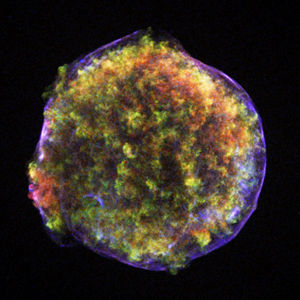
Plasma (matter)
- This article is about plasma in the sense of an ionized gas. The other common usage is plasma (blood).
In physics and chemistry, a plasma is one of the four principal states of matter. Plasma is typically an ionized gas, but it is usually considered a distinct state of matter in contrast to gases because of its unique properties. "Ionized" means that at least one electron is more free to move and is not bound to an atom or molecule.
In three states of matter—solid, liquid, and gas—the electrical interaction keeps the negative electrons and positive atomic nuclei in close proximity. In the fourth state of matter—a plasma—the electrical charges move independently, their energy being too great for stable atoms to form. For instance, at the temperature of the Sun's surface, all atoms have at least one electron missing while at temperatures deep inside the Sun, even the most massive of atoms have all their electrons stripped away. The atoms are fully ionized.
The free electric charges make the plasma electrically conductive so that it responds strongly to electromagnetic fields. The great loops seen on the sun's surface are composed of plasma flowing along magnetic field bundles.
A gas can be turned into a plasma in the following ways 1. It is heated to a high temperature (the sun is an example) 2. It is flooded with a discharge of high-energy electrons that strip the atoms bare (a neon tube uses this method) 3. It is flooded with high-energy photons, such as ultra-violet light (the sun's corona, which is only visible during a total eclipse, is made in this way)
Everyday examples of plasmas are sparks and lightning bolts.
History
This state of matter was first identified in a Crookes tube, and so described by Sir William Crookes in 1879 (he called it "radiant matter").[1] The nature of the Crookes tube "cathode ray" matter was subsequently identified by British physicist Sir J.J. Thomson in 1897 in his evening lecture to the Royal Institution on Friday, April 30. 1897,[2] and dubbed "plasma" by Irving Langmuir in 1928:
Except near the electrodes, where there are sheaths containing very few electrons, the ionized gas contains ions and electrons in about equal numbers so that the resultant space charge is very small. We shall use the name plasma to describe this region containing balanced charges of ions and electrons.[3]
In a short while, it was realized that plasma is actually a very common state for matter to be in. Indeed, plasmas are the most common phase of matter. Some estimates suggest that up to 99 percent of matter in the entire visible universe is plasma.[4][5] Essentially all of the visible light from space comes from stars, which are plasmas with a temperature such that they radiate strongly at visible wavelengths. Most of the ordinary (or baryonic) matter in the universe, however, is found in the intergalactic medium, which is also a plasma, but much hotter, so that it radiates primarily as x-rays. The current scientific consensus is that about 96 percent of the total energy density in the universe is not plasma or any other form of ordinary matter, but a combination of cold dark matter and dark energy. Since the space between the stars is filled with a plasma, albeit a very sparse one (see interstellar medium and intergalactic space), essentially the entire volume of the universe is plasma (see astrophysical plasmas). In the solar system, the planet Jupiter accounts for most of the non-plasma, only about 0.1 percent of the mass and 10−15 percent of the volume within the orbit of Pluto. Notable plasma physicist Hannes Alfvén also noted that due to their electric charge, very small grains also behave as ions and form part of plasma (see dusty plasmas).
| Common forms of plasma include | ||
|
|
|
Cosmology tells us that the matter in the universe was a uniform plasma for the first 100,000 years after the Big Bang. Only then, when the universe had cooled sufficiently, did the electric interaction start to unite the independent electric charges into neutral atoms. A million or so years later, gravity had fragmented that uniformity into stars, back to the plasma state.
Plasma properties and parameters

Definition of a plasma
Although a plasma is loosely described as an electrically neutral medium of positive and negative particles, a more rigorous definition requires three criteria to be satisfied:
- The plasma approximation: Charged particles must be close enough together that each particle influences many nearby charged particles, rather than just interacting with the closest particle (these collective effects are a distinguishing feature of a plasma). The plasma approximation is valid when the number of electrons within the sphere of influence—called the Debye sphere—is large.[7]
- Bulk interactions: The Debye sphere is small compared to the physical size of the plasma. This criterion means that interactions in the bulk of the plasma are more important than those at its edges, where boundary effects may take place.
