Progesterone
| |
| |
Progesterone
| |
| Systematic name | |
| IUPAC name pregn-4-ene-3,20-dione | |
| Identifiers | |
| CAS number | 57-83-0 |
| ATC code | G03DA04 |
| PubChem | 5994 |
| DrugBank | APRD00700 |
| Chemical data | |
| Formula | C21H30O2 |
| Mol. weight | 314.47 |
| Synonyms | 4-pregnene-3,20-dione |
| Physical data | |
| Melt. point | 126°C (259°F) |
| Spec. rot | [α]D |
| Pharmacokinetic data | |
| Bioavailability | prolonged absorption, half-life approx 25-50 hours |
| Protein binding | 96 percent-99 percent |
| Metabolism | hepatic to pregnanediols and pregnanolones |
| Half life | 34.8-55.13 hours |
| Excretion | renal |
| Therapeutic considerations | |
| Pregnancy cat. | B (USA) |
| Legal status | ? |
| Routes | oral, implant |
Progesterone is a steroid hormone in mammals that is involved in the female menstrual cycle, pregnancy (supporting gestation), and embryogenesis. The term progesterone also is used for natural or synthetic steroids that have a progesterone-like action on the body (Blakemore and Jennett 2001).
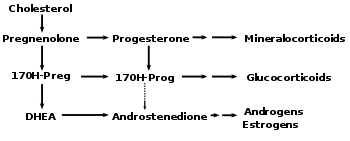
Progesterone belongs to a class of sex hormones called progestogens. Progesterone is the main progestogen produced in ovaries, particularly in the second half of the menstrual cycle after the occurrence of ovulation and the formation of a corpus luteum by the empty follicle (Blakemore and Jennett 2001). It is synthesized in the initial steps in the biosynthetic pathway involving the conversion of cholesterol to the sex steroids testosterone (an androgen) and estradiol (an estrogen), among others. It also is a precursor in the conversion to cortisol and aldosterone.
In addition to impacts on the reproductive system, progesterone also has impacts on the nervous system, immune system, skeletal system, thyroid function, and numerous other body functions. Human creativity has utilized natural progesterone and many synthetic analogues in many medical applications.
Chemistry
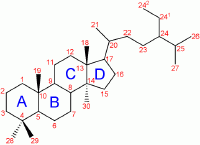
Progesterone is a steroid hormone. A steroid is any of a group of natural or synthetic, fat-soluble, organic compounds belonging to the class of lipids and characterized by a molecular core of four fused rings totaling 17 carbon atoms: Three six-carbon rings and one five-carbon ring fused together. The type of steroid is determined by the three-dimensional configuration and the type of additional side chains and rings.
Steroid hormones are steroids that act as hormones. Hormones are chemical messengers secreted by cells (including tissues and organs) in one part of a multicellular organism to travel to and coordinate the activities of different cells. An enormous range of chemicals are used for this type of cell-to-cell communication, including peptides (chains of amino acids) and steroids.
Progesterone is a type of progestogen, a subset of sex steroids, with androgens and estrogens being the other major sex steroids. Simply defined, progestogens (or progestagens) are a class of hormones that produce effects similar to progesterone. Progestoges that are synthetic are often referred to as progestins.
Like other steroids, progesterone consists of four interconnected cyclic hydrocarbons. Progesterone contains ketone and oxygenated functional groups, as well as two methyl branches. Like all steroid hormones, it is hydrophobic. This is mostly due to its lack of very polar functional groups. Progesterone has the chemical formula C21H30O2.
Progesterone was first isolated from hamster ovaries by Willard Myron Allen, who co-discovered it with his anatomy professor, George Washington Corner, at the University of Rochester Medical School. Allen first determined its melting point, molecular weight, and partial molecular structure. He also gave it the name Progesterone, derived from PROGEstational STERoidal ketONE. An avid mathematician, Allen recognized that the molecular weight of progesterone is 100 x π = 314 dalt.
Synthesis and role as precursor
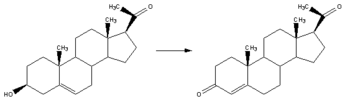
Progesterone, like all other steroid hormones, is synthesized from pregnenolone, a derivative of cholesterol. This conversion takes place in two steps. The 3-hydroxyl group is converted to a keto group and the double bond is moved to C-4, from C-5.
Progesterone is the precursor of the mineralocorticoid aldosterone. Likewise, after conversion to 17-hydroxyprogesterone, another natural progestogen, it is a precursor of cortisol and androstenedione. Androstenedione can be converted to testosterone, an androgen, and to the estrogen's estrone, and estradiol.
Sources and levels
Progesterone is produced in the adrenal glands, the gonads (specifically after ovulation in the corpus luteum), the brain, and, during pregnancy, in the placenta.
