Difference between revisions of "Hydrolysis" - New World Encyclopedia
Rosie Tanabe (talk | contribs) |
|||
| (16 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
| − | '''Hydrolysis''' is a [[chemical reaction]] | + | {{Images OK}}{{Approved}}{{copyedited}} |
| + | '''Hydrolysis''' is a [[chemical reaction]] in which a [[chemical compound]] decomposes by reaction with [[water]]. The reacting [[water (molecule)|water molecules]] are split into hydrogen (H<sup>+</sup>) and hydroxide (OH<sup>−</sup>) ions, which react with and break up (or "lyse") the other reacting compound.<ref>IUPAC Gold Book, [http://goldbook.iupac.org/H02902.html Hydrolysis]. Retrieved September 24, 2008.</ref> The term ''hydrolysis'' is also applied to the [[electrolysis]] of water (that is, breaking up of water molecules by an electric current) to produce [[hydrogen]] and [[oxygen]]. | ||
| + | {{toc}} | ||
| + | The hydrolysis reaction breaks down a variety of [[polymer]]s, including [[protein]]s, [[carbohydrate]]s, [[fat]]s, and [[nucleic acid]]s. Such reactions may be [[catalyst|catalyzed]] by [[acid]]s, [[base (chemistry)|bases]], or [[enzyme]]s (classified as ''[[hydrolase]]s''). Such reactions regularly occur during the process of [[digestion]] of [[food]] in living systems. In addition, hydrolysis is said to occur when a [[salt]] reacts with water to produce new [[ion (physics)|ions]] or [[precipitate]]s. | ||
| − | + | == Clarification of terminology == | |
| − | + | The hydrolysis reaction is distinct from a ''[[hydration reaction]]'', in which water molecules attach to molecules of the other reacting compound without breaking up the latter compound. Also, hydrolysis should not be confused with ''[[hydrogenolysis]],'' a reaction of hydrogen. | |
| − | |||
| − | |||
| − | |||
==Types== | ==Types== | ||
| + | There are various types of hydrolysis reactions, as noted below. | ||
| − | In [[organic chemistry]], hydrolysis can be considered | + | * In [[organic chemistry]], hydrolysis can be considered the reverse (or opposite) of [[condensation reaction|condensation]], a reaction in which two molecular fragments are joined for each water molecule produced. As hydrolysis may be a reversible reaction, condensation and hydrolysis can take place at the same time, with the position of equilibrium determining the amount of each product. A typical example is the hydrolysis of an ester to an acid and an alcohol. |
| − | :R<sup>1</sup>CO<sub>2</sub>R<sup>2</sup> + H<sub>2</sub>O {{unicode|⇌}} R<sup>1</sup>CO<sub>2</sub>H + R<sup>2</sup>OH | + | ::R<sup>1</sup>CO<sub>2</sub>R<sup>2</sup> + H<sub>2</sub>O {{unicode|⇌}} R<sup>1</sup>CO<sub>2</sub>H + R<sup>2</sup>OH |
| − | In [[inorganic chemistry]], the word is often applied to solutions of salts and the reactions by which they are converted to new ionic species or to precipitates (oxides, hydroxides, or salts) | + | * In [[inorganic chemistry]], the word is often applied to solutions of salts and the reactions by which they are converted to new ionic species or to precipitates (oxides, hydroxides, or salts). |
| − | In [[biochemistry]], hydrolysis is considered the reverse or opposite of [[dehydration synthesis]]. In hydrolysis, a water molecule (H<sub>2</sub>O), is added, whereas in dehydration synthesis, a molecule of water is removed. | + | * In [[biochemistry]], hydrolysis is considered the reverse or opposite of [[dehydration synthesis]]. In hydrolysis, a water molecule (H<sub>2</sub>O), is added, whereas in dehydration synthesis, a molecule of water is removed. |
| − | In [[electrochemistry]], hydrolysis can also refer to the [[electrolysis of water]]. | + | * In [[electrochemistry]], hydrolysis can also refer to the [[electrolysis of water]]. In hydrolysis, a voltage is applied across an aqueous medium, which produces a current and breaks the water into its constituents, [[hydrogen]] and [[oxygen]]. |
