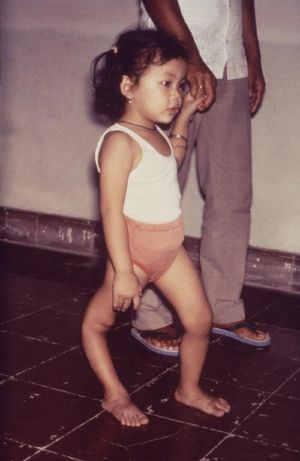Poliomyelitis
| Poliomyelitis | |
| File:Polio lores134.jpg A man with an atrophied right leg due to poliomyelitis | |
| ICD-10 | A80, B91 |
|---|---|
| ICD-9 | 045, 138 |
| DiseasesDB | 10209 |
| MedlinePlus | 001402 |
| eMedicine | ped/1843 pmr/6 |
| MeSH | C02.182.600.700 |
Poliomyelitis, or polio, is a highly contagious viral disease caused by any of three serotypes of poliovirus, with symptoms ranging from a mild, nonparalytic form to one that attacks the central nervous system and can result in temporary or permanent paralysis, and even death. Poliovirus is a form of enterovirus (one that infects the gastrointestinal tract) and this RNA virus enters through the mouth, spreading by direct exposure to an infected person, such as via saliva (oral-oral), or via transmission from foods contaminated by infected fecal matter (fecal-oral). Polio has also been known as infantile paralysis, since it most commonly infects younger children and could cause of lifetime of paralysis.
Although polio has caused paralysis and death for much of human history, major polio epidemics were unknown before the late 19th century. However, polio became one of the most dreaded childhood diseases of the 20th century, with major polio epidemics crippling thousands of people, mostly young children. In the 1950s, vaccines were developed, and with the launching of a global vaccination effort, the number of polio cases worldwide was reduced by 99% since 1985. There is hope that polio can be the second major human illness to be eradicated (after smallpox).
The effort to eradicate polio reflects a great spirit of cooperation, creativity, and sacrifice among individuals and organizations. From the pioneering scientists who developed vaccines, to the health care workers delivering the vaccines, to those volunteering time and money both for prevention and treatment, the effort reflects the best of the human spirit in rising to challenges. Among organizations, the World Health Organization, UNICEF, Rotary International, and the Gates Foundation have stood out in the effort toward the global eradication of the disease. Rotary International, a nonprofit organization, has alone immunized more than one billion children worldwide, at the cost of over 600 million, since it made a commitment in 1985 to the goal of immunizing all the children of the world. Its effort helped to inspire the Global Polio Eradication Initiative that began in 1988, which recently has received a big boost from well over a billion dollars invested by the Gates Foundation.
Overview
| Outcome | Proportion of cases[1] |
|---|---|
| Asymptomatic | 90–95% |
| Minor illness | 4–8% |
| Non-paralytic aseptic meningitis |
1–2% |
| Paralytic poliomyelitis | 0.1–0.5% |
| — Spinal polio | 79% of paralytic cases |
| — Bulbospinal polio | 19% of paralytic cases |
| — Bulbar polio | 2% of paralytic cases |
Poliomyelitis is a human infectious disease caused by caused by any of the three serotypes of poliovirus, and spread from person to person, primarily via the fecal-oral route.[2] It is believed to enter the body through the mouth and is absorbed into the blood and lymphatic system from the gastrointestinal tract. Transmission is often tied to poor sanitary conditions. It can be passed from an infected person to others as a result of contact, such as via saliva from an infected person or should a person engage in poor handwashing after use of the bathroom or eating, or via food contaminated with feces.[3]
Although around 90% of polio infections cause no symptoms at all, affected individuals can exhibit a range of symptoms if the virus enters the blood stream.[4]
Two basic patterns of polio infection are described: a minor illness that does not involve the central nervous system (CNS), sometimes called abortive poliomyelitis, and a major illness involving the CNS, which may be paralytic or non-paralytic.[5][3] In most people with a normal immune system, a poliovirus infection is asymptomatic. Rarely the infection produces minor symptoms; these may include upper respiratory tract infection (sore throat and fever), gastrointestinal disturbances (nausea, vomiting, abdominal pain, constipation or, rarely, diarrhea), and influenza-like illness.[1]
The virus enters the central nervous system in about 1% to 3% of infections. Most patients with CNS involvement develop non-paralytic aseptic meningitis, with symptoms of headache, neck, back, abdominal and extremity pain, fever, vomiting, lethargy and irritability.[6][7] Approximately 1 in 200 to 1 in 1000 cases progress to paralytic disease, in which the muscles become weak, floppy and poorly controlled, and finally completely paralyzed; this condition is known as acute flaccid paralysis and is caused by motor neurons being destroyed by the infection.[8][3]Different types of paralysis may occur, depending on the nerves involved.
Depending on the site of paralysis, paralytic poliomyelitis is classified as spinal, bulbar, or bulbospinal. Spinal polio is the most common form, characterized by asymmetric paralysis that most often involves the legs. Bulbar polio leads to weakness of muscles innervated by cranial nerves. Bulbospinal polio is a combination of bulbar and spinal paralysis.[1]Encephalitis, an infection of the brain tissue itself, can occur in rare cases and is usually restricted to infants. It is characterized by confusion, changes in mental status, headaches, fever, and less commonly seizures and spastic paralysis.[9]
Each of the three known types of poliovirus cause a different form of the disease. It is Type 1 that is the cause of epidemics and many of the cases of paralysis. The term itself derives from the Greek poliós (πολιός), meaning "gray", myelós (µυελός), referring to the "spinal cord", and the suffix -itis, which denotes inflammation.[6]
Poliomyelitis was first recognized as a distinct condition by Jakob Heine in 1840.[10] Its causative agent, poliovirus, was identified in 1908 by Karl Landsteiner.[10]
Polio had existed for thousands of years quietly as an endemic pathogen until the 1880s, when major epidemics began to occur in Europe; soon after, widespread epidemics appeared in the United States.[11] By 1910, much of the world experienced a dramatic increase in polio cases and frequent epidemics became regular events, primarily in cities during the summer months. These epidemics—which left thousands of children and adults paralyzed—provided the impetus for a "Great Race" towards the development of a vaccine. Developed in the 1950s, polio vaccines are credited with reducing the global number of polio cases per year from many hundreds of thousands to around a thousand.[12] Enhanced vaccination efforts led by the World Health Organization, UNICEF, and Rotary International, and now more recently, since 2005, Bill Gates and the Gates Foundation, could result in global eradication of the disease.[13][14][15] Over $9 billion dollars have been invested in the eradication campaign, with over $600 million given by the Rotary Foundation since 1985 and about $1.3 billion via Bill Gates and his foundation since 2005. However, the process of getting rid of the last one percent of the disease has been likened to "trying to squeeze Jell-O to death," with new outbreaks in countries where the virus had been eliminated.[14][15]
Causal agent: Poliovirus
| Poliovirus | ||||||||
|---|---|---|---|---|---|---|---|---|
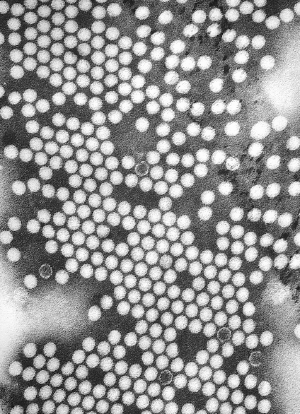
| TEM micrograph of poliovirus virions. TEM micrograph of poliovirus virions.
| ||||||||
| Virus classification | ||||||||
| ||||||||
| Human enterovirus C | ||||||||
|
Human enterovirus C[16] |
Poliovirus, the causative agent of poliomyelitis, is a human enterovirus and member of the family of Picornaviridae.[4] This group of RNA viruses colonize the gastrointestinal tract[2] — specifically the oropharynx and the intestine.
The structure of poliovirus is very simple, composed of a single (+) sense RNA genome enclosed in a protein shell called a capsid.[4] The single-stranded positive-sense RNA genome is about 7500 nucleotides long.[17] The viral particle is about 30 nanometers in diameter with icosahedral symmetry. Because of its short genome and its simple composition—only RNA and a non-enveloped icosahedral protein coat that encapsulates it—poliovirus is widely regarded as the simplest significant virus.[18] In addition to protecting the virus’s genetic material, the capsid proteins enable poliovirus to infect certain types of cells.
