Difference between revisions of "Riboflavin" - New World Encyclopedia
Rick Swarts (talk | contribs) |
Rick Swarts (talk | contribs) |
||
| Line 25: | Line 25: | ||
}} | }} | ||
}} | }} | ||
| + | '''Riboflavin''', also known as '''vitamin B<sub>2</sub>''', is a water-soluble organic compound in the [[vitamin B|vitamin B complex]] that is required for a number of [[metabolism|metabolic process]]es in living organisms. While [[plant]]s and most [[microorganism]]s are able to synthesize riboflavin, animals must acquire this essential nutrient in their diet. In most human diets, it is obtained from such common sources as [[milk]], leafy vegetables, whole [[grain]]s, [[liver]], egg white, [[cheese]], and fresh meat. | ||
| − | + | Riboflavin is an easily absorbed [[micronutrient]] with a key role in maintaining [[health]] in humans and animals. It is the central component of the [[cofactor (biochemistry)|cofactor]]s [[FAD]] and [[Flavin mononucleotide|FMN]] and is therefore required by all [[flavoprotein]]s. As such, vitamin B<sub>2</sub> is required for a wide variety of cellular processes. Like the other B vitamins, it plays a key role in energy metabolism, and is required for the [[metabolism]] of [[fat]]s, [[ketone bodies]], [[carbohydrate]]s, and [[protein]]s. It is involved in the support of the [[immune system|immune]] and [[nervous system]]s, formation of [[red blood cell]]s, cell reproduction, and activation of [[folic acid]] (vitamin B<sub>9</sub>) and pyridoxine (vitamin B<sub>6</sub>). | |
| − | + | ==Overview and structure== | |
| + | [[Vitamin]]s, such as riboflavin, are organic nutrients that are obtained through the diet and are essential in small amounts for normal [[metabolism|metabolic]] reactions in humans. Riboflavin is part of the [[vitamin B]] complex, a group of eight, chemically distinct, water-soluble vitamins that were once considered a single vitamin (like [[vitamin C]]), but now are seen as a complex of vitamins that have loosely similar properties and generally are found in the same foods. | ||
| + | |||
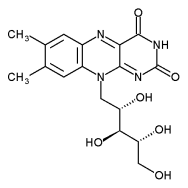
| + | Riboflavin has the chemical formula C<sub>17</sub>H<sub>20</sub>N<sub>4</sub>O<sub>6</sub>. It is an orange-yellow compound with that has a three-ring structure. It can be quickly broken down by heat and upon exposure to light it is converted to lumiflavin, a compound that destroys vitamin C. | ||
| + | The name "riboflavin" comes from "[[ribose]]" and "[[flavin]]." | ||
| − | + | ==Sources== | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | ==Importance== | |
| − | |||
| − | |||
| Line 84: | Line 78: | ||
[[Image:Riboflavin powder.jpg|thumb|Riboflavin powder.]] | [[Image:Riboflavin powder.jpg|thumb|Riboflavin powder.]] | ||
[[Image:Riboflavin solution.jpg|thumb|A riboflavin solution.]] | [[Image:Riboflavin solution.jpg|thumb|A riboflavin solution.]] | ||
| + | |||
| + | [[Milk]], [[cheese]], [[leafy green vegetable]]s, [[liver]], [[kidney]]s, legumes such as mature [[soybean]]s,<ref name=Brody>{{cite book | ||
| + | | last = Brody | ||
| + | | first = Tom | ||
| + | | title = Nutritional Biochemistry | ||
| + | | publisher = Academic Press | ||
| + | | date = 1999 | ||
| + | | location = San Diego | ||
| + | | isbn = 0-12-134836-9 | ||
| + | | oclc = 162571066 212425693 39699995 51091036}}</ref> [[yeast]] and [[almond]]s{{Fact|date=April 2008}} are good sources of vitamin B<sub>2</sub>, but exposure to light destroys riboflavin. | ||
| + | |||
Riboflavin is yellow or yellow-orange in color and in addition to being used as a [[food coloring]]{{Fact|date=September 2008}} it is also used to fortify some foods{{Fact|date=September 2008}}. It is used in [[baby food]]s, [[breakfast cereal]]s, [[pasta]]s, [[sauce]]s, [[processed cheese]], fruit drinks, vitamin-enriched milk products, some [[energy drink]]s, and is widely used in [[vitamin supplement]]s. | Riboflavin is yellow or yellow-orange in color and in addition to being used as a [[food coloring]]{{Fact|date=September 2008}} it is also used to fortify some foods{{Fact|date=September 2008}}. It is used in [[baby food]]s, [[breakfast cereal]]s, [[pasta]]s, [[sauce]]s, [[processed cheese]], fruit drinks, vitamin-enriched milk products, some [[energy drink]]s, and is widely used in [[vitamin supplement]]s. | ||
