Norepinephrine
| Norepinephrine[1] | |
|---|---|
| |
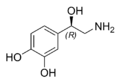
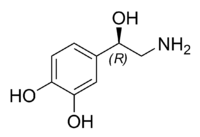
| Chemical name | 4-(2-Amino-1-hydroxyethyl)benzene-1,2-diol |
| Other names | Norepinephrine Noradrenaline |
| Chemical formula | C8H11NO3 |
| Molecular mass | 169.18 g/mol |
| CAS number | D: [149-95-1] L: [51-41-2] D/L: [138-65-8] |
| Density | ? g/cm3 |
| Melting point | L: 216.5-218 °C (decomp.) D/L: 191 °C (decomp.) |
| SMILES | OC1=CC=C(C(O)CN)C=C1O |
| Disclaimer and references | |
Norepinephrine or noradrenaline is a hormone and a neurotransmitter; it is secreted by the adrenal medulla as a hormone into the blood, and as a neurotransmitter from neurons. It has the chemical formula C8H11NO3.
Along with epinephrine (adrenaline), another hormone secreted by the adrenal medulla, norepinephrine underlies the fight-or-flight response to physical or mental stress, directly increasing heart rate, triggering the release of glucose from energy stores, and increasing skeletal muscle readiness, among other actions.
The harmony in nature is reflected in the complex coordination involved in the activity of norepinephrine. In response to a perceived stressful condition, the adrenal gland is stimulated to produce norepinephrine. This leads to a variety of intricate and coordinated responses in diverse parts of the body to prepare the organism for action. When the perceived emergency is over, the body returns to homeostasis.
Chemistry and overview
Like epinephrine, whose structure it resembles, norepinephrine is a phenethylamine, that is, a naturally occurring amine containing one amino group that is connected to an aromatic ring by a two-carbon chain, -CH2-CH2-.
Like epinephrine, norepinephrine also belongs to the family of compounds called catecholamine, a sympathomimetic monoamine derived from the amino acid tyrosine. Catecholamines are water soluble and are 50 percent bound to plasma proteins, so they circulate in the bloodstream. The most abundant catecholamines are epinephrine (adrenaline), norepinephrine (noradrenaline), and dopamine. Catecholamines as hormones are released by the adrenal glands in situations of stress, such as psychological stress or low blood sugar levels (Hoffman 1999).
Norepinephrine is synthesized by a series of enzymatic steps in the adrenal medulla from the amino acid tyrosine:
- The first reaction is the oxidation into dihydroxyphenylalanine (L-DOPA).
- This is followed by decarboxylation into the neurotransmitter dopamine.
- Last is the final β-oxidation into norepinephrine by dopamine beta hydroxylase.
The natural stereoisomer is L-(−)-(R)-norepinephrine. Norepinephrine can be converted to epinephrine.
Norepinephrine and epinephrine are the two main hormones excreted by the adrenal medulla. The adrenal gland, located atop the kidneys, is separated into two distinct structures, the adrenal medulla and the adrenal cortex. The adrenal medulla is at the center of the adrenal gland and is surrounded by the adrenal cortex, with the adrenal medulla taking up about one-quarter of the adrenal gland and the adrenal cortex the remaining three-quarters. Both structures receive regulatory input from the nervous system. The adrenal glands are chiefly responsible for regulating the stress response through the synthesis of corticosteroids and catecholamines (chiefly norepinephrine and epinephrine), including cortisol released in the adrenal cortex. About 20 percent of the total catecholamine release from the adrenal medulla is norepinephrine; the remainder is largely epinephrine (Klabunde 2007).
As a stress hormone, norepinephrine affects parts of the human brain where attention and responding actions are controlled. Norepinephrine is released when a host of physiological changes are activated by a stressful event. This is caused in part by activation of an area of the brain stem called the locus ceruleus. This nucleus is the origin of most norepinephrine pathways in the brain. Neurons that are activated by norepinephrine project bilaterally (send signals to both sides of the brain) from the locus ceruleus along distinct pathways to many locations, including the cerebral cortex, limbic system, and the spinal cord. It causes vasoconstriction (narrowing of the interior channel of blood vessels), increases heart rate, increases blood pressure, facilitates glycogenolysis (coversion of glycogen to glucose), and elevates the blood sugar level.
While epinephrine is mainly released from the adrenal medulla, norepinephrine has another major source—nerve endings.
Norepinephrine also has another major role beyond that of a hormone in that it acts as a neurotransmitter. Neurotransmitters are chemicals that are used to relay, amplify, and modulate electrical signals between a neuron and another cell. Norepinephrine is a neurotransmitter in the central nervous system and sympathetic nervous system where it is released from noradrenergic neurons during synaptic transmission. At synapses, norepinephrine acts on both alpha and beta adrenoreceptors.