Three serotypes of poliovirus have been identified—poliovirus type 1 (PV1), type 2 (PV2), and type 3 (PV3)—each with a slightly different capsid protein.[19] All three are extremely virulent and produce the same disease symptoms.[4] PV1 is the most commonly encountered form, and the one most closely associated with paralysis.[20]
The incubation time (to the first signs and symptoms) ranges from 3 to 35 days with a more common span of 6 to 20 days.[1] PV infects and causes disease in humans alone.[4]
Individuals who are exposed to the virus, either through infection or by immunization with polio vaccine, develop immunity. In immune individuals, IgA antibodies against poliovirus are present in the tonsils and gastrointestinal tract and are able to block virus replication; IgG and IgM antibodies against PV can prevent the spread of the virus to motor neurons of the central nervous system.[21] Infection or vaccination with one serotype of poliovirus does not provide immunity against the other serotypes, and full immunity requires exposure to each serotype.[21]
A rare condition with a similar presentation, non-poliovirus poliomyelitis, may result from infections with non-poliovirus enteroviruses.[22]
Poliovirus was first isolated in 1909 by Karl Landsteiner and Erwin Popper.[10] In 1981, the poliovirus genome was published by two different teams of researchers: by Vincent Racaniello and David Baltimore at MIT[23] and by Naomi Kitamura and others at the State University of New York, Stony Brook.[24] Poliovirus is one of the most well-characterized viruses, and has become a useful model system for understanding the biology of RNA viruses.
Classification
Polioviruses were formerly classified as a distinct species belonging to the genus Enterovirus in the family Picornaviridae. In 2008, the poliovirus species was eliminated from the genus Enterovirus and the three serotypes were assigned to the species Human enterovirus C, in the genus Enterovirus in the family Picornaviridae. The type species of the genus Enterovirus was changed from Poliovirus to Human enterovirus C.[25]
Life cycle
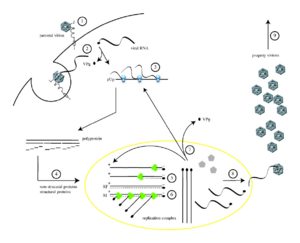
Poliovirus infects human cells by binding to an immunoglobulin-like receptor, CD155 (also known as the poliovirus receptor (PVR)), on the cell surface.[27][28][29] Interaction of poliovirus and CD155 facilitates an irreversible conformational change of the viral particle necessary for viral entry.[30][31] The precise mechanism poliovirus uses to enter the host cell has not been firmly established.[32] Attached to the host cell membrane, entry of the viral nucleic acid was thought to occur one of two ways: via the formation of a pore in the plasma membrane through which the RNA is then “injected” into the host cell cytoplasm, or that the virus is taken up by receptor-mediated endocytosis.[33] Recent experimental evidence supports the latter hypothesis and suggests that poliovirus binds to CD155 and is taken up via endocytosis. Immediately after internalization of the particle, the viral RNA is released.[34] However, any mechanism by which poliovirus enters the cell is very inefficient; as an infection is initiated only about 1% of the time.
Poliovirus is a positive stranded RNA virus. Thus the genome enclosed within the viral particle can be used as messenger RNA and immediately translated by the host cell. On entry, the virus hijacks the cell's translation machinery causing inhibition of cellular protein synthesis in favor of virus–specific protein production. Unlike the host cell's mRNAs, the 5' end of poliovirus RNA is extremely long—over 700 nucleotides—and is highly structured. This region of the viral genome is called internal ribosome entry site (IRES) and it directs translation of the viral RNA. Genetic mutations in this region prevent viral protein production.[35][36][37]
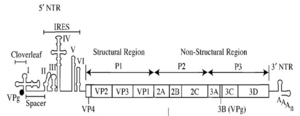
Poliovirus mRNA is translated as one long polypeptide. This polypeptide is then auto-cleaved by internal proteases into approximately 10 individual viral proteins, including:[18]
- 3Dpol, an RNA dependent RNA polymerase whose function is to copy and multiply the viral RNA genome.
- 2Apro and 3Cpro/3CDpro, proteases which cleave the viral polypeptide.
- VPg (3B), a small protein that binds viral RNA and is necessary for synthesis of viral positive and negative strand RNA.
- 2B.C.E., 2B, 2C, 3AB, 3A, 3B proteins which comprise the protein complex needed for virus replication.
- VP0, VP1, VP2, VP3, VP4 proteins of the viral capsid.
The assembly of new virus particles, (i.e., the packaging of progeny genome into a capsid that can survive outside the host cell) is poorly understood.[33] Fully assembled poliovirus leaves the confines of its host cell 4 to 6 hours following initiation of infection in cultured mammalian cells.[21] The mechanism of viral release from the cell is unclear,[17] but each dying cell can release up to 10,000 polio virions.[21]
Serotypes
There are three serotypes of poliovirus, PV1, PV2 , and PV3; each with a slightly different capsid protein. Capsid proteins define cellular receptor specificity and virus antigenicity. PV1 is the most common form encountered in nature; however, all three forms are extremely infectious.[10] Wild polioviruses can be found in approximately 10 countries. PV1 is highly localized to regions in India, Pakistan, Afghanistan, and Egypt, but following outbreaks of poliomeyletis in 2003–2004 it remains widespread in West and Central Africa. Wild poliovirus type 2 has probably been eradicated; it was last detected in October 1999 in Uttar Pradesh, India.[38][39] Wild PV3 is found in parts of only five countries (Nigeria, Niger, Pakistan, India, and Sudan).[21]
Specific strains of each serotype are used to prepare vaccines against polio. Inactive polio vaccine (IPV) is prepared by formalin inactivation of three wild, virulent reference strains, Mahoney or Brunenders (PV1), MEF-1/Lansing (PV2), and Saukett/Leon (PV3). Oral polio vaccine (OPV) contains live attenuated (weakened) strains of the three serotypes of poliovirus. Passaging the virus strains in monkey kidney epithelial cells introduces mutations in the viral IRES, and hinders (or attenuates) the ability of the virus to infect nervous tissue.[21]
Immune system avoidance
Poliovirus uses two key mechanisms to evade the immune system. First, it is capable of surviving the highly acidic conditions of the gastrointestinal tract, allowing the virus to infect the host and spread throughout the body via the lymphatic system.[18] Second, because it can replicate very quickly, the virus overwhelms the host organs before an immune response can be mounted.[23]
Individuals who are exposed to poliovirus, either through infection or by immunization with polio vaccine, develop immunity. In immune individuals, antibodies against poliovirus are present in the tonsils and gastrointestinal tract (specifically IgA antibodies) and are able to block poliovirus replication; IgG and IgM antibodies against poliovirus can prevent the spread of the virus to motor neurons of the central nervous system.[21] Infection with one serotype of poliovirus does not provide immunity against the other serotypes, however second attacks within the same individual are extremely rare.
Cloning and synthesis
In 1981, Racaniello and Baltimore used recombinant DNA technology to generate the first infectious clone of an animal RNA virus, poliovirus. DNA encoding the RNA genome of poliovirus was introduced into cultured mammalian cells and infectious poliovirus was produced.[41] Creation of the infectious clone propelled understanding of poliovirus biology, and has become a standard technology used to study many other viruses.