- Plasma frequency: The electron plasma frequency (measuring plasma oscillations of the electrons) is large compared to the electron-neutral collision frequency (measuring frequency of collisions between electrons and neutral particles). When this condition is valid, plasmas act to shield charges very rapidly (quasineutrality is another defining property of plasmas).
Ranges of plasma parameters
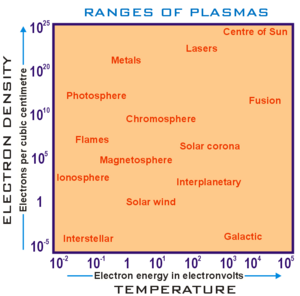
Plasma parameters can take on values varying by many orders of magnitude, but the properties of plasmas with apparently disparate parameters may be very similar (see plasma scaling). The following chart considers only conventional atomic plasmas and not exotic phenomena like quark gluon plasmas:
| Typical ranges of plasma parameters: orders of magnitude (OOM) | ||
| Characteristic | Terrestrial plasmas | Cosmic plasmas |
| Size in metres | 10−6 m (lab plasmas) to 102 m (lightning) (~8 OOM) | 10−6 m (spacecraft sheath) to 1025 m (intergalactic nebula) (~31 OOM) |
| Lifetime in seconds | 10−12 s (laser-produced plasma) to 107 s (fluorescent lights) (~19 OOM) | 101 s (solar flares) to 1017 s (intergalactic plasma) (~17 OOM) |
| Density in particles per cubic metre | 107 m-3 to 1032 m-3 (inertial confinement plasma) | 100 (i.e., 1) m-3 (intergalactic medium) to 1030 m-3 (stellar core) |
| Temperature in kelvins | ~0 K (crystalline non-neutral plasma[9]) to 108 K (magnetic fusion plasma) | 102 K (aurora) to 107 K (solar core) |
| Magnetic fields in teslas | 10−4 T (lab plasma) to 103 T (pulsed-power plasma) | 10−12 T (intergalactic medium) to 1011 T (near neutron stars) |
Degree of ionization

For plasma to exist, ionization is necessary. The word "plasma density" by itself usually refers to the electron density, that is, the number of free electrons per unit volume. The degree of ionization of a plasma is the proportion of atoms which have lost (or gained) electrons, and is controlled mostly by the temperature. Even a partially ionized gas in which as little as 1 percent of the particles are ionized can have the characteristics of a plasma (i.e., respond to magnetic fields and be highly electrically conductive).[10]
Temperatures
Plasma temperature is commonly measured in kelvins or electronvolts, and is (roughly speaking) a measure of the thermal kinetic energy per particle. In most cases the electrons are close enough to thermal equilibrium that their temperature is relatively well-defined, even when there is a significant deviation from a Maxwellian energy distribution function, for example due to UV radiation, energetic particles, or strong electric fields. Because of the large difference in mass, the electrons come to thermodynamic equilibrium among themselves much faster than they come into equilibrium with the ions or neutral atoms. For this reason the ion temperature may be very different from (usually lower than) the electron temperature. This is especially common in weakly ionized technological plasmas, where the ions are often near the ambient temperature.
Based on the relative temperatures of the electrons, ions and neutrals, plasmas are classified as thermal or non-thermal. Thermal plasmas have electrons and the heavy particles at the same temperature, that is, they are in thermal equilibrium with each other. Non-thermal plasmas, on the other hand, have the ions and neutrals at a much lower temperature (normally room temperature) whereas electrons are much "hotter."
Temperature controls the degree of plasma ionization. In particular, plasma ionization is determined by the electron temperature relative to the ionization energy (and more weakly by the density) in accordance with the Saha equation. A plasma is sometimes referred to as being hot if it is nearly fully ionized, or cold if only a small fraction (for example 1 percent) of the gas molecules are ionized (but other definitions of the terms hot plasma and cold plasma are common). Even in a "cold" plasma the electron temperature is still typically several thousand degrees Celsius. Plasmas utilized in plasma technology ("technological plasmas") are usually cold in this sense.