In mammals, norepinephrine is rapidly degraded to various metabolites. The principal metabolites are:
- Normetanephrine (via the enzyme catechol-O-methyl transferase, COMT)
- 3,4-Dihydroxymandelic acid (via monoamine oxidase, MAO)
- 3-Methoxy-4-hydroxymandelic acid (via MAO)
- 3-Methoxy-4-hydroxyphenylglycol (via MAO)
- Epinephrine
Clinical uses
Depression
Differences in the norepinephrine system are implicated in depression. Serotonin-norepinephrine reuptake inhibitors (SNRIs) are antidepressants that treat depression by increasing the amount of serotonin and norepinephrine available to postsynaptic cells in the brain. There is some recent evidence showing that the norepinephrine transporter also transports some dopamine as well, implying that SNRIs may also increase dopamine transmission. This is because SNRIs work by inhibiting reuptake, i.e., preventing the serotonin and norepinephrine transporters from taking their respective neurotransmitters back to their storage vesicles for later use. If the norepinephrine transporter normally recycles some dopamine too, then SNRIs will also enhance dopaminergic transmission. Therefore, the antidepressant effects associated with increasing norepinephrine levels may also be partly or largely due to the concurrent increase in dopamine (particularly in the prefrontal cortex).
Tricyclic antidepressants (TCAs) increase norepinephrine as well. Most of them also increase serotonin, but tend to have a lot of side effects due to actions on receptors for histamine and acetylcholine. These include tiredness, increased hunger, dry mouth, and blurred vision. For this reason, they have largely been replaced by newer selective reuptake drugs.
Vasoconstriction
Norepinephrine is used as a vasopressor medication (for example, brand name Levophed) for patients with critical hypotension. A vasopressor is any substance that acts to cause vasoconstriction (narrowing of the central space of blood vessels) and usually results in an increase of the blood pressure. Norepinephrine is given intravenously and acts on both alpha-1 and alpha-2 adrenergic receptors to cause vasoconstriction. Its effect in-vitro is often limited to the increasing of blood pressure through antagonising alpha-1 and alpha-2 receptors and causing a resultant increase in systemic vascular resistance.
In high doseage, and especially when it is combined with other vasopressors, norepinephrine can lead to limb ischemia and limb death. Norepinephrine is mainly used to treat patients in vasodilatory shock states such as septic shock (decreased tissue perfusion and oxygen delivery as a result of infection and sepsis) and neurogenic shock (shock caused by the sudden loss of the sympathetic nervous system signals to the smooth muscle in vessel walls) and has shown a survival benefit over dopamine.
Attention-deficit/hyperactivity disorder
Norepinephrine, along with dopamine, has come to be recognized as playing a large role in attention and focus. For people with Attention-Deficit/Hyperactivity Disorder ADD/ADHD, psychostimulant medications such as Ritalin/Concerta (methylphenidate), Dexedrine (dextroamphetamine), and Adderall (a mixture of dextroamphetamine and racemic amphetamine salts) are prescribed to help increase levels of norepinephrine and dopamine.
Strattera (atomoxetine) is a selective norepinephrine reuptake inhibitor, and is a unique ADD/ADHD medication, as it affects only norepinephrine, rather than dopamine. As a result, Strattera has a lower abuse potential. However, it may not be as effective as the psychostimulants are with many people who have ADD/ADHD. Consulting with a physician or nurse practitioner is needed to find the appropriate medication and dosage.
It should be noted that some individuals have demonstrated an ability to control ADD/ADHD over time without the use of medication.
Notes
- ↑ Merck Index, 11th Edition, 6612.
ReferencesISBN links support NWE through referral fees
- Bennett, M. 1999. "One Hundred Years of Adrenaline: The Discovery of Autoreceptors." Clin Auton Res 9(3): 145-159.
- Boron, W. F., and Boulpaep, E. L. 2005. Medical Physiology: A Cellular and Molecular Approach. Philadelphia, PA: Elsevier/Saunders. ISBN 1416023283.
- Hoffman, R. 1999. Hypoglycemia. Conscious Enlightenment Publishing, Chicago Conscious Choice. Retrieved April 13, 2007.
- Klabune, R. E. 2007. Circulating catecholamines. Cardiovascular Physiology Concepts. Retrieved April 14, 2007.
| Hormones and endocrine glands - edit |
|---|
|
Hypothalamus: GnRH - TRH - CRH - GHRH - somatostatin - dopamine | Posterior pituitary: vasopressin - oxytocin | Anterior pituitary: GH - ACTH - TSH - LH - FSH - prolactin - MSH - endorphins - lipotropin Thyroid: T3 and T4 - calcitonin | Parathyroid: PTH | Adrenal medulla: epinephrine - norepinephrine | Adrenal cortex: aldosterone - cortisol - DHEA | Pancreas: glucagon- insulin - somatostatin | Ovary: estradiol - progesterone - inhibin - activin | Testis: testosterone - AMH - inhibin | Pineal gland: melatonin | Kidney: renin - EPO - calcitriol - prostaglandin | Heart atrium: ANP Stomach: gastrin | Duodenum: CCK - GIP - secretin - motilin - VIP | Ileum: enteroglucagon | Liver: IGF-1 Placenta: hCG - HPL - estrogen - progesterone Adipose tissue: leptin, adiponectin Target-derived NGF, BDNF, NT-3 |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.