Difference between revisions of "Phenol" - New World Encyclopedia
(added credit and category tags, deleted foreign language links) |
|||
| Line 1: | Line 1: | ||
| − | + | {{Claimed}} | |
| − | |||
{| class="toccolours" border="1" style="float: right; clear: right; margin: 0 0 1em 1em; border-collapse: collapse;" | {| class="toccolours" border="1" style="float: right; clear: right; margin: 0 0 1em 1em; border-collapse: collapse;" | ||
! {{chembox header}}| '''Phenol''' | ! {{chembox header}}| '''Phenol''' | ||
| Line 11: | Line 10: | ||
|- | |- | ||
| Other names | | Other names | ||
| − | | Carbolic Acid<br/>Benzenol<br/>Phenylic Acid<br/>Hydroxybenzene<br>Phenic acid | + | | Carbolic Acid<br/>Benzenol<br/>Phenylic Acid<br/>Hydroxybenzene<br/>Phenic acid |
|- | |- | ||
| [[Chemical formula]] | | [[Chemical formula]] | ||
| Line 85: | Line 84: | ||
|- | |- | ||
| [[Phenol (data page)#Thermodynamic properties|Thermodynamic<br/>data]] | | [[Phenol (data page)#Thermodynamic properties|Thermodynamic<br/>data]] | ||
| − | | Phase behaviour<br>Solid, liquid, gas | + | | Phase behaviour<br/>Solid, liquid, gas |
|- | |- | ||
| [[Phenol (data page)#Spectral data|Spectral data]] | | [[Phenol (data page)#Spectral data|Spectral data]] | ||
| Line 99: | Line 98: | ||
|} | |} | ||
| − | '''Phenol''', also known under an older name of '''carbolic acid''', is a toxic, | + | '''Phenol''', also known under an older name of '''carbolic acid''', is a toxic, colorless [[crystal]]line [[solid]] with a distinctive sweet tarry [[odor]]. Its [[chemical formula]] is [[Carbon|C]]<sub>6</sub>[[Hydrogen|H]]<sub>5</sub>[[Oxygen|O]]H and its structure is that of a [[hydroxyl]] [[Functional group|group]] (-OH) [[chemical bond|bonded]] to a [[phenyl ring]]; it is thus an [[Simple aromatic ring|aromatic compound]]. |
| − | + | ||
==Phenols== | ==Phenols== | ||
{{main|phenols}} | {{main|phenols}} | ||
| Line 106: | Line 105: | ||
==Properties== | ==Properties== | ||
| − | Phenol has a limited [[solubility]] in water (8.3 g/100 ml). It is slightly acidic: the phenol molecule has weak tendencies to lose the H<sup>+</sup> ion from the hydroxyl group, resulting in the highly water-soluble '''phenoxide''' [[anion]] C<sub>6</sub>H<sub>5</sub>O<sup>−</sup>. Compared to [[aliphatic]] [[alcohol]]s, phenol shows much higher acidity; it even reacts with NaOH to lose H<sup>+</sup> whereas | + | Phenol has a limited [[solubility]] in water (8.3 g/100 ml). It is slightly acidic: the phenol molecule has weak tendencies to lose the H<sup>+</sup> ion from the hydroxyl group, resulting in the highly water-soluble '''phenoxide''' [[anion]] C<sub>6</sub>H<sub>5</sub>O<sup>−</sup>. Compared to [[aliphatic]] [[alcohol]]s, phenol shows much higher acidity; it even reacts with NaOH to lose H<sup>+</sup> whereas aliphatic alcohols do not. Contrary to popular belief, this has little to do with orbital overlap between the oxygen's lone pairs and the aromatic system. |
| − | While the aromaticity of the benzene ring allows the delocalization the negative charge throughout the ring to stabilizes the anion, the dominant effect is the induction from the sp<sup>2</sup> hybridized carbons (the comparatively more powerful inductive withdrawal of electron density that is provided by the sp<sup>2</sup> system compared to an sp<sup>3</sup> system allows for great stabilization of the oxyanion). In making this conclusion, we cite the pK<sub>a</sub> of acetone enol, which is 10.9 (in comparison to phenol with a pK<sub>a</sub> of 10.0). | + | While the aromaticity of the benzene ring allows the delocalization the negative charge throughout the ring to stabilizes the anion, the dominant effect is the induction from the sp<sup>2</sup> hybridized carbons (the comparatively more powerful inductive withdrawal of electron density that is provided by the sp<sup>2</sup> system compared to an sp<sup>3</sup> system allows for great stabilization of the oxyanion). In making this conclusion, we cite the pK<sub>a</sub> of acetone enol, which is 10.9 (in comparison to phenol with a pK<sub>a</sub> of 10.0). |
| − | |||
| − | |||
==Production== | ==Production== | ||
| Line 116: | Line 113: | ||
==Uses== | ==Uses== | ||