In 2002, researchers at SUNY Stony Brook succeeded in synthesizing poliovirus from its chemical code, producing the world's first synthetic virus.[42] Scientists first converted poliovirus's published RNA sequence, 7741 bases long, into a DNA sequence, as DNA was easier to synthesize. Short fragments of this DNA sequence were obtained by mail-order, and assembled. The complete viral genome was then assembled by a gene synthesis company. This whole painstaking process took two years. Nineteen markers were incorporated into the synthesized DNA, so that it could be distinguished from natural poliovirus. Enzymes were used to convert the DNA back into RNA, its natural state. Other enzymes were then used to translate the RNA into a polypeptide, producing functional viral particle. The newly minted synthetic virus was injected into PVR transgenic mice, to determine if the synthetic version was able to cause disease. The synthetic virus was able to replicate, infect, and cause paralysis or death in mice. However, the synthetic version was between 1,000 and 10,000 times less lethal than the original virus.[43]
Transmission
Poliomyelitis is highly contagious via the oral-oral (oropharyngeal source) and fecal-oral (intestinal source) routes.[21] In endemic areas, wild polioviruses can infect virtually the entire human population.[44] It is seasonal in temperate climates, with peak transmission occurring in summer and autumn.[21] These seasonal differences are far less pronounced in tropical areas.[44]
The time between first exposure and first symptoms, known as the incubation period, is usually 6 to 20 days, with a maximum range of 3 to 35 days.[23] Virus particles are excreted in the feces for several weeks following initial infection.[23] The disease is transmitted primarily via the fecal-oral route, by ingesting contaminated food or water. It is occasionally transmitted via the oral-oral route,[20] a mode especially visible in areas with good sanitation and hygiene.[21] Polio is most infectious between 7–10 days before and 7–10 days after the appearance of symptoms, but transmission is possible as long as the virus remains in the saliva or feces.[20]
Factors that increase the risk of polio infection or affect the severity of the disease include immune deficiency,[45]older age,[3] malnutrition,[46] recent tonsillectomy,[47] excessively strenuous physical activity immediately following the onset of paralysis,[48][3] skeletal muscle injury due to injection of vaccines or therapeutic agents,[49] and pregnancy.[50] Although the virus can cross the placenta during pregnancy, the fetus does not appear to be affected by either maternal infection or polio vaccination.[51] Maternal antibodies also cross the placenta, providing passive immunity that protects the infant from polio infection during the first few months of life.[52]
Pathophysiology and symptoms
Poliovirus enters the body through the mouth, infecting the first cells it comes in contact with—the pharynx (throat) and intestinal mucosa. It gains entry by binding to an immunoglobulin-like receptor, known as the poliovirus receptor or CD155, on the cell membrane.[28] The virus then hijacks the host cell's own machinery, and begins to replicate. Poliovirus divides within gastrointestinal cells for about a week, from where it spreads to the tonsils (specifically the follicular dendritic cells residing within the tonsilar germinal centers), the intestinal lymphoid tissue including the M cells of Peyer's patches, and the deep cervical and mesenteric lymph nodes, where it multiplies abundantly. The virus is subsequently absorbed into the bloodstream.[32]
Known as viremia, the presence of virus in the bloodstream enables it to be widely distributed throughout the body. Poliovirus can survive and multiply within the blood and lymphatics for long periods of time, sometimes as long as 17 weeks.[53] In a small percentage of cases, it can spread and replicate in other sites such as brown fat, the reticuloendothelial tissues, and muscle.[54] This sustained replication causes a major viremia, and leads to the development of minor influenza-like symptoms. Rarely, this may progress and the virus may invade the central nervous system (CNS), provoking a local inflammatory response. In most cases this causes a self-limiting inflammation of the meninges, the layers of tissue surrounding the brain, which is known as non-paralytic aseptic meningitis.[6] Penetration of the CNS provides no known benefit to the virus, and is quite possibly an incidental deviation of a normal gastrointestinal infection.[33] The mechanisms by which poliovirus spreads to the CNS are poorly understood, but it appears to be primarily a chance event—largely independent of the age, gender, or socioeconomic position of the individual.[33]
Paralytic polio
In around 1% of infections, poliovirus spreads along certain nerve fiber pathways, preferentially replicating in and destroying motor neurons within the spinal cord, brain stem, or motor cortex. This leads to the development of paralytic poliomyelitis, the various forms of which (spinal, bulbar, and bulbospinal) vary only with the amount of neuronal damage and inflammation that occurs, and the region of the CNS that is affected.
The destruction of neuronal cells produces lesions within the spinal ganglia; these may also occur in the reticular formation, vestibular nuclei, cerebellar vermis, and deep cerebellar nuclei.[33] Inflammation associated with nerve cell destruction often alters the color and appearance of the gray matter in the spinal column, causing it to appear reddish and swollen.[6] Other destructive changes associated with paralytic disease occur in the forebrain region, specifically the hypothalamus and thalamus.[33] The molecular mechanisms by which poliovirus causes paralytic disease are poorly understood.
Early symptoms of paralytic polio include high fever, headache, stiffness in the back and neck, asymmetrical weakness of various muscles, sensitivity to touch, difficulty swallowing, muscle pain, loss of superficial and deep reflexes, paresthesia (pins and needles), irritability, constipation, or difficulty urinating. Paralysis generally develops one to ten days after early symptoms begin, progresses for two to three days, and is usually complete by the time the fever breaks.[55]
The likelihood of developing paralytic polio increases with age, as does the extent of paralysis. In children, non-paralytic meningitis is the most likely consequence of CNS involvement, and paralysis occurs in only 1 in 1000 cases. In adults, paralysis occurs in 1 in 75 cases.[56] In children under five years of age, paralysis of one leg is most common; in adults, extensive paralysis of the chest and abdomen also affecting all four limbs—quadriplegia—is more likely.[57] Paralysis rates also vary depending on the serotype of the infecting poliovirus; the highest rates of paralysis (1 in 200) are associated with poliovirus type 1, the lowest rates (1 in 2,000) are associated with type 2.[58]
Spinal polio
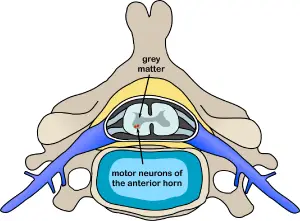
Spinal polio is the most common form of paralytic poliomyelitis; it results from viral invasion of the motor neurons of the anterior horn cells, or the ventral (front) gray matter section in the spinal column, which are responsible for movement of the muscles, including those of the trunk, limbs and the intercostal muscles.[8] Virus invasion causes inflammation of the nerve cells, leading to damage or destruction of motor neuron ganglia. When spinal neurons die, Wallerian degeneration takes place, leading to weakness of those muscles formerly innervated by the now dead neurons.[59] With the destruction of nerve cells, the muscles no longer receive signals from the brain or spinal cord; without nerve stimulation, the muscles atrophy, becoming weak, floppy and poorly controlled, and finally completely paralyzed.[8] Progression to maximum paralysis is rapid (two to four days), and is usually associated with fever and muscle pain.[59] Deep tendon reflexes are also affected, and are usually absent or diminished; sensation (the ability to feel) in the paralyzed limbs, however, is not affected.[59]
The extent of spinal paralysis depends on the region of the cord affected, which may be cervical, thoracic, or lumbar.[60] The virus may affect muscles on both sides of the body, but more often the paralysis is asymmetrical.[32] Any limb or combination of limbs may be affected—one leg, one arm, or both legs and both arms. Paralysis is often more severe proximally (where the limb joins the body) than distally (the fingertips and toes).[32]
Bulbar polio
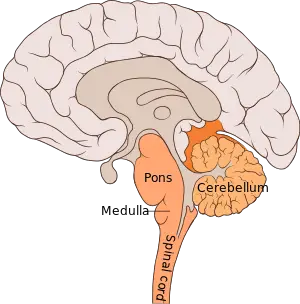
Making up about two percent of cases of paralytic polio, bulbar polio occurs when poliovirus invades and destroys nerves within the bulbar region of the brain stem.[1] The bulbar region is a white matter pathway that connects the cerebral cortex to the brain stem. The destruction of these nerves weakens the muscles supplied by the cranial nerves, producing symptoms of encephalitis, and causes difficulty breathing, speaking and swallowing.[7] Critical nerves affected are the glossopharyngeal nerve, which partially controls swallowing and functions in the throat, tongue movement and taste; the vagus nerve, which sends signals to the heart, intestines, and lungs; and the accessory nerve, which controls upper neck movement. Due to the effect on swallowing, secretions of mucus may build up in the airway causing suffocation.[55] Other signs and symptoms include facial weakness, caused by destruction of the trigeminal nerve and facial nerve, which innervate the cheeks, tear ducts, gums, and muscles of the face, among other structures; double vision; difficulty in chewing; and abnormal respiratory rate, depth, and rhythm, which may lead to respiratory arrest. Pulmonary edema and shock are also possible, and may be fatal.[60]
Bulbospinal polio
