Galactose
| Galactose | |
|---|---|
|

|
| Identifiers | |
| CAS number | [] |
| PubChem | |
| MeSH | |
| Properties | |
| Molecular formula | C6H12O6 |
| Molar mass | 180.08 |
| Melting point |
167 degrees C |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Galactose (Gal) is a six-carbon sugar and a monosaccharide that combines with glucose to form the disaccharide lactose, forms galactose polysaccharides in agar, is a component in various gums, mucilages, and pectins, and is also present in sugar beets. As a hexose sugar, galactose has the same formula as glucose, C6H12O6, but differs in the position of the hydroxyl group on carbon-4 (Bender and Bender 2005). Along with glucose and fructose, galactose is one of the three most important blood sugars in animals. Galactose also is known as brain sugar (Houghton Mifflin 1998).
Galactose is less sweet than glucose and sucrose. It is considered a nutritive sweetener because it has food energy. Galactose and glucose both play important roles in joining with lipids to form glycolipids and joining with proteins to form glycoproteins. Galactolipids are a major component of the membrane tissues of plants, while galactose joined in forming the more complex galactocerebrosides acts as an important component of membrane tissues in animal muscles and nerves.
Under normal conditions, the intricate coordination in the human body is remarkable, with lactose in food being broken down, via enzymes, into glucose and galactose, and these sugars being further metabolized. However, in rare cases (one per 62,000 births), this harmonious system is compromised by a genetic disorder, affecting an individual's ability to properly metabolize the sugar galactose. This leads to toxic levels of galactose building up in the blood, requiring the elimination of lactose and galactose from the diet. This condition is known as galactosemia.
Description
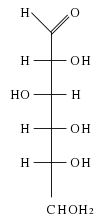
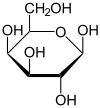
Galactose is a six-carbon monosaccharide. As a carbohydrate, galactose belongs to that class of biological molecules that contain primarily carbon (C) atoms flanked by hydrogen (H) atoms and hydroxyl (OH) groups (H-C-OH). As a hexose carbohydrate, galactose has the same molecular formula as glucose and fructose (C6H12O6) but a different atomic arrangement. It differs from glucose only in the position of the hydroxyl group on carbon-4 (Bender and Bender 2005). (The carbons of glucose are numbered beginning with the more oxidized end of the molecule, the carbonyl group.) In other words, galactose is an epimer of glucose, having a different configuration at only one of several stereogenic centers.
In galactose, the first and last -OH groups point the same way and the second and third -OH groups point the other way.
Galactose can be found in two enantiomers, L-galactose and D-galactose. Enantiomers are two stereoisomers that are related to each other by a reflection: They are mirror images of each other. Every stereocenter in one has the opposite configuration in the other. D-Galactose has the same configuration at its penultimate carbon as D-glyceraldehyde.
Galactose is less sweet than glucose and about one-third as sweet as sucrose (Bender and Bender 2005). It also is less soluble in water than glucose.
Sources and relationship to lactose
D-galactose is found in such sources as lactose (milk sugar), agar, gum arabic, sugar beets, seaweed, and nerve cell membranes. L-galactose is found in flaxseed mucilage, snail galactogen, agar, and other such sources. Often the two forms are found together, producing DL-galactose.
Galactose also is synthesized by the body, where it forms part of glycolipids and glycoproteins in several tissues. Galactose is an important component of cerebrosides, which are glycosphingolipids that are important components in animal muscle and nerve cell membranes. Myelin is the most well known cerebroside. Cerebrosides consist of a ceramide with a single sugar residue at the 1-hydroxyl moiety. The sugar residue can be either glucose or galactose; the two major types are therefore called glucocerebrosides and galactocerebrosides. Galactocerebrosides are typically found in neural tissue, while glucocerebrosides are found in other tissues.
Galactan is a polymer of the sugar galactose. It is found in hemicellulose and can be converted to galactose by hydrolysis.
Lactose is a main dietary source of galactose for humans. Lactose is a disaccharide that consists of β-D-galactose and β-D-glucose fragments bonded through a β1-4 glycosidic linkage. The hydrolysis of lactose to glucose and galactose is catalyzed by the enzyme lactase, a β-galactosidase. In the human body, glucose is changed into galactose in order to enable the mammary glands to secrete lactose. The β-galactosidase enzyme is produced by the lac operon in Escherichia coli (E. coli).
