Ethylene glycol
| Ethylene glycol | |
|---|---|
| |
| |
| |
| IUPAC name | Ethane-1,2-diol |
| Other names | Ethylene glycol Monoethylene glycol MEG 1,2-ethanediol |
| Identifiers | |
| CAS number | [] |
| SMILES | OCCO |
| Properties | |
| Molecular formula | C2H4(OH)2 |
| Molar mass | 62.068 g/mol |
| Density | 1.1132 g/cm³ |
| Melting point |
−12.9 °C (260 K) |
| Boiling point |
197.3 °C (470 K) |
| Solubility in water | Miscible with water in all proportions. |
| Viscosity | 16.1 mPa s [1] |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Harmful (Xn) |
| NFPA 704 |
|
| R-phrases | R22 R36 |
| S-phrases | S26 S36 S37 S39 S45 S53 |
| Flash point | 111 °C (closed cup) |
| Autoignition temperature |
410 °C |
| Related Compounds | |
| Related diols | Propylene glycol, diethylene glycol, triethylene glycol |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Ethylene glycol (also called monoethylene glycol (MEG); 1,2-ethanediol; or ethane-1,2-diol (IUPAC name)) is a chemical compound with the formula C2H4(OH)2. It is classified as a diol, that is, an alcohol with two -OH groups. In its pure form, it is an odorless, colorless, syrupy liquid with a sweet taste. However, it is toxic.
This compound is widely used as an antifreeze and coolant in automobiles, and as a deicing fluid for windshields and aircraft. It is being used to inhibit the formation of natural gas clathrates in long, multiphase, natural gas pipelines. It is also a good desiccant for natural gas production. The plastics industry uses ethylene glycol for the manufacture of polyester fibers and resins. In laboratories, ethylene glycol is used to precipitate out proteins from solution, often as an intermediary step in fractionation, purification, and crystallization.
History
Ethylene glycol was first prepared in 1859 by the French chemist Charles-Adolphe Wurtz. It was produced on a small scale during World War I, for use as a coolant and as an ingredient in explosives. Widespread industrial production began in 1937, when ethylene oxide, a component in its synthesis, became cheaply available.
When first introduced, it created a minor revolution in aircraft design because, when used in place of water as an engine coolant, its higher boiling point allowed for smaller radiators operating at higher temperatures. Prior to the widespread availability of ethylene glycol, many aircraft manufacturers tried to use evaporative cooling systems that used water at high pressure. Invariably, these proved to be unreliable and were easily damaged in combat because they took up large amounts of room on the plane and were easily hit by gunfire.
Production
Ethylene glycol is produced from ethylene, via the intermediate ethylene oxide. Ethylene oxide reacts with water to produce ethylene glycol according to the chemical equation
This reaction can be catalyzed by either acids or bases, or can occur at neutral pH under elevated temperatures. The highest yields of ethylene glycol occur at acidic or neutral pH with a large excess of water. Under these conditions, ethylene glycol yields of 90 percent can be achieved. The major byproducts are the ethylene glycol oligomers diethylene glycol, triethylene glycol, and tetraethylene glycol.
This molecule has been observed in space by Hollis, et al.[2]
Uses
Coolant
The major use of ethylene glycol is as an antifreeze, particularly in automobiles. Given its low freezing point, it is also used as a deicing fluid for windshields and aircraft. Ethylene glycol is also commonly used in chilled water air conditioning systems that place either the chiller or air handlers outside, or systems that must cool below the freezing temperature of water.
Hydrate inhibition
Ethylene glycol is being widely used to inhibit the formation of natural gas clathrates in long, multiphase pipelines that convey natural gas from remote gas fields back to an onshore processing facility. Ethylene glycol can be recovered from the natural gas and reused as an inhibitor after a purification treatment that removes water and inorganic salts.
Manufacturing
Ethylene glycol has become increasingly important in the plastics industry for the manufacture of polyester fibers and resins, including polyethylene terephthalate, which is used to make plastic bottles for soft drinks. The antifreeze capabilities of ethylene glycol have made it an important component of vitrification mixtures for low-temperature preservation of biological tissues and organs.
Minor uses of ethylene glycol include the manufacture of capacitors, as a chemical intermediate in the manufacture of 1,4-dioxane, and as an additive to prevent corrosion in liquid cooling systems for personal computers.
