Chitin
Chitin is a hard, semitransparent polysaccharide that is found in many places in the natural world, including serving as the main component of arthropod exoskeletons and the cell walls of some fungi.
Chitin, for example, is the main component of the shells of crustaceans, such as crabs, lobsters, and shrimp; and many insects, such as ants and beetles, have a covering made from chitin. Chitin is found in the cell walls of some molds, yeast, and algae. It also comprises parts of the jaws and body spines of certain worms (Nicol 1991).
Chitin is one of the most common organic (carbon-containing) compounds on Earth.
Chitin also has many important medical, industrial, and commercial uses. This reflection of human creativity was partially in response to an environmental problem. For many years, the shells of crustaceans such as crabs and shrimp were discarded during the process of producing seafood, resulting in a problem of pollution of coastal waters (Towle 1989). Spurred by funding of the U.S. Federal Government, research began to develop numerous ways to use the chitin in the shells. Today, chitin is used as a surgical thread, an artificial skin, artificial blood vessels, a protective seed coating against fungus, specialized contact lenses, diapers, and in wastewater purification processes, among other uses (Towle 1989, UDGCMS 2006).
Detailed Description
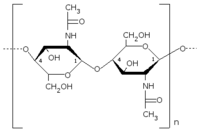
Chitin is a polysaccharide that is constructed from units of N-acetylglucosamine (more completely, N-acetyl-D-glucos-2-amine). These are linked together in β-1,4 fashion in a similar manner to the glucose units which form cellulose, which forms the primary structural component of green plants.
In effect chitin may be described as cellulose with one hydroxyl group on each monomer replaced by an acetylamine group. This allows for increased hydrogen bonding between adjacent polymers, giving the polymer increased strength. Both cellulose and chitin are long unbranched chains of glucose derivatives, and both materials contribute structure and strength, protecting the organisms (plants in the case of cellulose). Cellulose is by far the most abundant organic (carbon-containing) compound on Earth, and chitin is among the top three most abundant such compounds, along with starch.
In its unmodified form, chitin is translucent, pliable, resilient, and quite tough. In arthropods, however, it is frequently modified by being embedded in a hardened proteinaceous matrix, which forms much of the hard exoskeleton. The difference between unmodified and modified chitinous exoskeleton can be seen by comparing the soft body wall of a caterpillar to a beetle's hard body covering.
Chitin is an unusual substance as it is a naturally occurring polymer. Its breakdown is conducted by bacteria that are characterized by special chemical receptors to the sugars produced by the decomposition of chitin. If chitin is detected, they then produce enzymes to digest the chitin by reducing it to simple sugars and ammonia.
Chitin is closely related to chitosan (a more water-soluble derivative of chitin).
History and etymology
In 1821, Henri Braconnot, director of the botanical garden in Nancy, France, noticed that there was a material in mushrooms that did not dissolve in sulfuric acid. He called it fungine (Nicol 1991). In the 1830s, it was isolated in insects, and in 1859, chitosan, a derivative of chitin, was produced (UDGCMS 2006).
The English word "chitin" comes from the French word "chitine," which first appeared in 1836. These words were derived from the Latin word "chitōn," meaning mollusk, which in turn comes from the Greek word khitōn, meaning "tunic" or "frock." The Greek word "khitōn" can be traced to the Central Semitic word "kittan," which is from the Akkadian words "kitû" or "kita’um," meaning flax or linen, and originally the Sumerian word "gada" or "gida".[1]
A similar word, "chiton," refers to a marine animal with a protective shell (also known as a sea cradle).
Uses
Chitin's properties as a tough and strong material make it favorable as surgical thread. Its biodegradibility also means it wears away with time as the wound heals.
Chitin also has some unusual properties in that it accelerates healing in wounds in humans. Therefore, chitin is used as a wound-healing agent. It is also used in development of an artificial skin that the body does not reject (Towle 1989). Other medical uses include as anti-bacterial sponges and hospital dressings, artificial blood vessels, tumor inhibition, dental plaque inhibition, and blood cholesterol control (UDGCMS 2006).
Chitin is used industrially in many different processes. For example, chitin is used in water and wastewater purification. It has a strong positive charge, aiding its binding with negatively charged materials including oils, detergents, and other contaminants suspended in water, and chitosan's metal-binding properties help in removing toxic organic compounds, such as PCBs (UDGCMS 2006).
Chitin is also used to thicken and stabilize foods and pharmaceuticals, and it acts as a binder in dyes, fabrics, and adhesives. Ion-exchange resins can be made from chitin, and industrial separation membranes are made from chitin. Processes to size and strengthen paper also employ chitin. Chitin may also be used to make varnish, a seed coating to protect against fungus, specialized contact lenses, sponges, diapers, feminine napkins, and tampons (Towle 1989, UDGCMS 2006).
Notes
- ↑ Bartelby.com – Definition. Retrieved September 13, 2007.
ReferencesISBN links support NWE through referral fees
- Nicol, S. “Life after death for empty shells: Crustacean fisheries create a mountain of waste shells, made of a strong natural polymer, chitin. Now chemists are helping to put this waste to some surprising uses.” New Scientist. Vol. 1755, February 09, 1991.
- Towle, A. Modern Biology. Austin, TX: Holt, Rinehart and Winston, 1989. ISBN 0-03-013919-8.
- University of Delaware Graduate College of Marine Studies (UDGCMS). Chitin research Horseshoe Crab website, a joint effort of the Mid-Atlantic Sea Grant Programs and the National Oceanic and Atmospheric Administration. 2006. Retrieved September 13, 2007.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.