Approximately 19% of all paralytic polio cases have both bulbar and spinal symptoms; this subtype is called respiratory polio or bulbospinal polio.[1] Here, the virus affects the upper part of the cervical spinal cord (C3 through C5), and paralysis of the diaphragm occurs. The critical nerves affected are the phrenic nerve, which drives the diaphragm to inflate the lungs, and those that drive the muscles needed for swallowing. By destroying these nerves this form of polio affects breathing, making it difficult or impossible for the patient to breathe without the support of a ventilator. It can lead to paralysis of the arms and legs and may also affect swallowing and heart functions.[61]
Diagnosis
Paralytic poliomyelitis may be clinically suspected in individuals experiencing acute onset of flaccid paralysis in one or more limbs with decreased or absent tendon reflexes in the affected limbs that cannot be attributed to another apparent cause, and without sensory or cognitive loss.[62]Asymmetric flacid paralysis without sensory loss in a young adult or child, accompanied by a fever, is almost always indicative of polio.[3]
A laboratory diagnosis is usually made based on recovery of poliovirus from a stool sample or a swab of the pharynx. Antibodies to poliovirus can be diagnostic, and are generally detected in the blood of infected patients early in the course of infection.[1] Analysis of the patient's cerebrospinal fluid (CSF), which is collected by a lumbar puncture ("spinal tap"), reveals an increased number of white blood cells (primarily lymphocytes) and a mildly elevated protein level and no bacterial infection. Detection of virus in the CSF is diagnostic of paralytic polio, but rarely occurs.[1] Aseptic meningitis (no bacteria) cannot be distinguished clinically from nonparalytic poliomyelitis because of other agents and virus must be detected to confirm diagnosis.[3]
If poliovirus is isolated from a patient experiencing acute flaccid paralysis, it is further tested through oligonucleotide mapping (genetic fingerprinting), or more recently by PCR amplification, to determine whether it is "wild type" (that is, the virus encountered in nature) or "vaccine type" (derived from a strain of poliovirus used to produce polio vaccine).[63] It is important to determine the source of the virus because for each reported case of paralytic polio caused by wild poliovirus, it is estimated that another 200 to 3,000 contagious asymptomatic carriers exist.[64]
Treatment
There is no cure for polio. The focus of modern treatment has been on providing relief of symptoms, speeding recovery and preventing complications. Supportive measures include antibiotics to prevent infections in weakened muscles, analgesics for pain, moderate exercise and a nutritious diet.[65] Treatment of polio often requires long-term rehabilitation, including physical therapy, braces, corrective shoes and, in some cases, orthopedic surgery.[60]
Portable ventilators may be required to support breathing. Historically, a noninvasive negative-pressure ventilator, more commonly called an iron lung, was used to artificially maintain respiration during an acute polio infection until a person could breathe independently (generally about one to two weeks). Today, many polio survivors with permanent respiratory paralysis use modern jacket-type negative-pressure ventilators that are worn over the chest and abdomen.[66]
Other historical treatments for polio include hydrotherapy, electrotherapy, massage and passive motion exercises, and surgical treatments such as tendon lengthening and nerve grafting.[8] Devices such as rigid braces and body casts—which tended to cause muscle atrophy due to the limited movement of the user—were also touted as effective treatments.[67]
Prognosis
Patients with abortive polio infections (the minor of the two basic patterns) recover completely. In those that develop only aseptic meningitis, the symptoms can be expected to persist for two to ten days, followed by complete recovery.[68] In cases of spinal polio, if the affected nerve cells are completely destroyed, paralysis will be permanent; cells that are not destroyed but lose function temporarily may recover within four to six weeks after onset.[68] Half the patients with spinal polio recover fully; one quarter recover with mild disability and the remaining quarter are left with severe disability.[69] The degree of both acute paralysis and residual paralysis is likely to be proportional to the degree of viremia, and inversely proportional to the degree of immunity.[33] Spinal polio is rarely fatal.[55]
Without respiratory support, consequences of poliomyelitis with respiratory involvement include suffocation or pneumonia from aspiration of secretions.[66] Overall, 5–10% of patients with paralytic polio die due to the paralysis of muscles used for breathing. The mortality rate varies by age: 2–5% of children and up to 15–30% of adults die.[1] Bulbar polio often causes death if respiratory support is not provided;[61] with support, its mortality rate ranges from 25 to 75%, depending on the age of the patient.[1][70] When positive pressure ventilators are available, the mortality can be reduced to 15%.[71]
Recovery
Many cases of poliomyelitis result in only temporary paralysis.[8] Nerve impulses return to the formerly paralyzed muscle within a month, and recovery is usually complete in six to eight months.[68] The neurophysiological processes involved in recovery following acute paralytic poliomyelitis are quite effective; muscles are able to retain normal strength even if half the original motor neurons have been lost.[72] Paralysis remaining after one year is likely to be permanent, although modest recoveries of muscle strength are possible 12 to 18 months after infection.[68]
One mechanism involved in recovery is nerve terminal sprouting, in which remaining brainstem and spinal cord motor neurons develop new branches, or axonal sprouts.[73] These sprouts can reinnervate orphaned muscle fibers that have been denervated by acute polio infection,[74] restoring the fibers' capacity to contract and improving strength.[75] Terminal sprouting may generate a few significantly enlarged motor neurons doing work previously performed by as many as four or five units:[56] a single motor neuron that once controlled 200 muscle cells might control 800 to 1000 cells. Other mechanisms that occur during the rehabilitation phase, and contribute to muscle strength restoration, include myofiber hypertrophy—enlargement of muscle fibers through exercise and activity—and transformation of type II muscle fibers to type I muscle fibers.[74][76]
In addition to these physiological processes, the body possesses a number of compensatory mechanisms to maintain function in the presence of residual paralysis. These include the use of weaker muscles at a higher than usual intensity relative to the muscle's maximal capacity, enhancing athletic development of previously little-used muscles, and using ligaments for stability, which enables greater mobility.[76]
Complications
Residual complications of paralytic polio often occur following the initial recovery process.[7] Muscle paresis and paralysis can sometimes result in skeletal deformities, tightening of the joints, and movement disability. Once the muscles in the limb become flaccid, they may interfere with the function of other muscles. A typical manifestation of this problem is equinus foot (similar to club foot). This deformity develops when the muscles that pull the toes downward are working, but those that pull it upward are not, and the foot naturally tends to drop toward the ground. If the problem is left untreated, the Achilles tendons at the back of the foot retract and the foot cannot take on a normal position. Polio victims that develop equinus foot cannot walk properly because they cannot put their heel on the ground. A similar situation can develop if the arms become paralyzed.[77] In some cases, the growth of an affected leg is slowed by polio, while the other leg continues to grow normally. The result is that one leg is shorter than the other and the person limps and leans to one side, in turn leading to deformities of the spine (such as scoliosis).[77] Osteoporosis and increased likelihood of bone fractures may occur. Extended use of braces or wheelchairs may cause compression neuropathy, as well as a loss of proper function of the veins in the legs, due to pooling of blood in paralyzed lower limbs.[61][78] Complications from prolonged immobility involving the lungs, kidneys, and heart include pulmonary edema, aspiration pneumonia, urinary tract infections, kidney stones, paralytic ileus, myocarditis, and cor pulmonale.[61][78]
Post-polio syndrome
Around a quarter of individuals who survive paralytic polio in childhood develop additional symptoms decades after recovering from the acute infection, notably muscle weakness, extreme fatigue, or paralysis. This condition is known as post-polio syndrome (PPS) or post-polio sequelae.[75] The symptoms of PPS are thought to involve a failure of the over-sized motor units created during recovery from paralytic disease.[79][80] Factors that increase the risk of PPS include the length of time since acute poliovirus infection, the presence of permanent residual impairment after recovery from the acute illness, and both overuse and disuse of neurons.[75] Post-polio syndrome is not an infectious process, and persons experiencing the syndrome do not shed poliovirus.[1]
Prevention
Passive immunization
In 1950, William Hammon at the University of Pittsburgh purified the gamma globulin component of the blood plasma of polio survivors.[81] Hammon proposed that the gamma globulin, which contained antibodies to poliovirus, could be used to halt poliovirus infection, prevent disease, and reduce the severity of disease in other patients who had contracted polio. The results of a large clinical trial were promising; the gamma globulin was shown to be about 80% effective in preventing the development of paralytic poliomyelitis.[82] It was also shown to reduce the severity of the disease in patients that developed polio.[81] The gamma globulin approach was later deemed impractical for widespread use, however, due in large part to the limited supply of blood plasma, and the medical community turned its focus to the development of a polio vaccine.[83]
