Ethylene oxide
- "Oxirane" redirects here.For oxiranes as a class of molecules, see Epoxide.
| Ethylene oxide | |
|---|---|
|
|
| IUPAC name | epoxyethane |
| Other names | ethylene oxide, dimethylene oxide, oxirane, oxacyclopropane |
| Identifiers | |
| Abbreviations | EO |
| CAS number | [] |
| PubChem | |
| EINECS number | |
| KEGG | |
| MeSH | |
| ChEBI | |
| RTECS number | KX2450000 |
| SMILES | C1CO1 |
| InChI | InChI=1/C2H4O/c1-2-3-1/h1-2H2 |
| Properties | |
| Molecular formula | C2H4O |
| Molar mass | 44.05 g mol−1 |
| Appearance | colorless gas |
| Density | 0.882 g/mL, 7.360 lbs/gallon |
| Melting point |
−111.3 °C |
| Boiling point |
10.7 °C |
| Solubility in water | miscible |
| Thermochemistry | |
| Std enthalpy of formation ΔfH |
−52.6 kJ mol−1 |
| Standard molar entropy S |
243 J mol−1 K−1 |
| Hazards | |
| NFPA 704 |
|
| Flash point | −20 °C |
| Explosive limits | 3 to 100% |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Ethylene oxide (chemical formula C2H4O) is an important industrial chemical. It is also known as epoxyethane (IUPAC name), oxirane, and dimethylene oxide. At room temperature and ordinary atmospheric pressure, it is a flammable gas, so it is usually stored as a pressurized or refrigerated liquid.
This chemical is used mostly as an intermediate in the production of ethylene glycol, which in turn is converted into polyester fibers used in clothes and carpets, or into polyethylene terephthalate resin for plastic packaging film and bottles. Ethylene glycol is widely used as an automotive antifreeze, and it may be used for manufacturing fiberglass.
A minor but significant use of ethylene oxide is for the sterilization of foods and medical supplies. In addition, some of its derivatives are ingredients in cleaners, cosmetics, shampoos, plasticizers, ointments, and pharmaceuticals.
Ethylene oxide, however, is toxic by inhalation and is classified as a carcinogen. Symptoms of overexposure include headache and dizziness, and increased exposure leads to convulsions, seizure, and coma. It is also an irritant to the skin and respiratory tract.
History
Ethylene oxide was first prepared in 1859 by the French chemist Charles-Adolphe Wurtz.[1] He obtained it by treating 2-chloroethanol with a base. It achieved industrial importance during World War I as a precursor to both the coolant ethylene glycol and the chemical weapon mustard gas.
In 1931, Theodore Lefort, another French chemist, discovered a means to prepare ethylene oxide directly from ethylene and oxygen, using silver as a catalyst. Since 1940, this method has been the predominant means of production of ethylene oxide industrially.[2]
Production
For the industrial production of ethylene oxide, ethylene (H2C=CH2) is reacted with oxygen (O2) at 200–300 °C, in the presence of a silver catalyst (containing large silver nanoparticles) supported on alumina. Typically, chemical modifiers such as chlorine are also included. Pressures used for the reaction are in the neighborhood of 1-2 MPa. The chemical equation for this reaction is:
- H2C=CH2 + ½ O2 → C2H4O
The typical yield for this reaction under industrial conditions is 70-80 percent.
The above reaction takes place through an intermediate (oxametallacycle), leading to two possible reaction pathways, as follows.
- Ethylene oxide formation:
- H2C=CH2 + O → C2H4O
- Acetaldehyde formation:
- H2C=CH2 + O → CH3CHO
The latter pathway is the first step in complete combustion, which produces carbon dioxide and water:
- CH3CHO + 5/2 O2 → 2CO2 + 2H2O
In the laboratory, ethylene oxide can be conveniently produced by the action of an alkali hydroxide (OH−) on ethylene chlorohydrin (2-chloroethanol, CH2OH−CH2Cl), as follows.[3]
- CH2OH−CH2Cl + OH− → C2H4O + Cl− + H2O
Note that ethylene chlorohydrin can be readily prepared by the action of hypochlorous acid (HOCl) on ethylene.
Properties
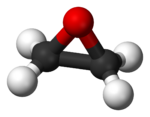
Ethylene oxide is a colorless, flammable gas at room temperature. Its melting point is −111.3 °C, and its boiling point is 10.7 °C. Thus, when refrigerated, it becomes a liquid. It has a faintly sweet odor. It is the simplest example of an epoxide.
Reactions
Most reactions of ethylene oxide result in opening its ring structure.
In industrial processes, it is reacted with water in the presence of sulfuric acid as catalyst. A tenfold molar excess of water is used to obtain ethylene glycol, as follows:
- C2H4O + H2O → HOCH2CH2OH
Various types of polyethylene glycol (PEG) or polyethylene oxide (PEO), which are polymers of ethylene glycol, are formed as secondary products of the reaction. The degree of polymerization increases as the proportion of water is reduced:
- n(CH2CH2O) + H2O → HO(CH2CH2O)nH
For example, under the appropriate conditions, the reaction produces diethylene glycol (HOCH2CH2OCH2CH2OH), triethylene glycol, and so forth.