| − | In [[polymer chemistry]], hydrolysis of [[polymer]]s can occur during high-temperature processing such as [[injection moulding]] leading to chain degradation and loss of product integrity. Polymers most at risk include [[Polyethylene terephthalate|PET]], [[polycarbonate]], [[nylon]] and other polymers made by [[step-growth polymerization]]. Such materials must be dried prior to | + | * In [[polymer chemistry]], hydrolysis of [[polymer]]s can occur during high-temperature processing such as [[injection moulding]] leading to chain degradation and loss of product integrity. Polymers most at risk include [[Polyethylene terephthalate|PET]], [[polycarbonate]], [[nylon]] and other polymers made by [[step-growth polymerization]]. Such materials must be dried prior to molding. |
== Hydrolysis of amide links == | == Hydrolysis of amide links == | ||
| − | |||
In the hydrolysis of an [[amide]] link into a [[carboxylic acid]] and an [[amine]] or [[ammonia]], the carboxylic acid has an hydroxyl group derived from a water molecule and the amine (or ammonia) gains the hydrogen ion. | In the hydrolysis of an [[amide]] link into a [[carboxylic acid]] and an [[amine]] or [[ammonia]], the carboxylic acid has an hydroxyl group derived from a water molecule and the amine (or ammonia) gains the hydrogen ion. | ||
| Line 29: | Line 29: | ||
A specific case of the hydrolysis of an amide link is the hydrolysis of [[peptide bond|peptides]] to smaller fragments or [[amino acid]]s. | A specific case of the hydrolysis of an amide link is the hydrolysis of [[peptide bond|peptides]] to smaller fragments or [[amino acid]]s. | ||
| − | Many [[polyamide]] polymers such as [[nylon 6,6]] are attacked and | + | Many [[polyamide]] polymers such as [[nylon 6,6]] are attacked and hydrolyzed in the presence of strong acids. Such attack leads to [[depolymerization]] and nylon products fail by fracturing when exposed to even small amounts of acid. Other polymers made by [[step-growth polymerization]] are susceptible to similar [[polymer degradation]] reactions. The problem is known as [[stress corrosion cracking]]. |
== Hydrolysis of polysaccharides == | == Hydrolysis of polysaccharides == | ||
| − | [[image:Sucrose-inkscape.svg|thumb|Sucrose. The glycoside bond is represented by the | + | [[image:Sucrose-inkscape.svg|thumb|Sucrose. The glycoside bond is represented by the central oxygen atom that holds the two monosaccharide units together.]] |
| + | |||
| + | In a [[polysaccharide]] molecule, [[monosaccharide]] subunits are linked together by [[glycosidic bond]]s. This bond can be cleaved by hydrolysis to yield monosaccharides. The best known disaccharide is [[sucrose]] (table sugar). Hydrolysis of sucrose yields [[glucose]] and [[fructose]]. | ||
| − | + | There are many [[enzymes]] that speed up the hydrolysis of polysaccharides. [[Invertase]] is used industrially to hydrolyze sucrose to so-called [[invert sugar]]. Invertase is an example of a [[glycoside hydrolase]] (glucosidase). [[Lactase]] is essential for digestive hydrolysis of [[lactose]] in milk. Deficiency of the enzyme in humans causes [[lactose intolerance]]. β-[[amylase]] catalyzes the conversion of [[starch]] to [[maltose]]. Malt made from [[barley]] is used as a source of β-amylase to break down starch into a form that can be used by yeast to produce [[beer]]. The hydrolysis of [[cellulose]] into glucose, known as ''saccharification,'' is catalyzed by [[cellulase]]. Animals such as cows ([[ruminants]]) are able to digest cellulose because of the presence of parasitic bacteria that produce cellulases. | |
== Hydrolysis of metal aqua ions == | == Hydrolysis of metal aqua ions == | ||
| − | + | Metal ions are [[Lewis acid]]s, and in aqueous solution they form "aqua ions," of the general formula M(H<sub>2</sub>O)<sub>n</sub><sup>m+</sup>.<ref>John Burgess, ''Metal Ions in Solution'' (Chichester: Ellis Horwood, 1978, ISBN 0470262931).</ref> The aqua ions are hydrolyzed, to a greater or lesser extent. The first hydrolysis step is given generically as | |