| − | Phenol has [[antiseptic]] properties | + | |
| + | Phenol has [[antiseptic]] properties and was used by Sir [[Joseph Lister]] (1827-1912) in his pioneering technique of antiseptic surgery, though the skin irritation caused by continual exposure to phenol eventually led to the substitution of aseptic (germ-free) techniques in surgery. It is also the active ingredient in some oral anesthetics such as [[Chloraseptic]] spray. Phenol was also the main ingredient of the [[Carbolic Smoke Ball]], a device sold in London designed to protect the user against influenza and other ailments. | ||
It is also used in the production of drugs (it is the starting material in the industrial production of [[aspirin]]), [[weedkiller]], and [[chemical synthesis|synthetic]] [[resins]] ([[Bakelite]], one of the first synthetic resins to be manufactured, is a [[polymer]] of phenol with [[formaldehyde]]). Exposure of the [[skin]] to concentrated phenol solutions causes chemical burns which may be severe; in [[laboratory|laboratories]] where it is used, it is usually recommended that [[polyethylene glycol]] solution is kept available for washing off splashes. Washing with large amounts of plain [[water]] (most labs have a safety shower or eye-wash) and removal of contaminated clothing are required, and immediate ER treatment for large splashes; particularly if the phenol is mixed with [[chloroform]] (a commonly used mixture in molecular biology for [[DNA]] purification). Notwithstanding the effects of concentrated solutions, it is also used in [[cosmetic surgery]] as an [[exfoliant]], to remove layers of dead skin. It is also used in [[phenolization]], a surgical procedure used to treat an [[ingrown nail]], in which it is applied to the toe to prevent regrowth of [[nail]]s. | It is also used in the production of drugs (it is the starting material in the industrial production of [[aspirin]]), [[weedkiller]], and [[chemical synthesis|synthetic]] [[resins]] ([[Bakelite]], one of the first synthetic resins to be manufactured, is a [[polymer]] of phenol with [[formaldehyde]]). Exposure of the [[skin]] to concentrated phenol solutions causes chemical burns which may be severe; in [[laboratory|laboratories]] where it is used, it is usually recommended that [[polyethylene glycol]] solution is kept available for washing off splashes. Washing with large amounts of plain [[water]] (most labs have a safety shower or eye-wash) and removal of contaminated clothing are required, and immediate ER treatment for large splashes; particularly if the phenol is mixed with [[chloroform]] (a commonly used mixture in molecular biology for [[DNA]] purification). Notwithstanding the effects of concentrated solutions, it is also used in [[cosmetic surgery]] as an [[exfoliant]], to remove layers of dead skin. It is also used in [[phenolization]], a surgical procedure used to treat an [[ingrown nail]], in which it is applied to the toe to prevent regrowth of [[nail]]s. | ||
| − | Injections of phenol have occasionally been used as a means of rapid [[execution]]. In particular, phenol was used as a means of extermination by the [[Nazi]]s during the [[Second World War]]. Phenol injections were given to thousands of people in [[Nazi concentration camps|concentration camps]], especially at [[Auschwitz-Birkenau]]. Injections were administered either by [[medical doctor]]s or by their [[physician assistant|assistant]]s; such injections were originally given [[Intravenous therapy|intravenous]]ly, more commonly in the [[arm]], but injection directly into the [[heart]], so as to induce nearly instant [[death]], was later preferred. One of the most famous inmates at Auschwitz to be murdered by carbolic acid injection was [[St. Maximilian Kolbe]], a Catholic priest who volunteered to undergo three weeks of starvation and dehydration in the place of another inmate and who was finally injected with carbolic acid so that the Nazis could make more room in their holding cells.<ref> | + | == Misuse by the [[Nazi]]s == |
| + | |||