Potentials
Since plasmas are very good conductors, electric potentials play an important role. The potential as it exists on average in the space between charged particles, independent of the question of how it can be measured, is called the plasma potential or the space potential. If an electrode is inserted into a plasma, its potential will generally lie considerably below the plasma potential due to the development of a Debye sheath. Due to the good electrical conductivity, the electric fields in plasmas tend to be very small. This results in the important concept of quasineutrality, which says that it is a very good approximation to assume that the density of negative charges is equal to the density of positive charges over large volumes of the plasma. [12]
It is, of course, possible to produce a plasma that is not quasineutral. An electron beam, for example, has only negative charges. The density of a non-neutral plasma must generally be very low, or it must be very small, otherwise it will be dissipated by the repulsive electrostatic force.
In astrophysical plasmas, Debye screening prevents electric fields from directly affecting the plasma over large distances (i.e., greater than the Debye length). But the existence of charged particles causes the plasma to generate and be affected by magnetic fields. This can and does cause extremely complex behavior, such as the generation of plasma double layers, an object that separates charge over a few tens of Debye lengths. The dynamics of plasmas interacting with external and self-generated magnetic fields are studied in the academic discipline of magnetohydrodynamics.
Magnetization
A plasma in which the magnetic field is strong enough to influence the motion of the charged particles is said to be magnetized. A common quantitative criterion is that a particle on average completes at least one gyration around the magnetic field before making a collision.[13]. It is often the case that the electrons are magnetized while the ions are not. Magnetized plasmas are anisotropic, meaning that their properties in the direction parallel to the magnetic field are different from those perpendicular to it. [14]
Comparison of plasma and gas phases
Plasma is often called the "fourth state of matter." It is distinct from other lower-energy states of matter; most commonly solid, liquid, and gas, although it is closely related to the gas phase in that it also has no definite form or volume. Physicists consider a plasma to be more than a gas because of a number of distinct properties including the following:
| Property | Gas | Plasma |
| Electrical Conductivity | Very low
| Usually very high
|
| Independently acting species | One
|
Two or three |
| Velocity distribution | Maxwellian
|
Often non-Maxwellian
|
| Interactions | Binary
|
Collective
|
Complex plasma phenomena
Although the underlying equations governing plasmas are relatively simple, plasma behavior is extraordinarily varied and subtle: the emergence of unexpected behavior from a simple model is a typical feature of a complex system. Such systems lie in some sense on the boundary between ordered and disordered behavior, and cannot typically be described either by simple, smooth, mathematical functions, or by pure randomness. The spontaneous formation of interesting spatial features on a wide range of length scales is one manifestation of plasma complexity. The features are interesting, for example, because they are very sharp, spatially intermittent (the distance between features is much larger than the features themselves), or have a fractal form. Many of these features were first studied in the laboratory, and have subsequently been recognized throughout the universe. Examples of complexity and complex structures in plasmas include:
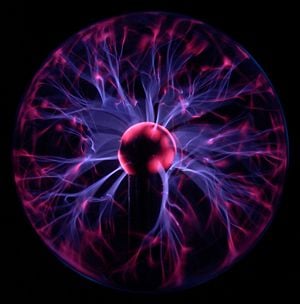
Filamentation
The striations or "stringy" things,[16] seen in many plasmas, like the plasma ball (image above), the aurora,[17] lightning,[18] electric arcs, solar flares,[19] and supernova remnants[20] They are sometimes associated with larger current densities, and are also called "magnetic ropes."[21]
Shocks or double layers
Narrow sheets with sharp gradients, such as shocks or double layers which support rapid changes in plasma properties. Double layers involve localized charge separation, which causes a large potential difference across the layer, but does not generate an electric field outside the layer. Double layers separate adjacent plasma regions with different physical characteristics, and are often found in current carrying plasmas. They accelerate both ions and electrons.

Electric fields and circuits
Quasineutrality of a plasma requires that plasma currents close on themselves in electric circuits. Such circuits follow Kirchhoff's circuit laws, and possess a resistance and inductance. These circuits must generally be treated as a strongly coupled system, with the behavior in each plasma region dependent on the entire circuit. It is this strong coupling between system elements, together with nonlinearity, which may lead to complex behavior. Electrical circuits in plasmas store inductive (magnetic) energy, and should the circuit be disrupted, for example, by a plasma instability, the inductive energy will be released as plasma heating and acceleration. This is a common explanation for the heating which takes place in the solar corona. Electric currents, and in particular, magnetic-field-aligned electric currents (which are sometimes generically referred to as Birkeland currents), are also observed in the Earth's aurora, and in plasma filaments.