| − | Metal ions are [[Lewis acid]]s, and in aqueous solution they form aqua ions, of the general formula M(H<sub>2</sub>O)<sub>n</sub><sup>m+</sup>.<ref>Burgess, | ||
: M(H<sub>2</sub>O)<sub>n</sub><sup>m+</sup> + H<sub>2</sub>O {{unicode|⇌}} M(H<sub>2</sub>O)<sub>n-1</sub>(OH)<sup>(m-1)+</sup> + H<sub>3</sub>O<sup>+</sup> | : M(H<sub>2</sub>O)<sub>n</sub><sup>m+</sup> + H<sub>2</sub>O {{unicode|⇌}} M(H<sub>2</sub>O)<sub>n-1</sub>(OH)<sup>(m-1)+</sup> + H<sub>3</sub>O<sup>+</sup> | ||
| Line 44: | Line 45: | ||
Thus the aqua ion is behaving as an acid in terms of [[Brønsted-Lowry acid-base theory]]. This is easily explained by considering the [[inductive effect]] of the positively charged metal ion, which weakens the O-H bond of an attached water molecule, making the liberation of a proton relatively easy. | Thus the aqua ion is behaving as an acid in terms of [[Brønsted-Lowry acid-base theory]]. This is easily explained by considering the [[inductive effect]] of the positively charged metal ion, which weakens the O-H bond of an attached water molecule, making the liberation of a proton relatively easy. | ||
| − | The [[Acid dissociation constant |dissociation constant]], pK<sub>a</sub>, for this reaction is more or less linearly related to the charge-to-size ratio of the metal ion.<ref name="bm"> | + | The [[Acid dissociation constant |dissociation constant]], pK<sub>a</sub>, for this reaction is more or less linearly related to the charge-to-size ratio of the metal ion.<ref name="bm">Charles F. Baes and Robert E. Mesmer, ''The Hydrolysis of Cations'' (New York: Wiley, 1976, ISBN 0471039853).</ref> Ions with low charges, such as Na<sup>+</sup> are very weak acids with almost imperceptible hydrolysis. Large divalent ions such as Ca<sup>2+</sup>, Zn<sup>2+</sup>, Sn<sup>2+</sup> and Pb<sup>2+</sup> have a pK<sub>a</sub> of 6 or more and would not normally be classed as acids, but small divalent ions such as Be<sup>2+</sup> are extensively hydrolyzed. Trivalent ions like Al<sup>3+</sup> and Fe<sup>3+</sup> are weak acids whose pK<sub>a</sub> is comparable to that of [[acetic acid]]. Solutions of salts such as BeCl<sub>2</sub> or Al(NO<sub>3</sub>)<sub>3</sub> in water are noticeably [[acidic]]; the hydrolysis can be [[le chatelier's principle|suppressed]] by adding an acid such as [[nitric acid]], making the solution more acidic. |
| − | Hydrolysis may proceed beyond the first step, often with the formation of polynuclear species. | + | Hydrolysis may proceed beyond the first step, often with the formation of polynuclear species.<ref name="bm"/> Some "exotic" species such as Sn<sub>3</sub>(OH)<sub>4</sub><sup>2+</sup><ref>N.N. Greenwood and A. Earnshaw, ''Chemistry of the Elements'' (Oxford: Butterworth-Heinemann, 2006).</ref> are well characterized. Hydrolysis tends to increase as [[pH]] rises leading, in many cases, to the precipitation of an hydroxide such as Al(OH)<sub>3</sub> or AlO(OH). These substances, the major constituents of [[bauxite]], are known as [[laterite]]s and are formed by leaching from rocks of most of the ions other than aluminum and iron and subsequent hydrolysis of the remaining aluminum and iron. |
| − | <ref name="bm"/> Some "exotic" species such as Sn<sub>3</sub>(OH)<sub>4</sub><sup>2+</sup> <ref> | ||
Ions with a formal charge of four are extensively hydrolyzed and salts of Zr<sup>4+</sup>, for example, can only be obtained from strongly acidic solutions. With oxidation states five and higher the concentration of the aqua ion in solution is negligible. In effect the aqua ion is a strong acid. For example, aqueous solutions of Cr(VI) contain CrO<sub>4</sub><sup>2-</sup>. | Ions with a formal charge of four are extensively hydrolyzed and salts of Zr<sup>4+</sup>, for example, can only be obtained from strongly acidic solutions. With oxidation states five and higher the concentration of the aqua ion in solution is negligible. In effect the aqua ion is a strong acid. For example, aqueous solutions of Cr(VI) contain CrO<sub>4</sub><sup>2-</sup>. | ||
| Line 60: | Line 60: | ||