Vaccine
Two types of vaccines are used throughout the world to combat polio. Both types induce immunity to polio, efficiently blocking person-to-person transmission of wild poliovirus, thereby protecting both individual vaccine recipients and the wider community (so-called herd immunity).[84]
The first candidate polio vaccine, based on one serotype of a live but attenuated (weakened) virus, was developed by the virologist Hilary Koprowski. Koprowski's prototype vaccine was given to an eight-year-old boy from Letchworth Village, New York on February 27, 1950. There were no side effects.Koprowski continued to work on the vaccine throughout the 1950s, leading to large-scale trials in the then Belgian Congo and the vaccination of seven million children in Poland against serotypes PV1 and PV3 between 1958 and 1960.[85]
The second inactivated virus vaccine was developed in 1952 by Jonas Salk, and announced to the world on April 12, 1955.[86] The Salk vaccine, or inactivated poliovirus vaccine (IPV), is based on poliovirus grown in a type of monkey kidney tissue culture (Vero cell line), which is chemically inactivated with formalin.[21] After two doses of IPV (given by injection), 90% or more of individuals develop protective antibody to all three serotypes of poliovirus, and at least 99% are immune to poliovirus following three doses.[1]
Subsequently, Albert Sabin developed another live, oral polio vaccine (OPV). It was produced by the repeated passage of the virus through non-human cells at sub-physiological temperatures.[87] The attenuated poliovirus in the Sabin vaccine replicates very efficiently in the gut, the primary site of wild poliovirus infection and replication, but the vaccine strain is unable to replicate efficiently within nervous system tissue.[88] A single dose of Sabin's oral polio vaccine produces immunity to all three poliovirus serotypes in approximately 50% of recipients. Three doses of live-attenuated OPV produce protective antibody to all three poliovirus types in more than 95% of recipients.[1] Human trials of Sabin's vaccine began in 1957, and in 1958 it was selected, in competition with the live vaccines of Koprowski and other researchers, by the US National Institutes of Health.[85] It was licensed in 1962 and rapidly became the only polio vaccine used worldwide.[85]
Because OPV is inexpensive, easy to administer, and produces excellent immunity in the intestine (which helps prevent infection with wild virus in areas where it is endemic), it has been the vaccine of choice for controlling poliomyelitis in many countries.[89] On very rare occasions (about 1 case per 750,000 vaccine recipients) the attenuated virus in OPV reverts into a form that can paralyze.[23] Most industrialized countries have switched to IPV, which cannot revert, either as the sole vaccine against poliomyelitis or in combination with oral polio vaccine.[90]
Eradication
WHO 2002
While now rare in the Western world, polio is still endemic to parts of South Asia and Africa. Following the widespread use of poliovirus vaccine in the mid-1950s, the incidence of poliomyelitis declined dramatically in many industrialized countries. In 1985, the Rotary Foundation commenced an effort to vaccinate children worldwide. In 1988, a coordinated global effort to eradicate polio, the Global Polio Eradication Initiative, was led by the World Health Organization, UNICEF, and The Rotary Foundation. These efforts have reduced the number of annual diagnosed cases by 99%; from an estimated 350,000 cases in 1988 to a low of 483 cases in 2001, after which it has remained at a level of about 1,000 cases per year (1,606 in 2009).[91][92] Polio is one of only two diseases currently the subject of a global eradication program, the other being Guinea worm disease. If the global Polio Eradication initiative is successful before that for Guinea worm or any other disease, it would be only the third time humankind has ever completely eradicated a disease, after smallpox in 1979[93] and rinderpest in 2010.[94]
A number of eradication milestones have already been reached, and several regions of the world have been certified polio-free. The Americas were declared polio-free in 1994.[95] In 2000, polio was officially eliminated in 36 Western Pacific countries, including China and Australia.[96][97] Europe was declared polio-free in 2002.[98]
As of 2006, polio remains endemic in Nigeria, India (specifically Uttar Pradesh and Bihar), Pakistan, and Afghanistan,[91][99] although it continues to cause epidemics in other nearby countries born of hidden or reestablished transmission.
History
The apparent effects of polio have been known since prehistory; Egyptian paintings and carvings depict otherwise healthy people with withered limbs, and children walking with canes at a young age.[10] The first clinical description was provided by the English physician Michael Underwood in 1789, where he refers to polio as "a debility of the lower extremities".[100] The work of physicians Jakob Heine in 1840 and Karl Oskar Medin in 1890 led to it being known as Heine-Medin disease.[101] The disease was later called infantile paralysis, based on its propensity to affect children.
Before the 20th century, polio infections were rarely seen in infants before six months of age, most cases occurring in children six months to four years of age.[102] Poorer sanitation of the time resulted in a constant exposure to the virus, which enhanced a natural immunity within the population. In developed countries during the late 19th and early 20th centuries, improvements were made in community sanitation, including better sewage disposal and clean water supplies. These changes drastically increased the proportion of children and adults at risk of paralytic polio infection, by reducing childhood exposure and immunity to the disease.
Small localized paralytic polio epidemics began to appear in Europe and the United States around 1900.[11] Outbreaks reached pandemic proportions in Europe, North America, Australia, and New Zealand during the first half of the 20th century. By 1950, the peak age incidence of paralytic poliomyelitis in the United States had shifted from infants to children aged five to nine years, when the risk of paralysis is greater; about one-third of the cases were reported in persons over 15 years of age.[103] Accordingly, the rate of paralysis and death due to polio infection also increased during this time.[11] In the United States, the 1952 polio epidemic became the worst outbreak in the nation's history. Of nearly 58,000 cases reported that year, 3,145 died and 21,269 were left with mild to disabling paralysis.[104] Intensive-care medicine has its origin in the fight against polio. Most hospitals in the 1950s had limited access to iron lungs for patients unable to breathe without mechanical assistance. The establishment of respiratory centers to assist the most severe polio patients was hence the harbinger of subsequent ICUs.[105]
The polio epidemics changed not only the lives of those who survived them, but also affected profound cultural changes, spurring grassroots fund-raising campaigns that would revolutionize medical philanthropy, and give rise to the modern field of rehabilitation therapy. As one of the largest disabled groups in the world, polio survivors also helped to advance the modern disability rights movement through campaigns for the social and civil rights of the disabled. The World Health Organization estimates that there are up to 20 million polio survivors worldwide.[106] In 1977, there were 254,000 persons living in the United States who had been paralyzed by polio.[107] According to doctors and local polio support groups, some 40,000 polio survivors with varying degrees of paralysis live in Germany, 30,000 in Japan, 24,000 in France, 16,000 in Australia, 12,000 in Canada and 12,000 in the United Kingdom.[106] Many notable individuals have survived polio and often credit the prolonged immobility and residual paralysis associated with polio as a driving force in their lives and careers.[108]
The disease was very well publicized during the polio epidemics of the 1950s, with extensive media coverage of any scientific advancements that might lead to a cure. Thus, the scientists working on polio became some of the most famous of the century. Fifteen scientists and two laymen who made important contributions to the knowledge and treatment of poliomyelitis are honored by the Polio Hall of Fame, which was dedicated in 1957 at the Roosevelt Warm Springs Institute for Rehabilitation in Warm Springs, Georgia, USA. In 2008, four organizations (Rotary International, the World Health Organization, the U.S. Centers for Disease Control, and UNICEF) were added to the Hall of Fame.[109][110]
Notes and references
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 W. Atkinson, J. Hamborsky, L. McIntyre, and S. Wolfe, "Poliomyelitis," in W. Atkinson, J. Hamborsky, L. McIntyre, and S. Wolfe, Epidemiology and Prevention of Vaccine-Preventable Diseases, 11th edition (Washington D.C.: Public Health Foundation, 2009). ISBN 9780017066084. Retrieved February 19, 2011.
- ↑ 2.0 2.1 J. I. Cohen, "Enteroviruses and Reoviruses," In D. L. Kasper, E. Braunwald, A. S. Fauci, et al., Harrison's Principles of Internal Medicine, 16th ed. (McGraw-Hill Professional, 2004). ISBN 0071402357.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 L. K. Bennington, "Polio," in J. L. Longe, The Gale Encyclopedia of Medicine, Second Edition, Volume 4 (Detroit: Gale Group/Thomson Learning, 2002). ISBN 0787654930.
- ↑ 4.0 4.1 4.2 4.3 4.4 K. J. Ryan, and C. G. Ray, "Enteroviruses," in K. J. Ryan and C. G. Ray, Sherris Medical Microbiology, 4th edition (McGraw Hill, 2004). ISBN 08385-8529-9.
- ↑ M. Falconer, and E. Bollenbach, "Late functional loss in nonparalytic polio," American Journal of Physical Medicine & Rehabilitation 79(2000), iss. 1: 19–23. PMID 10678598.
- ↑ 6.0 6.1 6.2 6.3 S. L. Chamberlin, and B. Narins (eds.), The Gale Encyclopedia of Neurological Disorders (Detroit: Thomson Gale, 2005) ISBN 078769150X.