| + | Injections of phenol have occasionally been used as a means of rapid [[execution]]. In particular, phenol was used as a means of extermination by the [[Nazi]]s during the [[Second World War]]. Phenol injections were given to thousands of people in [[Nazi concentration camps|concentration camps]], especially at [[Auschwitz-Birkenau]]. Injections were administered either by [[medical doctor]]s or by their [[physician assistant|assistant]]s; such injections were originally given [[Intravenous therapy|intravenous]]ly, more commonly in the [[arm]], but injection directly into the [[heart]], so as to induce nearly instant [[death]], was later preferred. One of the most famous inmates at Auschwitz to be murdered by carbolic acid injection was [[St. Maximilian Kolbe]], a Catholic priest who volunteered to undergo three weeks of starvation and dehydration in the place of another inmate and who was finally injected with carbolic acid so that the Nazis could make more room in their holding cells.<ref>[http://www.wsg-hist.uni-linz.ac.at/AUSCHWITZ/HTML/Phenol.html Auschwitz - Final Station Extermination: Killing Through Phenol Injection.] ''Johannes Kepler University, Linz, Austria''. Retrieved June 4, 2007.</ref> | ||
== See also == | == See also == | ||
| − | |||
| − | |||
| − | == | + | * [[Acid]] |
| + | * [[Benzene]] | ||
| + | |||
| + | == Notes == | ||
{{refs}} | {{refs}} | ||
| + | |||
| + | == References == | ||
| + | |||
| + | * McMurry, John. 2004. ''Organic Chemistry''. 6th ed. Belmont, CA: Brooks/Cole. ISBN 0534420052. | ||
| + | |||
| + | * Morrison, Robert T., and Robert N. Boyd. 1992. ''Organic Chemistry''. 6th ed. Englewood Cliffs, NJ: Prentice Hall. ISBN 0-13-643669-2. | ||
| + | |||
| + | * Solomons, T.W. Graham, and Fryhle, Craig B. 2004. ''Organic Chemistry''. 8th ed. Hoboken, NJ: John Wiley. ISBN 0471417998. | ||
==External links== | ==External links== | ||
| − | * [http://www.ilo.org/public/english/protection/safework/cis/products/icsc/dtasht/_icsc00/icsc0070.htm International | + | |
| + | * [http://www.ilo.org/public/english/protection/safework/cis/products/icsc/dtasht/_icsc00/icsc0070.htm Phenol, ICSC 0070.] ''International Occupational Safety and Health Information Centre''. Retrieved June 4, 2007. | ||
| + | |||
* [http://www.jtbaker.com/msds/englishhtml/p1949.htm Phenol Material Safety Data Sheet] | * [http://www.jtbaker.com/msds/englishhtml/p1949.htm Phenol Material Safety Data Sheet] | ||
* [http://www.npi.gov.au/database/substance-info/profiles/70.html National Pollutant Inventory - Phenol Fact Sheet] | * [http://www.npi.gov.au/database/substance-info/profiles/70.html National Pollutant Inventory - Phenol Fact Sheet] | ||
* [http://www.cdc.gov/niosh/npg/npgd0493.html NIOSH Pocket Guide to Chemical Hazards] | * [http://www.cdc.gov/niosh/npg/npgd0493.html NIOSH Pocket Guide to Chemical Hazards] | ||
* [http://www-cie.iarc.fr/htdocs/monographs/vol71/027-phenol.html IARC Monograph: "Phenol"] | * [http://www-cie.iarc.fr/htdocs/monographs/vol71/027-phenol.html IARC Monograph: "Phenol"] | ||
| − | |||
[[Category:Physical sciences]] | [[Category:Physical sciences]] | ||
Revision as of 20:53, 4 June 2007
| Phenol | |
|---|---|
| |
| Systematic name | Phenol |
| Other names | Carbolic Acid Benzenol Phenylic Acid Hydroxybenzene Phenic acid |
| Chemical formula | C6H5OH |
| SMILES | Oc1ccccc1 |
| Molar mass | 94.11 g/mol |
| Appearance | White Crystalline Solid |
| CAS number | [108-95-2] |
| Properties | |
| Density | 1.07 g/cm3 |
| Solubility in water | 8.3 g/100 ml (20 °C) |
| Melting point | 40.5 °C |
| Boiling point | 181.7 °C |
| Acidity (pKa) | 9.95 |
| Structure | |
| Molecular shape | planar |
| Dipole moment | ? D |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Toxic (T) Muta. Cat. 3 Corrosive (C) |
| NFPA 704 | |
| R-phrases | R23/24/25, R34, R48/23/24/25, R68 |
| S-phrases | S1/2, S24/25, S26, S28, S36/37/39, S45 |
| Flash point | 79 °C |
| Autoignition temperature | 715 °C |
| RTECS number | SJ3325000 |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Related compounds | Benzenethiol |
| Disclaimer and references | |
Phenol, also known under an older name of carbolic acid, is a toxic, colorless crystalline solid with a distinctive sweet tarry odor. Its chemical formula is C6H5OH and its structure is that of a hydroxyl group (-OH) bonded to a phenyl ring; it is thus an aromatic compound.