== Irreversibility of hydrolysis under physiological conditions == | == Irreversibility of hydrolysis under physiological conditions == | ||
| − | + | Under physiological conditions (i.e. in dilute aqueous solution), a hydrolytic cleavage reaction, where the concentration of a metabolic precursor is low (on the order of 10<sup>-3</sup> to 10<sup>-6</sup> molar) and the reaction is catalyzed by an enzyme, is essentially [[thermodynamics|thermodynamically]] irreversible. This theme of physiological irreversibility of hydrolysis is used consistently in metabolic pathways, since many biological processes are driven by the cleavage of [[anhydride|anhydrous]] [[pyrophosphate]] bonds. | |
| − | Under physiological conditions (i.e. in dilute aqueous solution), a hydrolytic cleavage reaction, where the concentration of a metabolic precursor is low (on the order of 10<sup>-3</sup> to 10<sup>-6</sup> molar), is essentially [[thermodynamics|thermodynamically]] irreversible. | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | This theme of physiological irreversibility of hydrolysis is used consistently in metabolic pathways, since many biological processes are driven by the cleavage of [[anhydride|anhydrous]] [[pyrophosphate]] bonds. | ||
== See also == | == See also == | ||
| − | * [[ | + | * [[Enzyme]] |
| − | + | * [[Polymer]] | |
| − | * [[Polymer | ||
| − | |||
* [[Water]] | * [[Water]] | ||
| Line 91: | Line 72: | ||
== References == | == References == | ||
| − | |||
* Baes, Charles F., and Robert E. Mesmer. 1976. ''The Hydrolysis of Cations.'' New York: Wiley. ISBN 0471039853. | * Baes, Charles F., and Robert E. Mesmer. 1976. ''The Hydrolysis of Cations.'' New York: Wiley. ISBN 0471039853. | ||
| − | |||
* Burgess, John. 1978. ''Metal Ions in Solution.'' Chichester: Ellis Horwood. ISBN 0470262931. | * Burgess, John. 1978. ''Metal Ions in Solution.'' Chichester: Ellis Horwood. ISBN 0470262931. | ||
| − | |||
* Frey, Perry A., and Adrian D. Hegeman. 2007. ''Enzymatic Reaction Mechanisms.'' Oxford: Oxford University Press. ISBN 978-0195122589. | * Frey, Perry A., and Adrian D. Hegeman. 2007. ''Enzymatic Reaction Mechanisms.'' Oxford: Oxford University Press. ISBN 978-0195122589. | ||
| − | + | * Richens, David T. 1997. ''The Chemistry of Aqua Ions: Synthesis, Structure, and Reactivity: A Tour Through the Periodic Table of the Elements.'' Chichester: Wiley. ISBN 0471970581. | |
| − | * Richens, David T. 1997. ''The Chemistry of Aqua Ions: Synthesis, Structure, and Reactivity : A Tour Through the Periodic Table of the Elements.'' Chichester: Wiley. ISBN 0471970581. | ||
| − | |||
* Silverman, Richard B. 2002. ''The Organic Chemistry of Enzyme-Catalyzed Reactions.'' San Diego: Academic Press. ISBN 0126437319. | * Silverman, Richard B. 2002. ''The Organic Chemistry of Enzyme-Catalyzed Reactions.'' San Diego: Academic Press. ISBN 0126437319. | ||
| − | |||
* Testa, Bernard, and Joachim M. Mayer. 2003. ''Hydrolysis in Drug and Prodrug Metabolism: Chemistry, Biochemistry, and Enzymology.'' Weinheim: Wiley-VCH. ISBN 390639025X. | * Testa, Bernard, and Joachim M. Mayer. 2003. ''Hydrolysis in Drug and Prodrug Metabolism: Chemistry, Biochemistry, and Enzymology.'' Weinheim: Wiley-VCH. ISBN 390639025X. | ||
==External links== | ==External links== | ||
| − | + | All links retrieved January 22, 2018. | |
| − | + | * [https://www.thebalance.com/what-is-hydrolysis-375589 An Explanation of the Process Hydrolysis]. ''the balance''. | |
| − | |||
| − | * [ | ||
[[Category:Physical sciences]] | [[Category:Physical sciences]] | ||
Latest revision as of 20:06, 22 January 2018
Hydrolysis is a chemical reaction in which a chemical compound decomposes by reaction with water. The reacting water molecules are split into hydrogen (H+) and hydroxide (OH−) ions, which react with and break up (or "lyse") the other reacting compound.[1] The term hydrolysis is also applied to the electrolysis of water (that is, breaking up of water molecules by an electric current) to produce hydrogen and oxygen.