In general, two sex hormones play a role in the control of the menstrual cycle in humans: Estradiol and progesterone. While estrogen peaks twice, during follicular growth and during the luteal phase, progesterone remains virtually absent prior to ovulation, but becomes critical in the luteal phase and during pregnancy. Many tests for ovulation check for the presence of progesterone.
Progesterone has been called "the hormone of pregnancy" because of its critical role in supporting the endometrium (uterine lining) and hence the survival of the conceptus (Bowen 2000). In humans, increasing amounts of progesterone are produced during pregnancy:
- Initially, the source is the corpus luteum that has been "rescued" by the presence of human chorionic gonadotropins (hCG) from the conceptus.
- However, after the eighth week, production of progesterone shifts over to the placenta. The placenta utilizes maternal cholesterol as the initial substrate, and most of the produced progesterone enters the maternal circulation, but some is picked up by the fetal circulation and is used as substrate for fetal corticosteroids. At term the placenta produces about 250 mg progesterone per day.
The placenta of all mammals produce progesterone but the quantity varies significantly (Bowen 2000). In some species, such as human, horses, sheep, and cats, sufficient progesterone is produced by the placenta that the ovaries and corpora lutea can be removed after establishment of the placenta without loss of the pregnancy (Bowen 2000). In others, such as cattle, pigs, goats, and dogs, luteal progesterone is essential throughout gestation because the placental secretions are not sufficient (Bowen 2000).
In general, levels are relatively low during the preovulatory phase of the menstrual cycle, rise after ovulation, and are elevated during the luteal phase. In women, progesterone levels tend to be less than 2 ng/ml prior to ovulation, and greater than 5 ng/ml after ovulation. If pregnancy occurs, progesterone levels are maintained at luteal levels initially. With the onset of the luteal-placental shift in progesterone support of the pregnancy levels start to rise further and may reach 100-200 ng/ml at term. Whether a decrease in progesterone levels is critical for the initiation of labor has been argued and may be species-specific. After delivery of the placenta and during lactation, progesterone levels are very low.
Progesterone levels are relatively low in children and postmenopausal women (NIH 2007). Adult males have levels similar to those in women during the follicular phase of the menstrual cycle.
Effects
Progesterone exerts its action primarily through the intracellular progesterone receptor, though a distinct, membrane bound progesterone receptor has recently been discovered.
It has a number of physiological effects, often regulatory, especially of the effects of estrogen. Estrogen often induces a multiplication of progesterone receptors.
Reproductive system
Progesterone has many roles relating to the development of the fetus:
- Progesterone converts the endometrium (inner membrane of the mammalian uterus) to its secretory stage to prepare the uterus for implantation. At the same time, progesterone affects the vaginal epithelium and cervical mucus. If pregnancy does not occur, progesterone levels will decrease, leading, in the human, to menstruation. Normal menstrual bleeding is progesterone withdrawal bleeding.
- During implantation and gestation, progesterone appears to decrease the maternal immune response to allow for the acceptance of the pregnancy.
- Progesterone decreases contractability of the uterine smooth muscle (Bowen 2000).
- Progesterone inhibits lactation during pregnancy. The fall in progesterone levels following delivery is one of the triggers for milk production.
- A drop in progesterone levels is possibly one step that facilitates the onset of labor.
The fetus metabolizes placental progesterone in the production of adrenal mineralo- and glucosteroids.
Nervous system
Progesterone, like pregnenolone and dehydroepiandrosterone, belongs to the group of neurosteroids that are found in high concentrations in certain areas in the brain and are synthesized there.
Neurosteroids affect synaptic functioning, are neuroprotective, and affect myelinization (Schumacher et al. 2004). They are being investigated for their potential to improve memory and cognitive ability.
Progesterone as a neuroprotectant affects regulation of apoptotic genes.
Progesterone's effect as a neurosteroid works predominantly through the GSK-3 beta pathway, as an inhibitor. (Other GSK-3 beta inhibitors include bipolar mood stabilizers, lithium, and valproic acid.)
Other systems
- Progesterone raises epidermal growth factor-1 levels, a factor often used to induce proliferation, and sustain cultures, of stem cells.
- Progesterone increases core temperature (thermogenic function) during ovulation (MCG 2007).
- Progesterone reduces spasm and relaxes smooth muscle. Bronchi are widened and mucus regulated. (Progesterone receptors are widely present in submucosal tissue.)
- Progesterone acts as an antiinflammatory agent and regulates the immune response.
- Progesterone reduces gall bladder activity (Hould et al. 1988).
- Progesterone normalizes blood clotting and vascular tone, zinc and copper levels, cell oxygen levels, and use of fat stores for energy.
- Progesterone assists in thyroid function, in bone building by osteoblasts, in bone, teeth, gums, joint, tendon, ligament and skin resilience, and in some cases healing by regulating various types of collagen, and in nerve function and healing by regulating myelin.
- Progesterone appears to prevent endometrial cancer (involving the uterine lining) by regulating the effects of estrogen.