Phenols
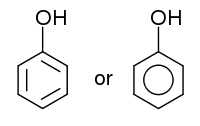
The word phenol is also used to refer to any compound which contains a six-membered aromatic ring, bonded directly to a hydroxyl group (-OH). In effect, phenols are a class of organic compounds of which the phenol discussed in this article is the simplest member.
Properties
Phenol has a limited solubility in water (8.3 g/100 ml). It is slightly acidic: the phenol molecule has weak tendencies to lose the H+ ion from the hydroxyl group, resulting in the highly water-soluble phenoxide anion C6H5O−. Compared to aliphatic alcohols, phenol shows much higher acidity; it even reacts with NaOH to lose H+ whereas aliphatic alcohols do not. Contrary to popular belief, this has little to do with orbital overlap between the oxygen's lone pairs and the aromatic system.
While the aromaticity of the benzene ring allows the delocalization the negative charge throughout the ring to stabilizes the anion, the dominant effect is the induction from the sp2 hybridized carbons (the comparatively more powerful inductive withdrawal of electron density that is provided by the sp2 system compared to an sp3 system allows for great stabilization of the oxyanion). In making this conclusion, we cite the pKa of acetone enol, which is 10.9 (in comparison to phenol with a pKa of 10.0).
Production
Phenol can be made from the partial oxidation of benzene or benzoic acid, by the cumene process, or by the Raschig process. It can also be found as a product of coal oxidation.
Uses
Phenol has antiseptic properties and was used by Sir Joseph Lister (1827-1912) in his pioneering technique of antiseptic surgery, though the skin irritation caused by continual exposure to phenol eventually led to the substitution of aseptic (germ-free) techniques in surgery. It is also the active ingredient in some oral anesthetics such as Chloraseptic spray. Phenol was also the main ingredient of the Carbolic Smoke Ball, a device sold in London designed to protect the user against influenza and other ailments.
It is also used in the production of drugs (it is the starting material in the industrial production of aspirin), weedkiller, and synthetic resins (Bakelite, one of the first synthetic resins to be manufactured, is a polymer of phenol with formaldehyde). Exposure of the skin to concentrated phenol solutions causes chemical burns which may be severe; in laboratories where it is used, it is usually recommended that polyethylene glycol solution is kept available for washing off splashes. Washing with large amounts of plain water (most labs have a safety shower or eye-wash) and removal of contaminated clothing are required, and immediate ER treatment for large splashes; particularly if the phenol is mixed with chloroform (a commonly used mixture in molecular biology for DNA purification). Notwithstanding the effects of concentrated solutions, it is also used in cosmetic surgery as an exfoliant, to remove layers of dead skin. It is also used in phenolization, a surgical procedure used to treat an ingrown nail, in which it is applied to the toe to prevent regrowth of nails.
Misuse by the Nazis
Injections of phenol have occasionally been used as a means of rapid execution. In particular, phenol was used as a means of extermination by the Nazis during the Second World War. Phenol injections were given to thousands of people in concentration camps, especially at Auschwitz-Birkenau. Injections were administered either by medical doctors or by their assistants; such injections were originally given intravenously, more commonly in the arm, but injection directly into the heart, so as to induce nearly instant death, was later preferred. One of the most famous inmates at Auschwitz to be murdered by carbolic acid injection was St. Maximilian Kolbe, a Catholic priest who volunteered to undergo three weeks of starvation and dehydration in the place of another inmate and who was finally injected with carbolic acid so that the Nazis could make more room in their holding cells.[1]
See also
Notes
- ↑ Auschwitz - Final Station Extermination: Killing Through Phenol Injection. Johannes Kepler University, Linz, Austria. Retrieved June 4, 2007.
ReferencesISBN links support NWE through referral fees
- McMurry, John. 2004. Organic Chemistry. 6th ed. Belmont, CA: Brooks/Cole. ISBN 0534420052.
- Morrison, Robert T., and Robert N. Boyd. 1992. Organic Chemistry. 6th ed. Englewood Cliffs, NJ: Prentice Hall. ISBN 0-13-643669-2.
- Solomons, T.W. Graham, and Fryhle, Craig B. 2004. Organic Chemistry. 8th ed. Hoboken, NJ: John Wiley. ISBN 0471417998.
External links
- Phenol, ICSC 0070. International Occupational Safety and Health Information Centre. Retrieved June 4, 2007.
- Phenol Material Safety Data Sheet
- National Pollutant Inventory - Phenol Fact Sheet
- NIOSH Pocket Guide to Chemical Hazards
- IARC Monograph: "Phenol"
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.