Cellular structure
Narrow sheets with sharp gradients may separate regions with different properties such as magnetization, density, and temperature, resulting in cell-like regions. Examples include the magnetosphere, heliosphere, and heliospheric current sheet. Hannes Alfvén wrote: "From the cosmological point of view, the most important new space research discovery is probably the cellular structure of space. As has been seen, in every region of space which is accessible to in situ measurements, there are a number of 'cell walls', sheets of electric currents, which divide space into compartments with different magnetization, temperature, density, etc."[23]
Critical ionization velocity
The Critical ionization velocity is the relative velocity between an (magnetized) ionized plasma and a neutral gas above which a runaway ionization process takes place. The critical ionization process is a quite general mechanism for the conversion of the kinetic energy of a rapidly streaming gas into ionization and plasma thermal energy. Critical phenomena in general are typical of complex systems, and may lead to sharp spatial or temporal features.
Ultracold plasma
[[Image:saturn_rings_false.jpg|thumb|170px|right|Saturn's rings in which certain effects have been suggested are due to dusty plasmas[24] [25] (false color image)[26]
It is possible to create ultracold plasmas, by using lasers to trap and cool neutral atoms to temperatures of 1 mK or lower. Another laser then ionizes the atoms by giving each of the outermost electrons just enough energy to escape the electrical attraction of its parent ion.
The key point about ultracold plasmas is that by manipulating the atoms with lasers, the kinetic energy of the liberated electrons can be controlled. Using standard pulsed lasers, the electron energy can be made to correspond to a temperature of as low as 0.1 K, a limit set by the frequency bandwidth of the laser pulse. The ions, however, retain the millikelvin temperatures of the neutral atoms. This type of non-equilibrium ultracold plasma evolves rapidly, and many fundamental questions about its behavior remain unanswered. Experiments conducted so far have revealed surprising dynamics and recombination behavior that are pushing the limits of our knowledge of plasma physics.
Non-neutral plasma
The strength and range of the electric force and the good conductivity of plasmas usually ensure that the density of positive and negative charges in any sizable region are equal ("quasineutrality"). A plasma that has a significant excess of charge density or that is, in the extreme case, composed of only a single species, is called a non-neutral plasma. In such a plasma, electric fields play a dominant role. Examples are charged particle beams, an electron cloud in a Penning trap, and positron plasmas.[27]
Dusty plasma and grain plasma
A dusty plasma is one containing tiny charged particles of dust (typically found in space) that also behaves like a plasma. A plasma containing larger particles is called a grain plasma.
Mathematical descriptions

To completely describe the state of a plasma, we would need to write down all the particle locations and velocities, and describe the electromagnetic field in the plasma region. However, it is generally not practical or necessary to keep track of all the particles in a plasma. Therefore, plasma physicists commonly use less detailed descriptions known as models, of which there are two main types:
Fluid model
Fluid models describe plasmas in terms of smoothed quantities like density and averaged velocity around each position (see Plasma parameters). One simple fluid model, magnetohydrodynamics, treats the plasma as a single fluid governed by a combination of Maxwell's Equations and the Navier Stokes Equations. A more general description is the two-fluid picture, where the ions and electrons are described separately. Fluid models are often accurate when collisionality is sufficiently high to keep the plasma velocity distribution close to a Maxwell-Boltzmann distribution. Because fluid models usually describe the plasma in terms of a single flow at a certain temperature at each spatial location, they can neither capture velocity space structures like beams or double layers nor resolve wave-particle effects.
Kinetic model
Kinetic models describe the particle velocity distribution function at each point in the plasma, and therefore do not need to assume a Maxwell-Boltzmann distribution. A kinetic description is often necessary for collisionless plasmas. There are two common approaches to kinetic description of a plasma. One is based on representing the smoothed distribution function on a grid in velocity and position. The other, known as the particle-in-cell (PIC) technique, includes kinetic information by following the trajectories of a large number of individual particles. Kinetic models are generally more computationally intensive than fluid models. The Vlasov equation may be used to describe how a system of particles evolves in an electromagnetic environment.
Fields of active research
Plasmas are the object of study of the academic field of plasma science or plasma physics, including sub-disciplines such as space plasma physics. It involves a large number of fields of active research and features across many journals.
