Difference between revisions of "Alpha decay" - New World Encyclopedia
| Line 3: | Line 3: | ||
[[Image:Alpha Decay.svg|thumb|left|Alpha decay.]] | [[Image:Alpha Decay.svg|thumb|left|Alpha decay.]] | ||
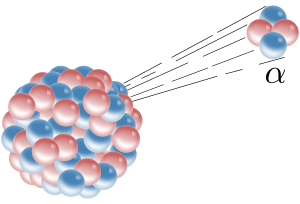
| − | + | '''Alpha decay''' is a type of [[radioactive decay]] in which an [[atomic nucleus]] emits an '''alpha particle'''. An alpha particle (or α particle, named after the first letter of the [[Greek alphabet]]) consists of two [[proton]]s and two [[neutron]]s bound together. It is identical to the nucleus of a [[helium]] atom and may therefore be written as He<sup>2+</sup> or <sup>4</sup><sub>2</sub>He. | |
| + | |||
| + | When an atomic nucleus releases an alpha particle, the atom "decays" (is transformed) into another atom with a [[atomic weight|mass number]] that is lower by 4 and an [[atomic number]] that is lower by 2. Alpha decay is a form of [[nuclear fission]] where the parent atom splits into two daughter products. | ||
| + | |||
| + | For example, alpha decay of radium atoms converts them to [[radon]] atoms, released as a gas. Also, most of the [[helium]] produced on [[Earth]] comes from the alpha decay of underground deposits of [[mineral]]s containing [[uranium]] or [[thorium]]. The helium is brought to the surface as a by-product of [[natural gas]] production. | ||
| + | |||
| − | |||
For example: | For example: | ||
:<math> | :<math> | ||
| Line 16: | Line 20: | ||
(The second form is preferred because the first form appears electrically unbalanced. Fundamentally, the recoiling nucleus is very quickly stripped of two electrons to neutralize the ionized helium cation.) | (The second form is preferred because the first form appears electrically unbalanced. Fundamentally, the recoiling nucleus is very quickly stripped of two electrons to neutralize the ionized helium cation.) | ||
| − | + | ||
| − | + | == Properties of alpha particles == | |
| + | |||
| + | Alpha particles are a highly [[ionizing radiation|ionizing]] form of [[particle radiation]], but they have low penetration. They are readily stopped by a sheet of paper. | ||
Alpha particles have a typical kinetic energy of 5 MeV (that is ≈0.13% of their total energy, i.e. 110 TJ/kg) and a speed of 15,000 km/s. This corresponds to a speed of around 0.05[[Speed of light|c]]. Because of their relatively large mass, +2 charge and relatively low velocity, they are very likely to interact with other atoms and lose their energy, so they are effectively absorbed within a few centimeters of air. | Alpha particles have a typical kinetic energy of 5 MeV (that is ≈0.13% of their total energy, i.e. 110 TJ/kg) and a speed of 15,000 km/s. This corresponds to a speed of around 0.05[[Speed of light|c]]. Because of their relatively large mass, +2 charge and relatively low velocity, they are very likely to interact with other atoms and lose their energy, so they are effectively absorbed within a few centimeters of air. | ||
| − | |||
| − | + | [[Image:Alphaspectrometer.jpg|thumb|left|Alpha source beneath a radiation detector.]] | |
| + | |||
| + | == Quantum tunneling effect == | ||
| − | + | In the classical view, escape of an alpha particle from an atomic nucleus is forbidden because the alpha particle is trapped in a "potential well" (energy minimum). By 1928, [[George Gamow]] solved the mystery of alpha decay by the theory known as "[[quantum tunneling]]." Applying the principles of [[quantum mechanics]], Gamow showed that an alpha particle has a tiny (but non-zero) probability of "tunneling" through the energy barrier and escaping from the nucleus. | |
| − | + | Unlike [[beta decay]], alpha decay is governed by the [[strong nuclear force]], which holds protons and neutrons together. | |
==Uses== | ==Uses== | ||