| + | |||
| + | |||
| + | |||
| + | |||
Large quantities of riboflavin are often included in multi-vitamins; often, the dose is far more than a normal human can use in a day. The excess is excreted in the urine, causing the urine to be colored bright yellow within a few hours of ingestion of the vitamin. | Large quantities of riboflavin are often included in multi-vitamins; often, the dose is far more than a normal human can use in a day. The excess is excreted in the urine, causing the urine to be colored bright yellow within a few hours of ingestion of the vitamin. | ||
Revision as of 21:30, 3 December 2008
| Riboflavin | |
|---|---|
| |
| General | |
| Systematic name | ? |
| Other names | ? |
| Molecular formula | ? |
| SMILES | ? |
| Molar mass | ?.?? g/mol |
| Appearance | ? |
| CAS number | [?-?-?] |
| Properties | |
| Density and phase | ? g/cm³, ? |
| Solubility in water | ? g/100 ml (?°C) |
| Melting point | ?°C (? K) |
| Boiling point | ?°C (? K) |
| Acidity (pKa) | ? |
| Basicity (pKb) | ? |
| Chiral rotation [α]D | ?° |
| Viscosity | ? cP at ?°C |
| Structure | |
| Molecular shape | ? |
| Coordination geometry |
? |
| Crystal structure | ? |
| Dipole moment | ? D |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | ? |
| NFPA 704 | |
| Flash point | ?°C |
| R/S statement | R: ? S: ? |
| RTECS number | ? |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other anions | ? |
| Other cations | ? |
| Related ? | ? |
| Related compounds | ? |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Riboflavin, also known as vitamin B2, is a water-soluble organic compound in the vitamin B complex that is required for a number of metabolic processes in living organisms. While plants and most microorganisms are able to synthesize riboflavin, animals must acquire this essential nutrient in their diet. In most human diets, it is obtained from such common sources as milk, leafy vegetables, whole grains, liver, egg white, cheese, and fresh meat.
Riboflavin is an easily absorbed micronutrient with a key role in maintaining health in humans and animals. It is the central component of the cofactors FAD and FMN and is therefore required by all flavoproteins. As such, vitamin B2 is required for a wide variety of cellular processes. Like the other B vitamins, it plays a key role in energy metabolism, and is required for the metabolism of fats, ketone bodies, carbohydrates, and proteins. It is involved in the support of the immune and nervous systems, formation of red blood cells, cell reproduction, and activation of folic acid (vitamin B9) and pyridoxine (vitamin B6).
Overview and structure
Vitamins, such as riboflavin, are organic nutrients that are obtained through the diet and are essential in small amounts for normal metabolic reactions in humans. Riboflavin is part of the vitamin B complex, a group of eight, chemically distinct, water-soluble vitamins that were once considered a single vitamin (like vitamin C), but now are seen as a complex of vitamins that have loosely similar properties and generally are found in the same foods.
Riboflavin has the chemical formula C17H20N4O6. It is an orange-yellow compound with that has a three-ring structure. It can be quickly broken down by heat and upon exposure to light it is converted to lumiflavin, a compound that destroys vitamin C.
The name "riboflavin" comes from "ribose" and "flavin."
Sources
Importance
Toxicity
Riboflavin is not toxic when taken orally, as its low solubility keeps it from being absorbed in dangerous amounts from the gut [1]. Although toxic doses can be administered by injection[1], any excess at nutritionally relevant doses is excreted in the urine[2], imparting a bright yellow color when in large quantities.
