HCl
The toxicity of phosgene is mainly due to the HCl that is released in this hydrolysis reaction.
History
Phosgene was synthesized by the chemist John Davy (1790-1868) in 1812 by exposing a mixture of carbon monoxide and chlorine to sunlight. He named it in reference to use of light to promote the reaction; from Greek, phos (light) and gene (born).[2] It gradually became important in the chemical industry as the 19th century progressed, particularly in dye manufacturing.
- Further information: Use of poison gas in World War I
Phosgene was stockpiled as part of U.S. military arsenals until well after World War II in the form of aerial bombs and mortar rounds.Cite error: Invalid <ref> tag; invalid names, e.g. too many, the United States began disposing of its stockpiles in 1969. Even before then, the importance of phosgene as a weapon had declined as the more lethal nerve agents entered stockpiles.
Production
Around 2 million tons are produced annually[3] for use in the synthesis of fine chemicals and polymers. Industrially, phosgene is produced by passing purified carbon monoxide and chlorine gas through a bed of highly porous carbon, which acts as a catalyst. The chemical equation for this reaction follows:
- CO + Cl2 → COCl2
The reaction is exothermic, therefore the reactor must be cooled to carry away the heat it produces. Typically, the reaction is conducted between 50 and 150 °C. Above 200 °C, phosgene decomposes back into carbon monoxide and chlorine.
Upon ultraviolet radiation in the presence of oxygen, chloroform slowly converts into phosgene via a radical reaction. To suppress this photodegradation, chloroform is often stored in brown-tinted glass containers.
Because of safety issues, phosgene is almost always produced and consumed within the same plant. It is listed on schedule 3 of the Chemical Weapons Convention: all production sites manufacturing more than 30 tonnes per year must be declared to the OPCW.[4] Although much less dangerous than nerve agents, phosgene is still regarded as a viable chemical warfare agent.
Uses
Phosgene is used chiefly in the production of polymers including polyurethanes, polycarbonates, and polyureas. It is also valuable in the preparation of fine chemicals.[5] In the laboratory for small-scale reactions, gaseous phosgene has increasingly been supplanted by more easily handled reagents that effect comparable transformations: diphosgene (chloroformic acid ester), which is a liquid at room temperature, or triphosgene, a crystalline substance. Following are three of many useful reactions involving phosgene.
Synthesis of carbonates
Diols react with phosgene to give either linear or cyclic carbonates (R = H, alkyl, aryl):
- HOCR2-X-CR2OH + COCl2 → 1/n [OCR2-X-CR2OC(O)-]n + 2 HCl
Polycarbonates are an important class of engineering thermoplastic, found for example in lenses in eye glasses.
Synthesis of isocyanates
The synthesis of isocyanates from amines illustrates the electrophilic character of this reagent and its use in introducing the equivalent of "CO2+" (R = alkyl, aryl):
- RNH2 + COCl2 → RN=C=O + 2 HCl
Such reactions are conducted in the presence of a base such as pyridine that absorbs the hydrogen chloride.
Synthesis of acid chlorides and esters
It is also used to produce acid chlorides:
- RCO2H + COCl2 → RC(O)Cl + HCl + CO2
Such acid chlorides react with amines and alcohols to give respectively amides and esters, which are common intermediates in the dye, pesticide, and pharmaceutical industries. Despite being an efficient method of synthesizing acyl chloride from carboxylic acids, laboratory safety issues led to the use of the less toxic thionyl chloride.
Safety
See MSDS. Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.[6] Like many reactive chlorides, phosgene combines with water in the tissues of the respiratory tract to form hydrochloric acid. Phosgene is stable when stored in dry steel containers.[7]. Phosgene is a member of a class of organic chemicals known as alkylating agents.[citation needed] These agents can react with both DNA and with enzymes (polymerases) that are responsible for replication of DNA in cells.
Notes
- ↑ Nakata, M.; Kohata, K.; Fukuyama, T.; Kuchitsu, K. “Molecular Structure of Phosgene as Studied by Gas Electron Diffraction and Microwave Spectroscopy. The rz Structure and Isotope Effect ” Journal of Molecular Spectroscopy 1980, Volume 83, Pages 105-117. Digital object identifier (DOI): 10.1016/0022-2852(80)90314-8
- ↑ John Davy (1812). On a Gaseous Compound of Carbonic Oxide and Chlorine. Philosophical Transactions of the Royal Society of London 102: 144-151.
- ↑ http://cbwinfo.com/Chemical/Pulmonary/CG.shtml
- ↑ http://www.opcw.org/html/db/cwc/eng/cwc_annex_verification_part_VIII.html
- ↑ Hamley, P. "Phosgene" Encyclopedia of Reagents for Organic Synthesis, 2001 John Wiley, New York. DOI: 10.1002/047084289X.rp149.
- ↑ Borak J., Diller W. F. (2001). Phosgene exposure: mechanisms of injury and treatment strategies. Journal of Occupational and Environmental Medicine 43 (2): 110-9. PMID 11227628.
- ↑ FM 3-8 Chemical Reference handbook; US Army; 1967
ReferencesISBN links support NWE through referral fees
<<Need 3 refs>>
External links
- Davy's account of his discovery of phosgene
- International Chemical Safety Card 0007
- NIOSH Pocket Guide to Chemical Hazards
- U.S. CDC Emergency Preparedness & Response
- U.S. EPA Acute Exposure Guideline Levels
- Regime For Schedule 3 Chemicals And Facilities Related To Such Chemicals, OPCW website
- CBWInfo website
- History of chemical warfare during World War I
- Use of Phosgene in WWII and in modern-day warfare (Refer to Section 4.C of the article)
Template:ChemicalSources
| Agents of Chemical Warfare | ||
|---|---|---|
| Blood agents: | Cyanogen chloride (CK) – Hydrogen cyanide (AC) | |
| Blister agents: | Lewisite (L) – Sulfur mustard gas (HD, H, HT, HL, HQ) – Nitrogen mustard gas (HN1, HN2, HN3) | |
| Nerve agents: | G-Agents: Tabun (GA) – Sarin (GB) – Soman (GD) – Cyclosarin (GF) | V-Agents: VE – VG – VM – VX | |
| Pulmonary agents: | Chlorine – Chloropicrin (PS) – Phosgene (CG) – Diphosgene (DP) | |
| Incapacitating agents: | Agent 15 (BZ) – KOLOKOL-1 | |
| Riot control agents: | Pepper spray (OC) – CS gas – CN gas (mace) – CR gas | |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