See also
Notes
- ↑ Sir William Crookes, On Radiant Mmatter: A lecture delivered to the British Association for the Advancement of Science, at Sheffield, Friday, August 22, 1879 (HardPress Publishing, 2012, ISBN 978-1290308670).
- ↑ J.J. Thomson, 'Cathode Rays" Philosophical Magazine 44(293) (1897). Retrieved May 21, 2018.
- ↑ Irving Langmuir, Oscillations in ionized gases. Proc. Nat. Acad. Sci. U.S. 14(8) (1928): 627-637. Retrieved May 21, 2018.
- ↑ D.A. Gurnett and A. Bhattacharjee, Introduction to Plasma Physics: With Space and Laboratory Applications (Cambridge, UK: Cambridge University Press, 2005, ISBN 0521364833).
- ↑ K. Scherer, H. Fichtner, and B. Heber, Space Weather: The Physics Behind a Slogan (New York, NY: Springer, 2005).
- ↑ Mike Carlowicz, Solar Wind Squeezes Some of Earth's Atmosphere into Space. NASA, December 8, 1998. Retrieved May 21, 2018.
- ↑ The radius is the Debye (screening) length) of a particular particle. The average number of particles in the Debye sphere is given by the plasma parameter, Λ.
- ↑ After Anthony L. Peratt, Advances in Numerical Modeling of Astrophysical and Space Plasmas. Astrophysics and Space Science 242(1/2) (1966(: 93-163. Retrieved May 2, 2018.
- ↑ C. Fred Driscoll, et al. The Nonneutral Plasma Group. University of California, San Diego. Retrieved May 22, 2018.
- ↑ The degree of ionization, α is defined as α = ni/(ni + na) where ni is the number density of ions and na is the number density of neutral atoms. The electron density is related to this by the average charge state <Z> of the ions through ne=<Z> ni where ne is the number density of electrons.
- ↑ Tim Stephens and Rachel A. Weintraub, Flashes in the Sky: Earth's Gamma-Ray Bursts Triggered by Lightning. NASA, February 17, 2005. Retrieved May 22, 2018.
- ↑ This equality (), but on the scale of the Debye length there can be charge imbalance. In the special case that double layers are formed, the charge separation can extend some tens of Debye lengths. The magnitude of the potentials and electric fields must be determined by means other than simply finding the net charge density. A common example is to assume that the electrons satisfy the Boltzmann relation:
- . Differentiating this relation provides a means to calculate the electric field from the density:
- .
- ↑ This is (i.e., where is the "electron gyrofrequency" and is the "electron collision rate")
- ↑ While electric fields in plasmas are usually small due to the high conductivity, the electric field associated with a plasma moving in a magnetic field is given by E = -v x B (where E is the electric field, v is the velocity, and B is the magnetic field), and is not affected by Debye shielding. See: Richard Fitzpatrick, Magnetized plasmas Introduction to Plasma Physics. Retrieved May 22, 2018.
- ↑ Dielectric Strength of Air. Retrieved May 22, 2018.
- ↑ J. R. Dickel, The Filaments in Supernova Remnants: Sheets, Strings, Ribbons, or? Bulletin of the American Astronomical Society 22 (1990): 832. Retrieved May 22, 2018.
- ↑ T. Grydeland, et al., Interferometric observations of filamentary structures associated with plasma instability in the auroral ionosphere. Geophysical Research Letters 30(6) (March 2003)1. Retrieved May 22, 2018.
- ↑ Gregory D. Moss, et al., Monte Carlo model for analysis of thermal runaway electrons in streamer tips in transient luminous events and streamer zones of lightning leaders. Journal of Geophysical Research 111 (February 2006): A2. Retrieved May 22, 2018.
- ↑ Lowell R. Doherty, Filamentary Structure in Solar Prominences. Astrophysical Journal 141 (1965): 251. Retrieved May 22, 2018.
- ↑ Jeff Hester and Paul Scowen, Hubble views the Crab Nebula M1: The Crab Nebula Filaments. NASA. Retrieved May 22, 2018.
- ↑ Yan-An Zhang, et al., A rope-shaped solar filament and a IIIb flare. Chinese Astronomy and Astrophysics 26(4) (2002): 442-450. Retrieved May 22, 2018.