Industrial synthesis
Various biotechnological processes have been developed for industrial scale riboflavin biosynthesis using different microorganisms, including filamentous fungi such as Ashbya gossypii, Candida famata and Candida flaveri as well as the bacteria Corynebacterium ammoniagenes and Bacillus subtilis[3]. The latter organism has been genetically modified to both increase the bacteria's production of riboflavin and to introduce an antibiotic (ampicillin) resistance marker, and is now successfully employed at a commercial scale to produce riboflavin for feed and food fortification purposes. The chemical company BASF has installed a plant in South Korea, which is specialized on riboflavin production using Ashbya gossypii. The concentrations of riboflavin in their modified strain are so high, that the mycelium has a reddish / brownish color and accumulates riboflavin crystals in the vacuoles, which will eventually burst the mycelium.
Riboflavin in food
Milk, cheese, leafy green vegetables, liver, kidneys, legumes such as mature soybeans,[4] yeast and almonds[citation needed] are good sources of vitamin B2, but exposure to light destroys riboflavin.
Riboflavin is yellow or yellow-orange in color and in addition to being used as a food coloring[citation needed] it is also used to fortify some foods[citation needed]. It is used in baby foods, breakfast cereals, pastas, sauces, processed cheese, fruit drinks, vitamin-enriched milk products, some energy drinks, and is widely used in vitamin supplements.
Large quantities of riboflavin are often included in multi-vitamins; often, the dose is far more than a normal human can use in a day. The excess is excreted in the urine, causing the urine to be colored bright yellow within a few hours of ingestion of the vitamin.
It is difficult to incorporate riboflavin into many liquid products because it has poor solubility in water. Hence the requirement for riboflavin-5'-phosphate (E101a), a more expensive but more soluble form of riboflavin.
Nutrition
Riboflavin deficiency
- Further information: Ariboflavinosis
Riboflavin is continuously excreted in the urine of healthy individuals[4], making deficiency relatively common when dietary intake is insufficient. However, riboflavin deficiency is always accompanied by deficiency of other vitamins[4].
A deficiency of riboflavin can be primary - poor vitamin sources in one's daily diet - or secondary, which may be a result of conditions that affect absorption in the intestine, the body not being able to use the vitamin, or an increase in the excretion of the vitamin from the body.
In humans, signs and symptoms of riboflavin deficiency (ariboflavinosis) include cracked and red lips, inflammation of the lining of mouth and tongue, mouth ulcers, cracks at the corners of the mouth (angular cheilitis), and a sore throat. A deficiency may also cause dry and scaling skin, fluid in the mucous membranes, and iron-deficiency anemia. The eyes may also become bloodshot, itchy, watery and sensitive to bright light.
Riboflavin deficiency is classically associated with the oral-ocular-genital syndrome. Angular cheilitis, photophobia, and scrotal dermatitis are the classic remembered signs.
In animals, riboflavin deficiency results in lack of growth, failure to thrive, and eventual death. Experimental riboflavin deficiency in dogs results in growth failure, weakness, ataxia, and inability to stand. The animals collapse, become comatose, and die. During the deficiency state, dermatitis develops together with hair-loss. Other signs include corneal opacity, lenticular cataracts, hemorrhagic adrenals, fatty degeneration of the kidney and liver, and inflammation of the mucus membrane of the gastrointestinal tract. Post-mortem studies in rhesus monkeys fed a riboflavin-deficient diet revealed that about one-third the normal amount of riboflavin was present in the liver, which is the main storage organ for riboflavin in mammals. These overt clinical signs of riboflavin deficiency are rarely seen among inhabitants of the developed countries. However, about 28 million Americans exhibit a common ‘sub-clinical’ stage[citation needed], characterized by a change in biochemical indices (e.g. reduced plasma erythrocyte glutathione reductase levels). Although the effects of long-term sub-clinical riboflavin deficiency are unknown, in children this deficiency results in reduced growth. Subclinical riboflavin deficiency has also been observed in women taking oral contraceptives, in the elderly, in people with eating disorders, and in disease states such as HIV, inflammatory bowel disease, diabetes and chronic heart disease. The fact that riboflavin deficiency does not immediately lead to gross clinical manifestations indicates that the systemic levels of this essential vitamin are tightly regulated.