Chemistry
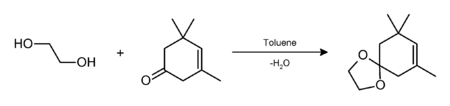
Ethylene glycol may also be used as a protecting group for carbonyl groups in organic synthesis. Reacting a ketone or aldehyde with ethylene glycol, in the presence of an acid catalyst (e.g. p-toluenesulfonic acid; BF3·Et2O), will give a cyclic acetal — a 1,3-dioxolane, which is resistant to bases and other nucleophiles. The 1,3-dioxolane protecting group can thereafter be removed, e.g. by further acid hydrolysis.[3] In this example, isophorone was protected using ethylene glycol with p-toluenesulfonic acid in moderate yield. Water was removed by azeotropic distillation to shift the equilibrium to the right.[4]
Laboratory use
Ethylene glycol is commonly used in laboratories to precipitate out proteins in solution. This is often an intermediary step in fractionation, purification, and crystallization. It can be used to protect functional groups from reacting during organic synthesis. The original functional group can be restored by simply adding water and acid.
Ethylene glycol is commonly used as a preservative for specimens in schools, frequently during dissection. It is said to be safer than formaldehyde, but the safety is questionable.
Other applications
Ethylene glycol's high boiling point and affinity for water make it an ideal desiccant for natural gas production. In the field, excess water vapor is usually removed by glycol dehydration. Ethylene glycol flows down from the top of a tower and meets a mixture of water vapor and hydrocarbon gases rising from the bottom. The glycol chemically removes the water vapor, allowing dry gas to exit from the top of the tower. The glycol and water are separated, and the glycol is cycled back through the tower.
Instead of removing water, ethylene glycol can also be used to depress the temperature at which hydrates are formed. The purity of glycol used for hydrate suppression (mono-ethylene glycol) is typically around 80 percent, whereas the purity of glycol used for dehydration (tri-ethylene glycol) is typically 95-99+ percent. Moreover, the injection rate for hydrate suppression is much lower than the circulation rate in a glycol dehydration tower.
Ethylene glycol is also used in the manufacture of some vaccines, but it is not itself present in these injections. It is used as a minor (1–2 percent) ingredient in shoe polish and also in some inks and dyes. Ethylene glycol has seen some use as a rot and fungal treatment for wood, both as a preventative and a treatment after the fact. It has been used in a few cases to treat partially rotted wooden objects to be displayed in museums. It is one of only a few treatments that are successful in dealing with rot in wooden boats, and is relatively cheap. Ethylene glycol may also be one of the minor ingredients in screen cleaning solutions, along with the main ingredient, isopropyl alcohol.
Toxicity
The most significant danger from ethylene glycol is ingestion. Due to its sweet taste, children and animals sometimes consume large quantities of it, if allowed access to it. Ethylene glycol may also be found as a contaminant in moonshine, distilled using an improperly washed car radiator. In many developed countries, a bittering agent called denatonium/denatonium benzoate (known in some locales by the trade name Bitrex), is generally added to ethylene glycol preparations as an "adversant," to prevent accidental ingestion. This embitterment is not yet widespread in the United States.
Ethylene glycol poisoning is a medical emergency, and in all cases, a poison control center should be contacted or medical attention should be sought. It is highly toxic, with an estimated lethal dose (LD100) in humans of approximately 1.4 ml/kg.[5] However, as little as 30 milliliters (2 tablespoons) can be lethal for adults.[6]
Symptoms
Symptoms of ethylene glycol poisoning usually follow a three-step progression, although poisoned individuals do not always develop each stage.[5] Stage 1 consists of neurological symptoms including victims appearing to be intoxicated, exhibiting symptoms such as dizziness, headaches, slurred speech, and confusion. Over time, the body metabolizes ethylene glycol into other toxins, first to glycolaldehyde, which is then oxidized to glycolic acid, glyoxylic acid, and finally oxalic acid. Stage 2 is a result of accumulation of these metabolites and consists of tachycardia, hypertension, hyperventilation, and metabolic acidosis. Stage 3 of ethylene glycol poisoning is the result of kidney injury, leading to acute kidney failure.[7] Oxalic acid reacts with calcium and forms calcium oxalate crystals in the kidney. An estimated 80 percent of kidney stones are calcium oxalate.