| − | [[americium#Isotopes|Americium-241]] is used in [[Smoke detector|smoke | + | The isotope [[americium#Isotopes|Americium-241]] emits alpha particles, and this property is used in some types of [[Smoke detector|smoke detector]]s. The alpha particles [[Ionization|ionize]] molecules in the air within a narrow gap, producing a small [[Electric current|current]]. This current can be easily interrupted by smoke particles. |
Alpha decay can provide a safe power source for [[radioisotope thermoelectric generator]]s used for [[space probe]]s and [[Artificial pacemaker|artificial heart pacemakers]]. Alpha decay is much more easily shielded against than other forms of radioactive decay. [[Plutonium-238]], for example, requires only 2.5 [[Millimetre|mm]] of [[lead]] shielding to protect against unwanted radiation. | Alpha decay can provide a safe power source for [[radioisotope thermoelectric generator]]s used for [[space probe]]s and [[Artificial pacemaker|artificial heart pacemakers]]. Alpha decay is much more easily shielded against than other forms of radioactive decay. [[Plutonium-238]], for example, requires only 2.5 [[Millimetre|mm]] of [[lead]] shielding to protect against unwanted radiation. | ||
| Line 47: | Line 54: | ||
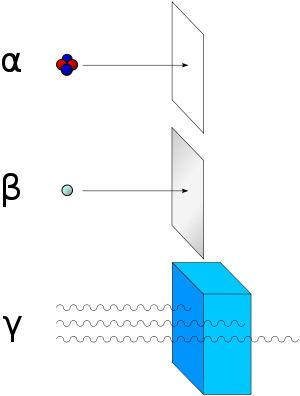
[[Image:Alfa_beta_gamma_radiation.svg|thumb|Alpha radiation consists of [[helium-4]] nuclei and is readily stopped by a sheet of paper. Beta radiation, consisting of [[electron]]s, is halted by an aluminum plate. Gamma radiation is eventually absorbed as it penetrates a dense material.]] | [[Image:Alfa_beta_gamma_radiation.svg|thumb|Alpha radiation consists of [[helium-4]] nuclei and is readily stopped by a sheet of paper. Beta radiation, consisting of [[electron]]s, is halted by an aluminum plate. Gamma radiation is eventually absorbed as it penetrates a dense material.]] | ||
| − | The | + | The alpha particle mass is 6.644656×10<sup>-27</sup> kg, which is equivalent to the energy of 3.72738 [[giga|G]][[electron volt|eV]]. The charge of an alpha particle is equal to +2e, where e is the magnitude of charge on an electron, e=1.602176462x10<sup>-19</sup>C. |
| − | Alpha particles are emitted by [[radioactive]] nuclei such as [[uranium]] or [[radium]] in a process known as | + | Alpha particles are emitted by [[radioactive]] nuclei such as [[uranium]] or [[radium]] in a process known as alpha decay. This sometimes leaves the nucleus in an excited state, with the emission of a [[gamma ray]] removing the excess [[energy]]. In contrast to [[beta decay]], alpha decay is mediated by the [[strong nuclear force]]. Classically, alpha particles do not have enough energy to escape the potential of the nucleus. However, the [[quantum tunneling]] effect allows them to escape. |
When an alpha particle is emitted, the [[atomic mass]] of an element goes down by roughly 4.0015 [[unified atomic mass unit|u]], due to the loss of 2 [[neutrons]] and 2 protons. The [[atomic number]] of the atom goes down by 2, as a result of the loss of 2 protons; the atom becomes a new element. An example of this is when radium becomes [[radon]] gas due to alpha decay. | When an alpha particle is emitted, the [[atomic mass]] of an element goes down by roughly 4.0015 [[unified atomic mass unit|u]], due to the loss of 2 [[neutrons]] and 2 protons. The [[atomic number]] of the atom goes down by 2, as a result of the loss of 2 protons; the atom becomes a new element. An example of this is when radium becomes [[radon]] gas due to alpha decay. | ||
Revision as of 15:03, 20 October 2007
| Nuclear physics | ||||||||||||||
| ||||||||||||||
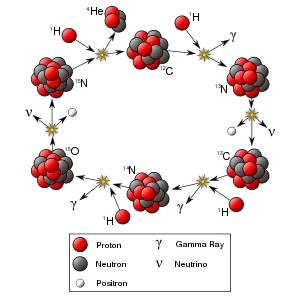
| Radioactive decay Nuclear fission Nuclear fusion
| ||||||||||||||
Alpha decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle. An alpha particle (or α particle, named after the first letter of the Greek alphabet) consists of two protons and two neutrons bound together. It is identical to the nucleus of a helium atom and may therefore be written as He2+ or 42He.