Diagnostic Testing of B2 Deficiency
A positive diagnostic test for measuring levels of riboflavin in serum is ascertained by measuring erythrocyte levels of glutathione reductase. A deficiency in riboflavin is indicated by changes in the color of the tongue, known as Magenta Tongue.[citation needed]
Clinical Uses
Riboflavin has been used in several clinical and therapeutic situations. For over 30 years, riboflavin supplements have been used as part of the phototherapy treatment of neonatal jaundice. The light used to irradiate the infants breaks down not only the toxin causing the jaundice, but the naturally occurring riboflavin within the infant's blood as well.
More recently there has been growing evidence that supplemental riboflavin may be a useful additive along with beta-blockers in the treatment of migraine headaches. [5]
Development is underway to use riboflavin to improve the safety of transfused blood by reducing pathogens found in collected blood. Riboflavin attaches itself to the nucleic acids (DNA and RNA) in cells, and when light is applied, the nucleic acids are broken, effectively killing those cells. The technology has been shown to be effective for inactivating pathogens in all three major blood components: (platelets, red blood cells, and plasma). It has been shown to inactivate a broad spectrum of pathogens, including known and emerging viruses, bacteria, and parasites.
Recently riboflavin has been used in a new treatment to slow or stop the progression of the corneal disorder keratoconus. This is called corneal collagen crosslinking (CXL). In corneal crosslinking, riboflavin drops are applied to the patient’s corneal surface. Once the riboflavin has penetrated through the cornea, Ultraviolet A light therapy is applied. This induces collagen crosslinking, which increases the tensile strength of the cornea. The treatment has been shown in several studies to stabilize keratoconus.
Industrial Uses
Because riboflavin is fluorescent under UV light, dilute solutions (0.015-0.025% w/w) are often used to detect leaks or to demonstrate coverage in an industrial system such a chemical blend tank or bioreactor. (See the ASME BPE section on Testing and Inspection for additional details.)
Good sources
Riboflavin is found naturally in asparagus, bananas, okra, chard, cottage cheese, milk, yogurt, meat, eggs and fish, each of which contain at least 0.1 mg of the vitamin per 3-10.5 oz (85-300 g) serving.[citation needed]
See also
- Ariboflavinosis (riboflavin deficiency)
- Flavin
- Riboflavin synthase
- Riboflavin kinase
ReferencesISBN links support NWE through referral fees
- ↑ 1.0 1.1 Unna, Klaus and Greslin, Joseph G. (1942). Studies on the toxicity and pharmacology of riboflavin. J Pharmacol Exp Ther 76 (1): 75–80.
- ↑ Zempleni, J and Galloway, JR and McCormick, DB (1996). Pharmacokinetics of orally and intravenously administered riboflavin in healthy humans. Am J Clin Nutr 63 (1): 54–66.
- ↑ Stahmann KP, Revuelta JL and Seulberger H. (2000). Three biotechnical processes using Ashbya gossypii, Candida famata, or Bacillus subtilis compete with chemical riboflavin production. Appl Microbiol Biotechnol 53 (5): 509–516.
- ↑ 4.0 4.1 4.2 Brody, Tom (1999). Nutritional Biochemistry. San Diego: Academic Press. ISBN 0-12-134836-9. OCLC 162571066 212425693 39699995 51091036.
- ↑ Sándor PS, Afra J, Ambrosini A, Schoenen J. Prophylactic treatment of migraine with beta-blockers and riboflavin: differential effects on the intensity dependence of auditory evoked cortical potentials. Headache. 2000 Jan;40(1):30-5.
External links
- Jane Higdon, "Riboflavin", Micronutrient Information Center, Linus Pauling Institute
- Mirasol PRT includes a brief description of riboflavin as an agent to inactivate pathogens.
| Vitamins |
|---|
| All B vitamins | All D vitamins |
| Retinol (A) | Thiamine (B1) | Riboflavin (B2) | Niacin (B3) | Pantothenic acid (B5) | Pyridoxine (B6) | Biotin (B7) | Folic acid (B9) | Cyanocobalamin (B12) | Ascorbic acid (C) | Ergocalciferol (D2) | Cholecalciferol (D3) | Tocopherol (E) | Naphthoquinone (K) |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.