- ↑ A Star with two North Poles. NASA, April 22, 2003. Retrieved May 22, 2018.
- ↑ Hannes Alfvén, Cosmic Plasma (Springer, 1981, ISBN 978-9027711519).
- ↑ M. Horanyi, et al., Dusty Plasma Effects in Saturn's Rings. American Geophysical Union, Fall Meeting 2004. Retrieved May 22, 2018.
- ↑ Hiroshi Kikuchi (ed.), Dusty and Dirty Plasmas, Noise, and Chaos in Space and in the Laboratory (New York, NY: Plenum Press, 1994, ISBN 0306448394).
- ↑ Saturn: Rings NASA. Retrieved May 22, 2018.
- ↑ R.G. Greaves, M.D. Tinkle, and C. M. Surko, Creation and uses of positron plasmas. Physics of Plasmas 1(5) (1994): 1439-1446. Retrieved May 22, 2018.
- ↑ Evolution of the Solar System. NASA. Retrieved May 22, 2018.
ReferencesISBN links support NWE through referral fees
- Alfvén, Hannes. Cosmic Plasma. Springer, 1981. ISBN 978-9027711519
- Bellan, Paul M. Fundamentals of Plasma Physics. Cambridge, UK: Cambridge University Press, 2006. ISBN 0521821169
- Chen, Francis F. Introduction to Plasma Physics and Controlled Fusion. New York, NY: Springer, 2006. ISBN 0306413329
- Crookes, Sir William. On Radiant Mmatter: A lecture delivered to the British Association for the Advancement of Science, at Sheffield, Friday, August 22, 1879. HardPress Publishing, 2012. ISBN 978-1290308670
- Fridman, Alexander A. and Lawrence A. Kennedy. Plasma Physics and Engineering. Boca Raton, FL: CRC Press, 2004. ISBN 1560328487
- Gurnett, D. A. and A. Bhattacharjee. Introduction to Plasma Physics: With Space and Laboratory Applications. Cambridge, UK: Cambridge University Press, 2005. ISBN 0521364833
- Kikuchi, Hiroshi (ed.). Dusty and Dirty Plasmas, Noise, and Chaos in Space and in the Laboratory. New York, NY: Plenum Press, 1994. ISBN 0306448394
- Scherer, K., H. Fichtner, and B. Heber. Space Weather: The Physics Behind a Slogan. New York, NY: Springer, 2005. ISBN 3540229078
External links
All links retrieved March 7, 2026.
- Plasma Science and Technology.
- Plasma on the Internet comprehensive list of plasma related links.
- Coalition for Plasma Science.
| States of Matter |
|---|
| Solid | Liquid | Gas | Plasma | Colloid | Supercritical fluid | Superfluid | Supersolid | Degenerate matter | Quark-gluon plasma | Fermionic condensate | Bose–Einstein condensate | Strange matter | Melting point | Boiling point | Triple point | Critical point | Equation of state | Cooling curve |
| Nuclear technology | |
|---|---|
| Nuclear engineering | Nuclear physics | Nuclear fission | Nuclear fusion | Radiation | Ionizing radiation | Atomic nucleus | Nuclear reactor | Nuclear safety |
| Nuclear material | Nuclear fuel | Fertile material | Thorium | Uranium | Enriched uranium | Depleted uranium | Plutonium |
| Nuclear power | Nuclear power plant | Radioactive waste | Fusion power | Future energy development | Inertial fusion power plant | Pressurized water reactor | Boiling water reactor | Generation IV reactor | Fast breeder reactor | Fast neutron reactor | Magnox reactor | Advanced gas-cooled reactor | Gas-cooled fast reactor | Molten salt reactor | Liquid-metal-cooled reactor | Lead-cooled fast reactor | Sodium-cooled fast reactor | Supercritical water reactor | Very high temperature reactor | Pebble bed reactor | Integral Fast Reactor | Nuclear propulsion | Nuclear thermal rocket | Radioisotope thermoelectric generator |
| Nuclear medicine | PET | Radiation therapy | Tomotherapy | Proton therapy | Brachytherapy |
| Nuclear weapons | History of nuclear weapons | Nuclear warfare | Nuclear arms race | Nuclear weapon design | Effects of nuclear explosions | Nuclear testing | Nuclear delivery | Nuclear proliferation | List of states with nuclear weapons | List of nuclear tests |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.