- ↑ 7.0 7.1 7.2 C. Leboeuf, The Late Effects of Polio: Information for Health Care Providers (Commonwealth Department of Community Services and Health, 1992). ISBN 1875412050.
- ↑ 8.0 8.1 8.2 8.3 8.4 H. W. A. Frauenthal and J. V. V. Manning, Manual of Infantile Paralysis, With Modern Methods of Treatment (Philadelphia Davis, 1914).
- ↑ L. D. H. Wood, J. B. Hall, and G. D. Schmidt, Principles of Critical Care, 3rd edition (McGraw-Hill Professional, 2005). ISBN 0071416404.
- ↑ 10.0 10.1 10.2 10.3 10.4 J. R. Paul, A History of Poliomyelitis (New Haven, CT: Yale University Press, 1971). ISBN 0300013248.
- ↑ 11.0 11.1 11.2 B. Trevelyan, M. Smallman-Raynor, and A. Cliff, "The spatial dynamics of poliomyelitis in the United States: From epidemic emergence to vaccine-induced retreat, 1910–1971," Ann Assoc Am Geogr 95(2005)iss. 2: 269–93. PMID 16741562.
- ↑ R. Aylward, "Eradicating polio: Today's challenges and tomorrow's legacy," Ann Trop Med Parasitol 100(2006)iss. 5–6: 401–13. PMID 16899145.
- ↑ D. Heymann, "Global polio eradication initiative," Bull. World Health Organ. 84(2006)iss. 8: 595. PMID 16917643. Retrieved February 19, 2011.
- ↑ 14.0 14.1 D. G. McNeil, "In battle against polio, a call for a final salvo," New York Times February 1, 2011. Retrieved February 19, 2011.
- ↑ 15.0 15.1 D. G. McNeil, "Can polio be eradicated? A skeptic now thinks so," New York Times February 14, 2011. Retrieved February 19, 2011.
- ↑ International Committee on Taxonomy of Viruses (ICTV), ICTV 2009 Master Species List Version 9 (ICTV, 2009). Retrieved February 19, 2011.
- ↑ 17.0 17.1 J. Hogle, "Poliovirus cell entry: Common structural themes in viral cell entry pathways," Annu Rev Microbiol 56(2002): 677–702. PMID 12142481.
- ↑ 18.0 18.1 18.2 D. S. Goodsell, The Machinery of Life (New York: Copernicus, 1998). ISBN 0387982736.
- ↑ S. L. Katz, A. A. Gershon, S. Krugman, and J. Peter, Krugman's Infectious Diseases of Children (St. Louis, MO: Mosby, 2004). ISBN 0323017568.
- ↑ 20.0 20.1 20.2 L. K. Ohri, and J. G. Marquess, "Polio: Will we soon vanquish an old enemy?" Drug Benefit Trends 11(1999)iss. 6: 41–54. Retrieved February 20, 2011.
- ↑ 21.00 21.01 21.02 21.03 21.04 21.05 21.06 21.07 21.08 21.09 21.10 O. Kew, R. Sutter, E. de Gourville, W. Dowdle, and M. Pallansch, "Vaccine-derived polioviruses and the endgame strategy for global polio eradication," Annu Rev Microbiol 59(2005): 587–635. PMID 16153180. Cite error: Invalid
<ref>tag; name "Kew_2005" defined multiple times with different content - ↑ K. C. Gorson, and A. H. Ropper, "Nonpoliovirus poliomyelitis simulating Guillain-Barré syndrome," Archives of Neurology 58(2001)iss. 9: 1460–4. PMID 11559319. Retrieved February 19, 2011.
- ↑ 23.0 23.1 23.2 23.3 23.4 V. Racaniello and D. Baltimore, "Molecular cloning of poliovirus cDNA and determination of the complete nucleotide sequence of the viral genome," Proceedings of the National Academy Science USA 78(1981)iss. 8: 4887–91. PMID 6272282. Cite error: Invalid
<ref>tag; name "Racaniello" defined multiple times with different content Cite error: Invalid<ref>tag; name "Racaniello" defined multiple times with different content - ↑ N. Kitamura, B. Semler, P. Rothberg, et. al., "Primary structure, gene organization and polypeptide expression of poliovirus RNA," Nature 291(1981)iss. 5816: 547–53. PMID 6264310.
- ↑ E. B. Carstens, and L. A. Ball, "Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2008)," Archives of Virology 154(2009)iss. 7: 1181–1188. Retrieved February 20, 2011.
- ↑ 26.0 26.1 N. H. De Jesus, "Epidemics to eradication: The modern history of poliomyelitis," Virol. J.4(2007): 70. PMID 17623069. Retrieved February 19, 2011.
- ↑ C. Mendelsohn, E. Wimmer, and V. R. Racaniello, "Cellular receptor for poliovirus: Molecular cloning, nucleotide sequence, and expression of a new member of the immunoglobin superfamily," Cell 56(1989)iss. 5: 855–865. PMID 2538245.
- ↑ 28.0 28.1 Y. He, S. Mueller, P. Chipman, et al., "Complexes of poliovirus serotypes with their common cellular receptor, CD155," J Virol 77(2003)iss. 8: 4827–35. PMID 12663789. Retrieved February 20, 2011.
- ↑ T. H. Dunnebacke, J. D. Levinthal, and R. C. Williams, [http://jvi.asm.org/cgi/pmidlookup?view=long&pmid=4309884 "Entry and release of poliovirus as observed by electron microscopy of cultured cells," J. Virol. 4(1969)iss. 4: 505–13. PMID 4309884. Retrieved February 20, 2011.
- ↑ G. Kaplan, M. S. Freistadt, and V. R. Racaniello, "Neutralization of poliovirus by cell receptors expressed in insect cells," J. Virol. 64(1990)iss. 10: 4697–702. PMID 2168959. Retrieved February 20, 2011.
- ↑ A. Gomez Yafal, G. Kaplan, V. R. Racaniello, and J. M. Hogle, "Characterization of poliovirus conformational alteration mediated by soluble cell receptors," Virology 197(1993)iss. 1: 501–505. PMID 8212594.
- ↑ 32.0 32.1 32.2 32.3 M. Yin-Murphy, and J. W. Almond, "Picornaviruses: The enteroviruses: Polioviruses," in S. Baron et al. (eds.), Baron's Medical Microbiology, 4th edition(Univ of Texas Medical Branch, 1996). ISBN 0963117211. Retrieved February 20, 2011.
- ↑ 33.0 33.1 33.2 33.3 33.4 33.5 33.6 S. Mueller, E. Wimmer, and J. Cello, "Poliovirus and poliomyelitis: A tale of guts, brains, and an accidental event," Virus Res 111(2005)iss. 2: 175–93. PMID 15885840.
- ↑ B. Brandenburg, L. Y. Lee, M. Lakadamyali, M. J. Rust, X. Zhuang, and J. M. Hogle, "Imaging poliovirus entry in live cells," PLOS Biology 5(2007)iss. 7: e183. PMID 17622193.
- ↑ C. Y. Chen, and P. Sarnow, "Initiation of protein synthesis by the eukaryotic translational apparatus on circular RNAs," Science 268(1995)iss. 5209: 415–7. PMID 7536344.
- ↑ J. Pelletier, and N. Sonenberg, "Internal initiation of translation of eukaryotic mRNA directed by a sequence derived from poliovirus RNA," Nature 334(1988)iss. 6180: 320–325. PMID 2839775.
- ↑ S. K. Jang, H. G. Kräusslich, M. J. Nicklin, G. M. Duke, A. C. Palmenberg, and E. Wimmer, "A segment of the 5' nontranslated region of encephalomyocarditis virus RNA directs internal entry of ribosomes during in vitro translation," J. Virol. 62(1988)iss. 8: 2636–43. PMID 2839690. Retrieved February 20, 2011.
- ↑ Wkly Epidemiol. Rec. (no authors listed), [http://www.ncbi.nlm.nih.gov/pubmed/11315462 "Transmission of wild poliovirus type 2: Apparent global interruption" Wkly. Epidemiol. Rec. 76(2001)iss. 13: 95–7. PMID 11315462. Retrieved February 20, 2011.
- ↑ Centers for Disease Control (CDC), "Apparent global interruption of wild poliovirus type 2 transmission," CDC (2001). Retrieved February 20, 2011.
- ↑ D. S. Goodsell, "Poliovirus and Rhinovirus: August 2001 Molecule of the Month," RCSB Protein Data Bank (2001). Retrieved February 20, 2011.
