Bakelite
Bakelite is a material based on a thermosetting phenol formaldehyde resin developed in 1907–1909, by the Belgian-American chemist Leo Baekeland. It was the first plastic made from synthetic components. It was used for its electrically nonconductive and heat-resistant properties in radio and telephone casings and electrical insulators, and was also used in such diverse products as kitchenware, jewelry, pipe stems, and children's toys.
In 1993, Bakelite was designated an ACS National Historical Chemical Landmark in recognition of its significance as the world's first synthetic plastic.[1] Bakelite AG, a German company, owns the trademark rights in many countries around the world.[2]
Given the cost, complexity of production, and brittle nature of Bakelite, it is no longer used extensively for general consumer products. Nonetheless, the retro appeal of old Bakelite products and labor-intensive manufacturing has made them quite collectible in recent years. Also, Bakelite-type materials are used for various industrial applications in the electronics, power generation, and aerospace industries, and under a variety of commercial brand names.
Business history
Bakelite Corp. was formed, in 1922, from the consolidation of three companies. General Bakelite Co., Condensite Corp. and Redmanol Chemical Products Company, an early plastics manufacturer formed in 1913, by chemist L.H. Baekeland. The American Catalin Corporation acquired the Bakelite formulas, in 1927, and currently manufactures Bakelite cast resins.
Bakelite Limited was formed, in 1926, from the amalgamation of three suppliers of phenol formaldehyde materials: The Damard Lacquer Company Limited of Birmingham, Mouldensite Limited of Darley Dale, and Redmanol Chemical Products Company of London. Around 1928, a new factory opened in Tyseley, Birmingham, England. (The building was demolished in 1998.) The company was acquired by the Union Carbide and Carbon Corporation in 1939.
Properties
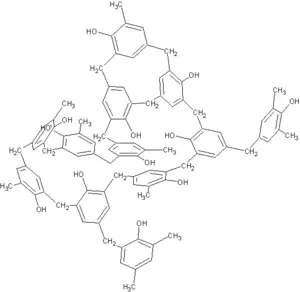
Bakelite is based on a phenol formaldehyde resin with the chemical name polyoxybenzyl methylene glycol anhydride. It was formed by the reaction under heat and pressure of phenol, a toxic, colorless crystalline solid, and formaldehyde, a simple organic compound, generally with a wood flour filler.
The phenolic sheet is a hard, dense material made by applying heat and pressure to layers of paper or glass cloth impregnated with synthetic resin. These layers of laminations are usually of cellulose paper, cotton fabrics, synthetic yarn fabrics, glass fabrics or unwoven fabrics. When heat and pressure are applied to the layers, a chemical reaction (polymerization) transforms the layers into a high-pressure thermosetting industrial laminated plastic. When rubbed, original Bakelite has a telltale odor.
Bakelite Phenolic is produced in dozens of commercial grades and with various additives to meet diverse mechanical, electrical and thermal requirements. Some common types and their properties are noted below.
- PAPER REINFORCED PHENOLIC NEMA XX per MIL-I-24768 PBG: Normal electrical applications, moderate mechanical strength, continuous operating temperature of 250°F.
- CANVAS REINFORCED PHENOLIC NEMA C per MIL-I-24768 TYPE FBM NEMA CE per MIL-I-24768 TYPE FBG: Good mechanical and impact strength with continuous operating temperature of 250°F.
- LINEN REINFORCED PHENOLIC NEMA L per MIL-I-24768 TYPE FBI NEMA LE per MIL-I-24768 TYPE FEI: Good mechanical and electrical strength. Recommended for intricate high strength parts. Continuous operating temperature 250°F.
- NYLON REINFORCED PHENOLIC NEMA N-1 per MIL-I-24768 TYPE NPG: Superior electrical properties under humid conditions, fungus resistant, continuous operating temperature of 160°F.
Applications
Bakelite is not extensively used for general consumer products any more, due to the cost and complexity of production and its brittle nature. However, it was used in the past in a myriad applications, such as saxophone mouthpieces, cameras, solid-body electric guitars, rotary-dial telephones, early machine guns, and appliance casings. It was at one point considered for the manufacture of coins, due to a shortage of traditional manufacturing material.
An exception to the overall decline is the use in small, precision-shaped components where their specific properties are required, such as molded disc brake cylinders, saucepan handles, electrical plugs and switches, and electrical iron parts. Today, Bakelite-type materials are manufactured and produced in the form of sheets, rods and tubes for many industrial applications in the electronics, power generation, and aerospace industries, and under a variety of commercial brand names.
See also
Notes
- ↑ New Products. American Chemical Society. Retrieved August 25, 2008.
- ↑ Our reference—Protection of brands. Bakelite AG. Retrieved August 25, 2008.
ReferencesISBN links support NWE through referral fees
- Battle, Dee, and Alayne Lesser. The Best of Bakelite & Other Plastic Jewelry. A Schiffer Book for Collectors. Atglen, PA: Schiffer Pub, 1996. ISBN 0887409016.
- Mauzy, Barbara E. Bakelite in the Kitchen. A Schiffer Book for Collectors. Atglen, PA: Schiffer Pub, 2001. ISBN 0764313797.
- Meikle, Jeffrey L. American Plastic: A Cultural History. New Brunswick, NJ: Rutgers University Press, 1997. ISBN 0813522358.
External links
All links retrieved August 26, 2023.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.