Treatment
Initial treatment consists of stabilizing the patient and gastric decontamination. As ethylene glycol is rapidly absorbed, gastric decontamination needs to be performed soon after ingestion to be of benefit. Gastric lavage or nasogastric aspiration of gastric contents are the most common methods employed in ethylene glycol poisoning. Ipecac-induced vomiting or activated charcoal (charcoal does not adsorb glycols) are not recommended. [5]
The antidotes for ethylene glycol poisoning are ethanol or fomepizole; antidotal treatment forms the mainstay of management following ingestion. Ethanol (usually given IV as a 5 or 10 percent solution in 5 percent dextrose and water, but also sometimes given in the form of a strong spirit such as whisky, vodka or gin) acts by competing with ethylene glycol for the enzyme alcohol dehydrogenase thus limiting the formation of toxic metabolites. Fomepizole acts by inhibiting alcohol dehydrogenase, thus blocking the formation of the toxic metabolites.[8]
In addition to antidotes, hemodialysis can be used to enhance the removal of unmetabolized ethylene glycol, as well as its metabolites from the body. Hemodialysis has the added benefit of correcting other metabolic derangements or supporting deteriorating kidney function caused by ethylene glycol ingestion. Often both antidotal treatment and hemodialysis are used together in the treatment of poisoning.
Industrial hazards
Ethylene glycol can begin to break down at 230° – 250°F (110° – 121°C). Note that breakdown can occur when the system bulk (average) temperature is below these limits because surface temperatures in heat exchangers and boilers can be locally well above these temperatures.
The electrolysis of ethylene glycol solutions with a silver anode results in an exothermic reaction. In the Apollo 1 fire catastrophe, a coolant consisting of ethylene glycol and water was implicated as a possible cause via this reaction.
See also
Notes
- ↑ Glenn Elert. 2008. Viscosity. The Physics Hypertextbook. Retrieved September 21, 2008.
- ↑ J. M. Hollis, F. J. Lovas, P. R. Jewell, and L. H. Coudert. May 20, 2002. Interstellar Antifreeze: Ethylene Glycol. The AstroPhysical Journal. 571:L59–L62.
- ↑ Theodora W. Greene, and Peter G. M. Wuts. 1999. Protective Groups in Organic Synthesis, 3rd ed. (New York: Wiley), 312-322. ISBN 0471160199.
- ↑ J. H. Babler, N. C. Malek, and M. J. Coghlan. 1978. Selective hydrolysis of α,β- and β,γ-unsaturated ketals: method for deconjugation of β,β-disubstituted α,β-unsaturated ketones. J. Org. Chem. 43(9): 1821–1823. (doi = 10.1021/jo00403a047).
- ↑ 5.0 5.1 5.2 J. Brent, 2001. Current management of ethylene glycol poisoning. Drugs 61(7): 979–88. (pmid = 11434452; doi = 10.2165/00003495-200161070-00006).
- ↑ D. Field, 1985. Acute ethylene glycol poisoning. Crit. Care Med. 13(10): 872–3. (pmid = 4028762; doi = 10.1097/00003246-198510000-00024).
- ↑ D.G. Barceloux, E.P. Krenzelok, K. Olson, and W. Watson. 1999. American Academy of Clinical Toxicology Practice Guidelines on the Treatment of Ethylene Glycol Poisoning. Ad Hoc Committee. J. Toxicol. Clin. Toxicol. 37(5): 537–560. (pmid=10497633; doi=10.1081/CLT-100102445).
- ↑ J. Brent, K. McMartin, S. Phillips, K. Burkhart, J. Donovan, M. Wells, and K. Kulig. 1999. Fomepizole for the treatment of ethylene glycol poisoning. Methylpyrazole for Toxic Alcohols Study Group. New Engl. J. Med. 340(11): 832–838. (pmid = 10080845).
ReferencesISBN links support NWE through referral fees
- Greene, Theodora W. and Peter G. M. Wuts. Protective Groups in Organic Synthesis, 3rd ed. New York: Wiley, 1999. ISBN 0471160199
- McMurry, John. Organic Chemistry, 6th ed. Belmont, CA: Thomson-Brooks/Cole, 2004. ISBN 0534420052.
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry, 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692.
- Solomons, T.W. Graham, and Craig B. Fryhle. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998.
- Yaws, Carl L. The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals: Physical Properties for More Than 41,000 Organic and Inorganic Chemical Compounds: Coverage for C1 to C100 Organics and Ac to Zr Inorganics. Houston: Gulf Pub. Co., 2005. ISBN 978-0976511373.
External links
All links retrieved March 22, 2024.
- Ethylene Glycol and Propylene Glycol Toxicity. Agency for Toxic Substances and Disease Registry (ATSDR).
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.