When an atomic nucleus releases an alpha particle, the atom "decays" (is transformed) into another atom with a mass number that is lower by 4 and an atomic number that is lower by 2. Alpha decay is a form of nuclear fission where the parent atom splits into two daughter products.
For example, alpha decay of radium atoms converts them to radon atoms, released as a gas. Also, most of the helium produced on Earth comes from the alpha decay of underground deposits of minerals containing uranium or thorium. The helium is brought to the surface as a by-product of natural gas production.
For example:
although this is typically written as:
(The second form is preferred because the first form appears electrically unbalanced. Fundamentally, the recoiling nucleus is very quickly stripped of two electrons to neutralize the ionized helium cation.)
Properties of alpha particles
Alpha particles are a highly ionizing form of particle radiation, but they have low penetration. They are readily stopped by a sheet of paper.
Alpha particles have a typical kinetic energy of 5 MeV (that is ≈0.13% of their total energy, i.e. 110 TJ/kg) and a speed of 15,000 km/s. This corresponds to a speed of around 0.05c. Because of their relatively large mass, +2 charge and relatively low velocity, they are very likely to interact with other atoms and lose their energy, so they are effectively absorbed within a few centimeters of air.
Quantum tunneling effect
In the classical view, escape of an alpha particle from an atomic nucleus is forbidden because the alpha particle is trapped in a "potential well" (energy minimum). By 1928, George Gamow solved the mystery of alpha decay by the theory known as "quantum tunneling." Applying the principles of quantum mechanics, Gamow showed that an alpha particle has a tiny (but non-zero) probability of "tunneling" through the energy barrier and escaping from the nucleus.
Unlike beta decay, alpha decay is governed by the strong nuclear force, which holds protons and neutrons together.
Uses
The isotope Americium-241 emits alpha particles, and this property is used in some types of smoke detectors. The alpha particles ionize molecules in the air within a narrow gap, producing a small current. This current can be easily interrupted by smoke particles.
Alpha decay can provide a safe power source for radioisotope thermoelectric generators used for space probes and artificial heart pacemakers. Alpha decay is much more easily shielded against than other forms of radioactive decay. Plutonium-238, for example, requires only 2.5 mm of lead shielding to protect against unwanted radiation.
Toxicity
Because they are heavy and charged, alpha particles tend to have a very short mean free path, and therefore lose their kinetic energy within a short distance of their source. This can result in several MeV being deposited in a relatively small area. If they penetrate live tissue, this can cause significant cellular damage. Generally, external alpha radiation is not harmful because alpha particles are completely absorbed by a few centimeters of air. Even touching an alpha source is usually not harmful; the thin layer of dead skin cells in the outermost layer of the skin will absorb them. However, if a substance radiating alpha particles is ingested, inhaled by, injected into, or introduced through the skin (shrapnel, corrosive chemicals) into an organism it may result in a high dose to that area.
Radon is a naturally occurring, radioactive gas found in soil, rock, and sometimes groundwater. When radon gas is inhaled, some of the radon particles stick to the inner lining of the lung. The particles that remain continue to decay over time, emitting alpha particles which may damage cells in the lung tissue.[1]. The death of Marie Curie at age 66 from leukemia was likely caused by prolonged exposure to high doses of ionizing radiation. Curie worked extensively with Radium, which decays into Radon[2], along with other radioactive materials that emit beta and gamma rays. Shrapnel deposited in the body from depleted uranium poses another such internal risk of alpha particle radiation dose.
The 2006 assassination of Russian dissident Alexander Litvinenko is thought to have been caused by poisoning with Polonium-210, an alpha emitter.
Alpha particle
The alpha particle mass is 6.644656×10-27 kg, which is equivalent to the energy of 3.72738 GeV. The charge of an alpha particle is equal to +2e, where e is the magnitude of charge on an electron, e=1.602176462x10-19C.
Alpha particles are emitted by radioactive nuclei such as uranium or radium in a process known as alpha decay. This sometimes leaves the nucleus in an excited state, with the emission of a gamma ray removing the excess energy. In contrast to beta decay, alpha decay is mediated by the strong nuclear force. Classically, alpha particles do not have enough energy to escape the potential of the nucleus. However, the quantum tunneling effect allows them to escape.
When an alpha particle is emitted, the atomic mass of an element goes down by roughly 4.0015 u, due to the loss of 2 neutrons and 2 protons. The atomic number of the atom goes down by 2, as a result of the loss of 2 protons; the atom becomes a new element. An example of this is when radium becomes radon gas due to alpha decay.