Medical applications
The use of progesterone and its analogues have many medical applications—both to address acute situations, and to address the long-term decline of natural progesterone levels. Because of the poor bioavailability of progesterone when taken orally, many synthetic progestins have been designed. However, the roles of progesterone may not be fulfilled by the synthetic progestins which in some cases were designed solely to mimic progesterone's uterine effects.
Bioavailability
Progesterone is poorly absorbed by oral ingestion unless micronized and in oil, or with fatty foods; it does not dissolve in water. Products such as Prometrium, Uteregestan, Minagest, and Microgest are, therefore, capsules containing micronized progesterone in oil. Generally this is peanut oil, which may cause serious [allergy|[allergic reactions]] in some people, but compounding pharmacies, which have the facilities and licenses to make their own products, can use alternatives.
Vaginal and rectal application is also effective, with products such as CRINONE and PROCHIEVE bioadhesive progesterone vaginal gels (the only progesterone products FDA-approved for use in infertility and during pregnancy) and Cycff, which is progesterone in cocoa butter in the form of pessaries. Progesterone can be given by injection, but because it has a short half-life the injections need to be daily. Implants, for a longer period, are also available. Marketing of progesterone pharmaceutical products, country to country, varies considerably, with many countries having no oral progesterone products marketed, but they can usually be specially imported by pharmacies through international wholesalers.
"Natural progesterone" products derived from yams, do not require a prescription. Wild yams contain a plant steroid called diosgenin, which the human body cannot metabolize into progesterone. Diosgenin can only be chemically processed into progesterone in labs.
Specific uses
- Progesterone is used medically to control anovulatory bleeding. It is also used to prepare the uterine lining in infertility therapy and to support early pregnancy. Patients with recurrent pregnancy loss due to inadequate progesterone production may receive progesterone.
- Progesterone is being investigated as potentially beneficial in treating multiple sclerosis, since the characteristic deterioration of nerve myelin insulation halts during pregnancy, when progesterone levels are raised; deterioration commences again when the levels drop.
- Progesterone receptor antagonists, or selective progesterone receptor modulators (SPRM)s, such as RU-486 (Mifepristone), can be used to prevent conception or induce medical abortions.
Note that oral birth control pills do not contain progesterone but a progestin.
Aging
Since most progesterone in males is created during testicular production of testosterone, and most in females by the ovaries, the shutting down (whether by natural or chemical means), or removal, of those inevitably causes a considerable reduction in progesterone levels. Previous concentration upon the role of progestagens (progesterone and molecules with similar effects) in female reproduction, when progesterone was simply considered a "female hormone," obscured the significance of progesterone elsewhere in both sexes.
The tendency for progesterone to have a regulatory effect, the presence of progesterone receptors in many types of body tissue, and the pattern of deterioration (or tumor formation) increasing in later years when progesterone levels have dropped, is prompting widespread research into the potential value of maintaining progesterone levels in both males and females.
Additional images
ReferencesISBN links support NWE through referral fees
- Blakemore, C. and S. Jennett. 2001. The Oxford Companion to the Body. New York: Oxford University Press. ISBN 019852403X
- Hould, F., G. Fried, A. Fazekas, S. Tremblay, and W. Mersereau. 1988. Progesterone receptors regulate gallbladder motility. J Surg Res 45(6): 505-512.
- Medical College of Georgia (MCG), Robert B. Greenblatt, M.D. Library. 2007. Progesterone action. Medical College of Georgia. Retrieved August 2, 2007.
- NIH Clinical Center (NIH). 2007. Historical reference range: Progesterone. National Institutes of Health. Retrieved August 2, 2007.
- Schumacher, M., R. Guennoun, F. Robert, et al. 2004. Local synthesis and dual actions of progesterone in the nervous system: neuroprotection and myelination. Growth Horm IGF Res. 14(Suppl A): S18-33.
| Hormones and endocrine glands - edit |
|---|
|
Hypothalamus: GnRH - TRH - CRH - GHRH - somatostatin - dopamine | Posterior pituitary: vasopressin - oxytocin | Anterior pituitary: GH - ACTH - TSH - LH - FSH - prolactin - MSH - endorphins - lipotropin Thyroid: T3 and T4 - calcitonin | Parathyroid: PTH | Adrenal medulla: epinephrine - norepinephrine | Adrenal cortex: aldosterone - cortisol - DHEA | Pancreas: glucagon- insulin - somatostatin | Ovary: estradiol - progesterone - inhibin - activin | Testis: testosterone - AMH - inhibin | Pineal gland: melatonin | Kidney: renin - EPO - calcitriol - prostaglandin | Heart atrium: ANP Stomach: gastrin | Duodenum: CCK - GIP - secretin - motilin - VIP | Ileum: enteroglucagon | Liver: IGF-1 Placenta: hCG - HPL - estrogen - progesterone Adipose tissue: leptin, adiponectin Target-derived NGF, BDNF, NT-3 |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.