- ↑ V. Racaniello, and D. Baltimore, "Cloned poliovirus complemenatry DNA is infectious in mammalian cells," Science 214(1981)iss. 453: 916–9. PMID 6272391.
- ↑ J. Cello, A. V. Paul, and E. Wimmer, "Chemical synthesis of poliovirus cDNA: Generation of infectious virus in the absence of natural template," Science 297(2002)iss. 5583: 1016–8. PMID 12114528.
- ↑ J. Couzin, "Virology. Active poliovirus baked from scratch," Science 297(2002)iss. 5579: 174–5. PMID 12114601.
- ↑ 44.0 44.1 S. P. Parker (ed.), McGraw-Hill Concise Encyclopedia of Science & Technology (New York: McGraw-Hill, 1998) ISBN 0070526591.
- ↑ L. Davis, D. Bodian, D. Price, I. Butler, and J. Vickers, "Chronic progressive poliomyelitis secondary to vaccination of an immunodeficient child," New England Journal of Medicine 297(1977)iss. 5: 241–5. PMID 195206.
- ↑ R. Chandra, "Reduced secretory antibody response to live attenuated measles and poliovirus vaccines in malnourished children," British Medical Journal 2(1975)iss. 5971: 583–5. PMID 1131622.
- ↑ A. Miller, "Incidence of poliomyelitis; the effect of tonsillectomy and other operations on the nose and throat," Calif Med 77(1952)iss. 1: 19–21. PMID 12978882.
- ↑ D. Horstmann, "Acute poliomyelitis relation of physical activity at the time of onset to the course of the disease," Journal of the American Medical Association 142(1950)iss. 4: 236–41. PMID 15400610}}
- ↑ M. Gromeier, and E. Wimmer, "Mechanism of injury-provoked poliomyelitis," J. Virol. 72(1998)iss. 6: 5056–60. PMID 9573275.
- ↑ C. E. Evans, "Factors influencing the occurrence of illness during naturally acquired poliomyelitis virus infections, Bacteriol Rev 24(1960)iss. 4: 341–52. PMID 13697553.
- ↑ Joint Committee on Vaccination and Immunisation (A. Salisbury, M. Ramsay, and K. Noakes (eds.)), "Chapter 26:Poliomyelitis," in Immunisation Against Infectious Disease, 2006 | (Edinburgh: Stationery Office, 2006). ISBN 0113225288.
- ↑ A. Sauerbrei, A. Groh, A. Bischoff, J. Prager, and P. Wutzler, " Antibodies against vaccine-preventable diseases in pregnant women and their offspring in the eastern part of Germany," Med Microbiol Immunol 190(2002)iss. 4: 167–72. PMID 12005329.
- ↑ K. Todar, "Polio," in K. Todar, The Microbial World website (Madison, WI: University of Wisconsin, 2006). Retrieved February 20, 2011.
- ↑ A. Sabin, "Pathogenesis of poliomyelitis; reappraisal in the light of new data," Science 123(1956)iss. 3209: 1151–7. PMID 13337331.
- ↑ 55.0 55.1 55.2 A. Silverstein, V. Silverstein, and L. S. Nunn, Polio (Berkeley Heights, NJ: Enslow Publishers, 2001) ISBN 0766015920.
- ↑ 56.0 56.1 A. C. Gawne, and L. S. Halstead, "Post-polio syndrome: Pathophysiology and clinical management," Critical Review in Physical Medicine and Rehabilitation 7(1995): 147–88. Retrieved February 20, 2011.
- ↑ G. R. Young, "Occupational therapy and the postpolio syndrome," American Journal of Occupational Therapy 43(1989)iss. 2: 97–103. PMID 2522741. Retrieved February 20, 2011.
- ↑ N. Nathanson, and J. Martin, "The epidemiology of poliomyelitis: Enigmas surrounding its appearance, epidemicity, and disappearance," Am J Epidemiol 110(1979)iss. 6: 672–92. PMID 400274.
- ↑ 59.0 59.1 59.2 J. Cono, and L. N. Alexander, "Chapter 10, Poliomyelitis," in Vaccine Preventable Disease Surveillance Manual, 3rd ed. (Centers for Disease Control and Prevention, 2002). Retrieved February 20, 2011.
- ↑ 60.0 60.1 60.2 Lippincott Williams & Wilkins, Professional Guide to Diseases (Professional Guide Series) (Ambler, PA: Lippincott Williams & Wilkins, 2005). ISBN 158255370X.
- ↑ 61.0 61.1 61.2 61.3 W. G. Hoyt, N. Miller, and F. Walsh, Walsh and Hoyt's Clinical Neuro-ophthalmology (Hagerstown, MD: Lippincott Williams & Wilkins, 2005). ISBN 0781748143.
- ↑ Centers for Disease Control and Prevention (CDC), [ftp://ftp.cdc.gov/pub/Publications/mmwr/rr/rr4610.pdf "Case definitions for infectious conditions under public health surveillance," ''Morbidity and Mortality Weekly Report 46(1997)iss. RR-10: 26–7. PMID 9148133. Retrieved February 20, 2011.
- ↑ C. Chezzi, "Rapid diagnosis of poliovirus infection by PCR amplification," Journal of Clinical Microbiology 34(1996)iss. 7: 1722–5. PMID 8784577.
- ↑ A. Gawande, "The mop-up: eradicating polio from the planet, one child at a time," The New Yorker January 12, 2004, pages 34-40.
- ↑ T. M. Daniel, and F. C. Robbins, Polio (Rochester, NY: University of Rochester Press, 1997) ISBN 1580460666.
- ↑ 66.0 66.1 A. Goldberg, "Noninvasive mechanical ventilation at home: building upon the tradition," Chest 121(2002)iss. 2: 321–4. PMID 11834636.
- ↑ S. Oppewal, "Sister Elizabeth Kenny, an Australian nurse, and treatment of poliomyelitis victims," Image J Nurs Sch 29(1997)iss. 1: 83–7. PMID 9127546.
- ↑ 68.0 68.1 68.2 68.3 D. Neumann, "Polio: Its impact on the people of the United States and the emerging profession of physical therapy," Journal of Orthopaedic and Sports Physical Therapy 34(2004)iss. 8: 479–92. PMID 15373011. Retrieved February 21, 2011.
- ↑ S. J. Cuccurullo, Physical Medicine and Rehabilitation Board Review (Demos Medical Publishing, 2004). ISBN 1888799455. Retrieved February 21, 2011.
- ↑ A. H. Miller, and L. S. Buck, "Tracheotomy in bulbar poliomyelitis," California Medicine 72(1950)iss. 1: 34–6. PMID 15398892.
- ↑ G. Wackers, Constructivist Medicine (PhD-thesis) (Maastricht: Universitaire Pers Maastricht, 1994). Retrieved February 21, 2011.
- ↑ A. Sandberg, B. Hansson,a nd E. Stålberg, "Comparison between concentric needle EMG and macro EMG in patients with a history of polio," Clinical Neurophysiology 110(1999)iss. 11: 1900–8. PMID 10576485.
- ↑ N. R. Cashman, J. Covault, R. L. Wollman, and J. R. Sanes, "Neural cell adhesion molecule in normal, denervated, and myopathic human muscle," Ann. Neurol. 21(1987)iss. 5: 481–9. PMID 3296947.
- ↑ 74.0 74.1 J. C. Agre, A. A. Rodríquez, and J. A. Tafel, "Late effects of polio: Critical review of the literature on neuromuscular function," Archives of Physical Medicine and Rehabilitation 72(1991)iss. 11: 923–31. PMID 1929813.
- ↑ 75.0 75.1 75.2 D. A. Trojan, and N. R. Cashman, "Post-poliomyelitis syndrome," Muscle Nerve 31(2005)iss. 1: 6–19. PMID 15599928.
- ↑ 76.0 76.1 G. Grimby, G. Einarsson, M. Hedberg, and A. Aniansson, "Muscle adaptive changes in post-polio subjects," Scandinavian Journal of Rehabilitation Medicine 21(1989)iss. 1: 19–26. PMID 2711135.
- ↑ 77.0 77.1 Sanofi Pasteur, "Poliomyelitis virus (picornavirus, enterovirus), after-effects of the polio, paralysis, deformations," "Conquering polio," Sanofi Pasteur (2006). Retrieved February 21, 2011.