Similarly, reaction with ammonia can yield ethanolamine, diethanolamine, or triethanolamine.
Ethylene oxide is also important in the manufacture of surfactants and other detergents, in a process called ethoxylation.
One class of ethylene oxide derivatives that has attracted much scientific attention consists of the crown ethers, which are cyclic oligomers of ethylene oxide. These compounds have the ability to make ionic compounds such as salts soluble in nonpolar solvents, which they otherwise will not dissolve in. However, the high cost of these compounds has largely confined their use to the laboratory rather than industrial practice.
Uses
The main use of ethylene oxide is as an intermediate in the production of various other chemicals. A prominent derivative is ethylene glycol, which in turn can be converted into polyester fibers or polyethylene terephthalate (PET) resin. Polyester fibers are used in clothing, carpets, and upholstery; PET resin is a recyclable plastic used for packaging film and bottles. In addition, ethylene glycol is known for its use as an automotive coolant and antifreeze; and it is used in the manufacture of fiberglass, for such items as bathtubs, bowling balls, and jet skis.
Ethylene oxide gas kills bacteria (and their endospores), mold, and fungi. It can therefore be used to sterilize substances that would be damaged by techniques such as pasteurization that rely on heat. Ethylene oxide sterilization for the preservation of spices was patented in 1938 by the American chemist Lloyd Hall, and it is still used in that role. Additionally, ethylene oxide is widely used to sterilize medical supplies such as bandages, sutures, and surgical implements. The overwhelming majority of medical items are sterilized with ethylene oxide.
Preferred methods of sterilization have been (a) the traditional chamber sterilization method, in which a chamber is flooded with a mix of ethylene oxide and other gases that are later aerated, and (b) the more recent gas diffusion method, developed in 1967, which relies on a bag that encloses the elements to be sterilized and acts as a mini-chamber to minimize gas consumption and make the process economically feasible for small loads. Other names for the latter method are the Anprolene method, bag sterilization method, or micro-dose sterilization method.
Some derivatives of ethylene oxide serve as ingredients in household and industrial cleaners, cosmetics, shampoos, heat-transfer liquids, plasticizers, ointments, and pharmaceuticals.
Given its high flammability and wide explosive concentration range in air, ethylene oxide is sometimes used as the fuel component of a fuel-air explosive.
Health effects
Ethylene oxide is toxic by inhalation. Symptoms of overexposure include headache and dizziness, progressing with increasing exposure to convulsions, seizure, and coma. It is also an irritant to the skin and respiratory tract, and inhaling its vapors may cause the lungs to fill with fluid several hours after exposure.[4]
Ethylene oxide is usually stored as a pressurized or refrigerated liquid. At room temperature and pressure, it rapidly evaporates, potentially causing frostbite in cases of skin exposure.
Laboratory animals exposed to ethylene oxide for their entire lives have had a higher incidence of liver cancer. However, studies on human beings who have worked with ethylene oxide for extended periods and may have experienced low doses during that time have found no increase in cancer risk. Chronic ethylene oxide exposure may increase the risk of cataracts in humans.
Animals exposed to ethylene oxide have been found to suffer from higher rates of mutations and miscarriages. Effects of the chemical on humans have not been well studied, but it is considered probable that ethylene oxide exposure has similar effects on human reproduction.
Ethylene oxide is classified as carcinogenic to humans by the International Agency for Research on Cancer (IARC) of the World Health Organization.[5]
See also
- Acetaldehyde
- Epoxy
- Ethylene glycol
- Polyethylene glycol
Notes
- ↑ A. Wurtz, 1859, Compt. rend 48:101-104.
- ↑ P.P. McClellan, 1950, Manufacture and Uses of Ethylene Oxide and Ethylene Glycol, Ind. Eng. Chem 42:2402-2407. DOI 10.1021/ie50492a013.
- ↑ Andrew Streitwieser and Clayton H. Heathcock, Introduction to Organic Chemistry (New York: Macmillan, 1976, ISBN 0024180106).
- ↑ Material Safety Data Sheet: Ethylene Oxide Andersen Sterilizers, Haw River, NC. Retrieved May 8, 2008.
- ↑ IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol. 60: Some Industrial Chemicals World Health Organization. Retrieved May 8, 2008.
ReferencesISBN links support NWE through referral fees
- McMurry, John. Organic Chemistry, 6th ed. Belmont, CA: Thomson-Brooks/Cole, 2004. ISBN 0534420052
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry, 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0-13-643669-2
- Solomons, T.W. Graham, and Craig B. Fryhle. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998
- Streitwieser, Andrew, and Clayton H. Heathcock. Introduction to Organic Chemistry. New York: Macmillan, 1976. ISBN 0024180106
External links
All links retrieved March 22, 2024.
- Ethylene Oxide National Institute for Occupational Safety and Health.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