The hydrolysis reaction breaks down a variety of polymers, including proteins, carbohydrates, fats, and nucleic acids. Such reactions may be catalyzed by acids, bases, or enzymes (classified as hydrolases). Such reactions regularly occur during the process of digestion of food in living systems. In addition, hydrolysis is said to occur when a salt reacts with water to produce new ions or precipitates.
Clarification of terminology
The hydrolysis reaction is distinct from a hydration reaction, in which water molecules attach to molecules of the other reacting compound without breaking up the latter compound. Also, hydrolysis should not be confused with hydrogenolysis, a reaction of hydrogen.
Types
There are various types of hydrolysis reactions, as noted below.
- In organic chemistry, hydrolysis can be considered the reverse (or opposite) of condensation, a reaction in which two molecular fragments are joined for each water molecule produced. As hydrolysis may be a reversible reaction, condensation and hydrolysis can take place at the same time, with the position of equilibrium determining the amount of each product. A typical example is the hydrolysis of an ester to an acid and an alcohol.
- R1CO2R2 + H2O ⇌ R1CO2H + R2OH
- In inorganic chemistry, the word is often applied to solutions of salts and the reactions by which they are converted to new ionic species or to precipitates (oxides, hydroxides, or salts).
- In biochemistry, hydrolysis is considered the reverse or opposite of dehydration synthesis. In hydrolysis, a water molecule (H2O), is added, whereas in dehydration synthesis, a molecule of water is removed.
- In electrochemistry, hydrolysis can also refer to the electrolysis of water. In hydrolysis, a voltage is applied across an aqueous medium, which produces a current and breaks the water into its constituents, hydrogen and oxygen.
- In polymer chemistry, hydrolysis of polymers can occur during high-temperature processing such as injection moulding leading to chain degradation and loss of product integrity. Polymers most at risk include PET, polycarbonate, nylon and other polymers made by step-growth polymerization. Such materials must be dried prior to molding.
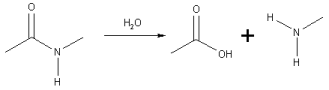
Hydrolysis of amide links
In the hydrolysis of an amide link into a carboxylic acid and an amine or ammonia, the carboxylic acid has an hydroxyl group derived from a water molecule and the amine (or ammonia) gains the hydrogen ion.
A specific case of the hydrolysis of an amide link is the hydrolysis of peptides to smaller fragments or amino acids.
Many polyamide polymers such as nylon 6,6 are attacked and hydrolyzed in the presence of strong acids. Such attack leads to depolymerization and nylon products fail by fracturing when exposed to even small amounts of acid. Other polymers made by step-growth polymerization are susceptible to similar polymer degradation reactions. The problem is known as stress corrosion cracking.
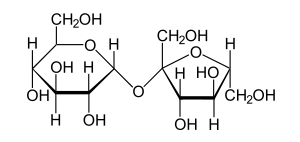
Hydrolysis of polysaccharides
In a polysaccharide molecule, monosaccharide subunits are linked together by glycosidic bonds. This bond can be cleaved by hydrolysis to yield monosaccharides. The best known disaccharide is sucrose (table sugar). Hydrolysis of sucrose yields glucose and fructose.
There are many enzymes that speed up the hydrolysis of polysaccharides. Invertase is used industrially to hydrolyze sucrose to so-called invert sugar. Invertase is an example of a glycoside hydrolase (glucosidase). Lactase is essential for digestive hydrolysis of lactose in milk. Deficiency of the enzyme in humans causes lactose intolerance. β-amylase catalyzes the conversion of starch to maltose. Malt made from barley is used as a source of β-amylase to break down starch into a form that can be used by yeast to produce beer. The hydrolysis of cellulose into glucose, known as saccharification, is catalyzed by cellulase. Animals such as cows (ruminants) are able to digest cellulose because of the presence of parasitic bacteria that produce cellulases.