- ↑ 78.0 78.1 Mayo Clinic Staff, [http://www.mayoclinic.com/health/polio/DS00572/DSECTION=complications "Polio: Complications," Mayo Foundation for Medical Education and Research (MFMER) (2009). Retrieved February 21, 2011.
- ↑ J. Ramlow, M. Alexander, R. LaPorte, C. Kaufmann, and L. Kuller, "Epidemiology of the post-polio syndrome," Am. J. Epidemiol. 136(1992)iss. 7: 769–86. PMID 1442743.
- ↑ K. Lin, and Y. Lim, "Post-poliomyelitis syndrome: Case report and review of the literature," Ann Acad Med Singapore 34(2005)iss. 7: 447–9. PMID 16123820. Retrieved February 21, 2011.
- ↑ 81.0 81.1 W. Hammon, "Passive immunization against poliomyelitis," Monogr Ser World Health Organ 26(1955): 357–70. PMID 14374581.
- ↑ W. Hammon, L. Coriell, E. Ludwig, et al., "Evaluation of Red Cross gamma globulin as a prophylactic agent for poliomyelitis. 5. Reanalysis of results based on laboratory-confirmed cases," J Am Med Assoc 156(1954)iss. 1: 21–7. PMID
- ↑ C. Rinaldo, "Passive immunization against poliomyelitis: The Hammon gamma globulin field trials, 1951–1953," American Journal of Public Health 95(2005)iss. 5: 790–9. PMID 15855454.
- ↑ P. Fine, and I. Carneiro, "Transmissibility and persistence of oral polio vaccine viruses: Implications for the global poliomyelitis eradication initiative," Am J Epidemiol 150(1999)iss. 10: 1001–21. PMID 10568615. Retrieved February 21, 2011.
- ↑ 85.0 85.1 85.2 Sanofi Pasteur, "Competition to develop an oral vaccine," Sanofi Pasteur (2005). Retrieved February 20, 2011.
- ↑ B. Spice, "Tireless polio research effort bears fruit and indignation: The Salk vaccine: 50 years later," Pittsburgh Post-Gazette April 4, 2005. Retrieved February 21, 2011.
- ↑ A. B. Sabin, and L. R. Boulger, "History of Sabin attenuated poliovirus oral live vaccine strains," J Biol Stand 1(1973): 115–8.
- ↑ A. Sabin, M. Ramos-Alvarez, J. Alvarez-Amezquita, et al., "Live, orally given poliovirus vaccine. Effects of rapid mass immunization on population under conditions of massive enteric infection with other viruses," JAMA 173(1960): 1521–6. PMID 14440553.
- ↑ Committee on Infectious Diseases, American Academy of Pediatrics, "Poliomyelitis prevention: Recommendations for use of inactivated poliovirus vaccine and live oral poliovirus vaccine," Pediatrics 99(1997)iss. 2: 300–5. PMID 9024465. Retrieved February 21, 2011.
- ↑ World Health Organization (WHO), [http://web.archive.org/web/20080606170542/http://www.who.int/ith/vaccines/2007_routine_use/en/index11.html "Vaccines for routine use: Poliomyelitis," WHO (2008). Retrieved February 21, 2011.
- ↑ 91.0 91.1 Centers for Disease Control and Prevention (CDC), "Update on vaccine-derived polioviruses," Morbidity and Mortality Weekly Report 55(2006)iss. 40: 1093-7. PMID 17035927 . Retrieved February 20, 2011.
- ↑ Centers for Disease Control and Prevention (CDC), "Progress toward interruption of wild poliovirus transmission—worldwide, January 2007-April 2008," Morbidity and Mortality Weekly Report 57(2008)iss. 18: 489-94. PMID 18463607. Retrieved February 20, 2011.
- ↑ World Health Organization (WHO), "Smallpox," WHO (2011). Retrieved February 21, 2011.
- ↑ P. Ghosh, "Rinderpest virus has been wiped out, scientists say," BBC News October 14, 2010. Retrieved February 21, 2011.
- ↑ Centers for Disease Control and Prevention (CDC), "Certification of poliomyelitis eradication—the Americas, 1994," Morbidity and Mortality Weekly Report 43(1994)iss. 39: 720-722. PMID 7522302. Retrieved February 20, 2011.
- ↑ Health Education Research, [http://her.oxfordjournals.org/cgi/reprint/16/1/109.pdf "General news. Major milestone reached in global polio eradication: Western Pacific Region is certified polio-free," Health Educ Res 16(2001)iss. 1: 109. Retrieved February 21, 2011.
- ↑ R. D'Souza, M. Kennett, and C. Watson, "Australia declared polio free," Commun Dis Intell 26(2002)iss. 2: 253–60. PMID 12206379.
- ↑ World Health Organization, "Europe achieves historic milestone as region is declared polio-free," European Region of the World Health Organization June 21, 2002. Retrieved February 21, 2011.
- ↑ P. E. M. Fine, "Polio: Measuring the protection that matters most," J Infect Dis 200(2009)iss. 5: 673-675. PMID 19624277.
- ↑ M. Underwood, "Debility of the lower extremities," in M. Underwood, A Treatise on the Dieases [sic of Children, With General Directions for the Management of Infants From the Birth], vol. 2 (Philadelphia: T. Dobson, 1793) Retrieved February 22, 2011.
- ↑ J. Pearce, "Poliomyelitis (Heine-Medin disease)," J Neurol Neurosurg Psychiatry 76(2005)iss. 1: 128. PMID 15608013.
- ↑ S. Robertson, 'Module 6: Poliomyelitis," (Geneva, Switzerland: World Health Organization, 1993). Retrieved February 22, 2011.
- ↑ J. L. Melnick, "Poliomyelitis," pages 558-576 in J. Melnick, Tropical and Geographical Medicine, 2nd ed. (McGraw-Hill, 1990). ISBN 007068328X.
- ↑ E. Zamula, "A new challenge for former polio patients," FDA Consumer 25(1991)iss. 5: 21–5. Retrieved February 22, 2011.
- ↑ H. G. Wooten, "The voices of polio in Texas: Hot packs, warm springs and cold facts," joint meeting of East Texas Historical Association and West Texas Historical Association in Fort Worth, Texas, February 27, 2010; there were more than a half-dozen serious polio outbreaks in Texas between 1942 and 1955.
- ↑ 106.0 106.1 March of Dimes, After effects of polio can harm survivors 40 years later," March of Dimes (2001). Retrieved February 22, 2011.
- ↑ N. M. Frick, and R. L. Bruno, "Post-polio sequelae: Physiological and psychological overview," Rehabilitation Literature 47(1986)iss. 5–6: 106–11. PMID 3749588.
- ↑ R. L. Bruno, The Polio Paradox: Understanding and Treating "Post-Polio Syndrome" and Chronic Fatigue (New York: Warner Books, 2002). ISBN 0446690694.
- ↑ W. Skinner, "Four added to Polio Hall of Fame at Warm Springs," Times-Herald (Newnan, GA): November 15, 2008. Retrieved February 22, 2011.
- ↑ Centers for Disease Control and Prevention (CDC), "CDC Inducted into Polio Hall of Fame," CDC (January 23, 2009). Retrieved February 22, 2011.
Further reading
- Daniel, T. M., and F. C. Robbins. 1999. Polio. Rochester, NY: University of Rochester. ISBN 1580460666.
- Gould, T. 1997. A Summer Plague: Polio and Its Survivors. New Haven, CT: Yale University Press. ISBN 0300072767.
- Kluger, J. 2004. Splendid Solution: Jonas Salk and the Conquest of Polio. New York: G. P. Putnam's Sons. ISBN 0399152164. (A social history of the effects of polio on early 20th Century America and the search for a vaccine.)
- Oshinsky, D. M. 2005. Polio: An American Story. Oxford: Oxford University Press. ISBN 0195152948. (A Pulitzer Prize-winning account of the development of the Salk and Sabin rivalry, and of the key role of the National Foundation for Infantile Paralysis/March of Dimes).
- Shaffer, M. M., and B. Seytre. 2005. The Death of a Disease: A History of the Eradication of Poliomyelitis. New Brunswick, NJ: Rutgers University Press. ISBN 0813536774.
- Shell, M. 2005. Polio and Its Aftermath: The Paralysis of Culture. Cambridge: Harvard University Press. ISBN 0674013158.
- Wilson, D. J. 2005. Living With Polio: The Epidemic and Its Survivors. Chicago: University of Chicago Press. ISBN 0226901033.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.