Liver galactose metabolism
Galactose in the bloodstream travels to the liver. In the liver, galactose (Gal) is converted to glucose-6-phosphate (Glc-6-P) in the following reactions:
galacto- uridyl phosphogluco-
kinase transferase mutase
gal --------> gal 1 P ------------------> glc 1 P -----------> glc 6 P
^ \
/ v
UDP-glc UDP-gal
^ /
\___________/
epimerase
Enzymes that catalyze the reactions are in italic. Mutations that disable either the kinase, the transferase, or the epimerase block galactose metabolism, producing galactosemia. The cyclic reaction at the center of the diagram shows that uridyl-transferase catalyzes Gal-1-P and UDP-Glc (uracil-diphosphate-glucose) to react in such a way that the Gal displaces the Glc from its bond with UDP yielding UDP-Gal and Glc-1-P. (Biocarta)
Clinical significance
Some studies have suggested a possible link between galactose in milk and ovarian cancer (Cramer 1989; Cramer et al. 1989). Other studies show no correlation, even in the presence of defective galactose metabolism (Goodman et al. 2002; Fung et al. 2003). More recently, pooled analysis done by the Harvard School of Public Health showed no specific correlation between lactose containing foods and ovarian cancer, and showed statistically insignificant increases in risk for consumption of lactose at ≥30 grams/day (Genkinger et al. 2006). More research is necessary to ascertain possible risks.
Some ongoing studies suggest that galactose may have a role in treatment of focal segmental glomerulosclerosis (a kidney disease resulting in kidney failure and proteinuria). This effect is likely to be a result of binding of galactose to FSGS factor.
Metabolic disorders
There are some important disorders involving galactose.
Galactosemia is a rare genetic metabolic disorder that affects an individual's ability to properly metabolize the sugar galactose. Goppert first described the disease in 1917 (Goppert 1917), with its cause as a defect in galactose metabolism being identified by a group led by Herman Kalckar in 1956 (Isselbacher et al. 1956). Incidence of the most common or classic type of galactosemia is about one per 62,000 births (Merck).
ReferencesISBN links support NWE through referral fees
- Bender, D. A., and A. E. Bender. 2005. A Dictionary of Food and Nutrition. New York: Oxford University Press. ISBN 0198609612.
- Biocarta. Leloir pathway of galactose metabolism Charting pathways of life. Retrieved August 4, 2008.
- Cramer, D. 1989. Lactase persistence and milk consumption as determinants of ovarian cancer risk Am J Epidemiol 130(5): 904-10. Retrieved May 11, 2008.
- Cramer, D., B. Harlow, W. Willett, W. Welch, D. Bell, R. Scully, W. Ng, and R. Knapp. 1989. Galactose consumption and metabolism in relation to the risk of ovarian cancer Lancet 2(8654): 66-71. Retrieved May 11, 2008.
- Fung, W. L. A., H. Risch, J. McLaughlin, B. Rosen, D. Cole, D. Vesprini, and S. A. Narod. 2003. The N314D polymorphism of galactose-1-phosphate uridyl transferase does not modify the risk of ovarian cancer Cancer Epidemiol Biomarkers Prev 12(7): 678-80. Retrieved May 11, 2008.
- Genkinger, J. M., D. J. Hunter, D. Spiegelman, K. E. Anderson, A. Arslan, W. L. Beeson, J. E. Buring, G. E. Fraser, J. L. Freudenheim, R. A. Goldbohm, S. E. Hankinson, D. R. Jacobs, A. Koushik, J. V. Lacey, S. C. Larsson, M. Leitzmann, M. L. McCullough, A. B. Miller, C. Rodriguez, T. E. Rohan, L. J. Schouten, R. Shore, E. Smit, A. Wolk, S. M. Zhang, and S. A. Smith-Warner. 2006. Dairy products and ovarian cancer: A pooled analysis of 12 cohort studies Cancer Epidemiol Biomarkers Prev 15: 364-372. Retrieved May 11, 2008.
- Goodman, M. T., A. H. Wu , K.-H. Tung, K. McDuffie, D. W. Cramer, L. R. Wilkens, K. Terada, J. K. V. Reichardt, and W. G. Ng. 2002. Association of galactose-1-phosphate uridyltransferase activity and N314D genotype with the risk of ovarian cancer Am. J. Epidemiol 156(8): 693-701. Retrieved May 11, 2008.
- Goppert, F. 1917. Galaktosurie nach Milchzuckergabe bei angeborenem, familiaerem chronischem Leberleiden. Klin Wschr 54:473-477.
- Houghton Mifflin Company. 1998. Compact American Medical Dictionary: A Concise and Up-to-date Guide to Medical Terms. Boston, Mass: Houghton Mifflin. ISBN 0395884098.
- Isselbacher, K. J., E. P. Anderson, K. Kurahashi, and H. M. Kalckar. 1956. Congenital galactosemia, a single enzymatic block in galactose metabolism Science 13(123): 635-6. Retrieved May 11, 2008.
- Merck. Carbohydrate metabolism disorders—Galactosemia. Retrieved August 8, 2008.
| |||||||||||||||||||||||||||||||||||||||||||||||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.