Hydrolysis of metal aqua ions
Metal ions are Lewis acids, and in aqueous solution they form "aqua ions," of the general formula M(H2O)nm+.[2] The aqua ions are hydrolyzed, to a greater or lesser extent. The first hydrolysis step is given generically as
- M(H2O)nm+ + H2O ⇌ M(H2O)n-1(OH)(m-1)+ + H3O+
Thus the aqua ion is behaving as an acid in terms of Brønsted-Lowry acid-base theory. This is easily explained by considering the inductive effect of the positively charged metal ion, which weakens the O-H bond of an attached water molecule, making the liberation of a proton relatively easy.
The dissociation constant, pKa, for this reaction is more or less linearly related to the charge-to-size ratio of the metal ion.[3] Ions with low charges, such as Na+ are very weak acids with almost imperceptible hydrolysis. Large divalent ions such as Ca2+, Zn2+, Sn2+ and Pb2+ have a pKa of 6 or more and would not normally be classed as acids, but small divalent ions such as Be2+ are extensively hydrolyzed. Trivalent ions like Al3+ and Fe3+ are weak acids whose pKa is comparable to that of acetic acid. Solutions of salts such as BeCl2 or Al(NO3)3 in water are noticeably acidic; the hydrolysis can be suppressed by adding an acid such as nitric acid, making the solution more acidic.
Hydrolysis may proceed beyond the first step, often with the formation of polynuclear species.[3] Some "exotic" species such as Sn3(OH)42+[4] are well characterized. Hydrolysis tends to increase as pH rises leading, in many cases, to the precipitation of an hydroxide such as Al(OH)3 or AlO(OH). These substances, the major constituents of bauxite, are known as laterites and are formed by leaching from rocks of most of the ions other than aluminum and iron and subsequent hydrolysis of the remaining aluminum and iron.
Ions with a formal charge of four are extensively hydrolyzed and salts of Zr4+, for example, can only be obtained from strongly acidic solutions. With oxidation states five and higher the concentration of the aqua ion in solution is negligible. In effect the aqua ion is a strong acid. For example, aqueous solutions of Cr(VI) contain CrO42-.
- Cr(H2O)6+ → CrO42- + 2 H2O + 8 H+
Note that reactions such as
- 2 CrO42- + H2O ⇌ Cr2O72- + 2 OH-
are formally hydrolysis reactions as water molecules are split up yielding hydroxide ions. Such reactions are common among polyoxometalates.
Irreversibility of hydrolysis under physiological conditions
Under physiological conditions (i.e. in dilute aqueous solution), a hydrolytic cleavage reaction, where the concentration of a metabolic precursor is low (on the order of 10-3 to 10-6 molar) and the reaction is catalyzed by an enzyme, is essentially thermodynamically irreversible. This theme of physiological irreversibility of hydrolysis is used consistently in metabolic pathways, since many biological processes are driven by the cleavage of anhydrous pyrophosphate bonds.
See also
Notes
- ↑ IUPAC Gold Book, Hydrolysis. Retrieved September 24, 2008.
- ↑ John Burgess, Metal Ions in Solution (Chichester: Ellis Horwood, 1978, ISBN 0470262931).
- ↑ 3.0 3.1 Charles F. Baes and Robert E. Mesmer, The Hydrolysis of Cations (New York: Wiley, 1976, ISBN 0471039853).
- ↑ N.N. Greenwood and A. Earnshaw, Chemistry of the Elements (Oxford: Butterworth-Heinemann, 2006).
ReferencesISBN links support NWE through referral fees
- Baes, Charles F., and Robert E. Mesmer. 1976. The Hydrolysis of Cations. New York: Wiley. ISBN 0471039853.
- Burgess, John. 1978. Metal Ions in Solution. Chichester: Ellis Horwood. ISBN 0470262931.
- Frey, Perry A., and Adrian D. Hegeman. 2007. Enzymatic Reaction Mechanisms. Oxford: Oxford University Press. ISBN 978-0195122589.
- Richens, David T. 1997. The Chemistry of Aqua Ions: Synthesis, Structure, and Reactivity: A Tour Through the Periodic Table of the Elements. Chichester: Wiley. ISBN 0471970581.
- Silverman, Richard B. 2002. The Organic Chemistry of Enzyme-Catalyzed Reactions. San Diego: Academic Press. ISBN 0126437319.
- Testa, Bernard, and Joachim M. Mayer. 2003. Hydrolysis in Drug and Prodrug Metabolism: Chemistry, Biochemistry, and Enzymology. Weinheim: Wiley-VCH. ISBN 390639025X.
External links
All links retrieved January 22, 2018.
- An Explanation of the Process Hydrolysis. the balance.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.