The energy of alpha particles varies, with higher energy alpha particles being emitted from larger nuclei, but most alpha particles have energies of between 3 and 7 MeV. This is a substantial amount of energy for a single particle, but their high mass means alpha particles have a lower speed (with a typical kinetic energy of 5 MeV the speed is 15,000 km/s) than any other common type of radiation (β particles, γ rays, neutrons, etc). Because of their charge and large mass, alpha particles are easily absorbed by materials and can travel only a few centimeters in air. They can be absorbed by tissue paper or the outer layers of human skin (about 40 micrometres, equivalent to a few cells deep) and so are not generally dangerous to life unless the source is ingested or inhaled. Because of this high mass and strong absorption, however, if alpha radiation does enter the body (most often because radioactive material has been inhaled or ingested), it is the most destructive form of ionizing radiation. It is the most strongly ionizing, and with large enough doses can cause any or all of the symptoms of radiation poisoning. It is estimated that chromosome damage from alpha particles is about 100 times greater than that caused by an equivalent amount of other radiation. The alpha emitter polonium-210 is suspected of playing a role in lung and bladder cancer related to tobacco smoking.
Most smoke detectors contain a small amount of the alpha emitter americium-241. This isotope is extremely dangerous if inhaled or ingested, but the danger is minimal if the source is kept sealed. Many municipalities have established programs to collect and dispose of old smoke detectors, rather than let them go into the general waste stream.
Because alpha particles occur naturally, but can have energy high enough to participate in a nuclear reaction, study of them led to much early knowledge of nuclear physics. The physicist Ernest Rutherford famously used alpha particles to infer that J. J. Thomson's Plum pudding model of the atom was fundamentally flawed. Rutherford's famous gold foil experiment was conducted by his students Hans Geiger and Ernest Marsden. A narrow beam of alpha particles was set up, passing through very thin (only a few hundred atoms thick) gold foil. The alpha particles were detected by a zinc sulfide screen, which emits a flash of light upon an alpha particle collision. Rutherford hypothesisized that, assuming the "plum pudding" model of the atom was correct, the positively charged alpha particles would be only slightly deflected, if at all, by the dispersed positive charge predicted. It was found that some of the alpha particles were deflected at much larger angles than expected, with some even bouncing back. Although most of the alpha particles went straight through as expected, Rutherford commented that the few particles that were deflected was akin to shooting a fifteen inch shell at tissue paper only to have it bounce off, again assuming the "plum pudding" theory was correct. It was soon determined that the positive charge of the atom was concentrated in a small area in the center of the atom, hence making the positive charge dense enough to deflect any positively charged alpha particles that happened to come close to what was later termed the nucleus (it was not known at the time that alpha particles were themselves nuclei nor was the existence of protons or neutrons known). Rutherford's experiment subsequently led to the Bohr model and later the modern wave-mechanical model of the atom.
Rutherford's work also improved on previous measurements of the ratio of an alpha particle's mass to charge, allowing him to deduce that alpha particles were helium nuclei.[3]
In computer technology, DRAM 'soft errors' were linked to alpha particles in 1978 in Intel's DRAM chips. The discovery led to strict control of radioactive elements in the packaging of semiconductor materials, and the problem was largely considered 'solved'.
See also
Notes
ReferencesISBN links support NWE through referral fees
- Krane, Kenneth S., and David Halliday. 1988. Introductory Nuclear Physics. New York: Wiley. ISBN 047180553X.
- Martin, Brian. 2006. Nuclear and Particle Physics: An Introduction. Hoboken, NJ: Wiley. ISBN 0470025328.
- Poenaru, D. N. 1996. Nuclear Decay Modes. Fundamental and Applied Nuclear Physics Series. Philadelphia: Institute of Physics. ISBN 0750303387.
- Seiden, Abraham. 2004. Particle Physics: A Comprehensive Introduction. San Francisco, CA: Addison Wesley. ISBN 0805387366.
- Tipler, Paul and Ralph Llewellyn. 2002. Modern Physics. 4th ed. New York, NY: W.H. Freeman. ISBN 0-7167-4345-0.
- Turner, James E. 1995. Atoms, Radiation, and Radiation Protection. 2nd ed. New York: Wiley. ISBN 0471595810.
| |||||||||||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.