Gamma ray
Gamma rays (γ rays) is the popular name applied to gamma radiation, the most energetic form of electromagnetic radiation and thus the electromagnetic radiation with the shortest wavelengths ( meters) and highest frequencies (Failed to parse (Conversion error. Server ("https://wikimedia.org/api/rest_") reported: "Cannot get mml. Server problem."): {\displaystyle 3x10^{19}-3x10^{25}} cycles per second).
By comparison, visible light occupies the middle range of the electromagnetic spectrum with moderate frequencies and radio waves occupy the low range with the lowest frequencies and longest wavelengths.
Gamma rays of specific frequencies are produced during electron-positron annihilation, radioactive decay (nuclear disintegration), and as synchrotron radiation from elementary particles or nuclei accelerated in high energy regions of the universe. Due to their high energy content, gamma rays can cause serious damage when absorbed by living cells.
This type of radiation also has a number of useful applications. For example, it is used to sterilize medical equipment, remove bacteria from foods, and maintain the freshness of fruits and vegetables. Gamma rays are also used for diagnostics in nuclear medicine and for performing what is called gamma-knife surgery to treat some types of cancer.
Properties
Matter interaction
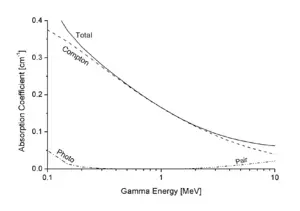
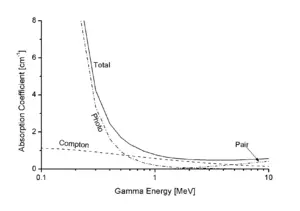
When a gamma ray passes through matter, the probability for absorption in a thin layer is proportional to the thickness of that layer. This leads to an exponential decrease of intensity with thickness.
- Failed to parse (Conversion error. Server ("https://wikimedia.org/api/rest_") reported: "Cannot get mml. Server problem."): {\displaystyle I(d)=I_{0}\cdot e^{-\mu d}}
Here, μ is the absorption coefficient, measured in cm−1, and d is the thickness of the material in cm. Also, μ = n×σ, where n is the number of atoms per cm3 in the material, and σ is the absorption cross section in cm2.
In passing through matter, gamma radiation ionizes atoms in that matter via three main processes: directly through the photoelectric effect and Compton scattering and indirectly through pair production.
- Photoelectric Effect: This describes the case in which a photon of gamma radiation interacts with and transfers its energy to an atomic electron giving that electron enough of an energy boost for it to escape from the atom. The kinetic energy of the resulting photoelectron is equal to the energy of the incident gamma photon minus the binding energy of the electron. The photoelectric effect is the dominant energy transfer mechanism for X-ray and gamma-ray photons with energies below 50 keV (thousand electron volts), but it is much less important at higher energies.
- Compton Scattering: In this phenomenon an incident gamma photon carries such an excess of the amount of energy required to induce the escape of an electron as a photoelectron, that the gamma photon's interaction induces release of the electron while the remainder of the photon's energy is emitted as a new, lower energy gamma photon with an emission direction different from that of the incident gamma photon. The probability of Compton scatter decreases with increasing photon energy. Compton scattering is thought to be the principal absorption mechanism for gamma rays in the intermediate energy range 100 keV to 10 MeV (megaelectronvolts), an energy range comprising most of the gamma radiation produced in a nuclear explosion. Compton scattering is relatively independent of the atomic number of the absorbing material.
- Pair Production: A photon of gamma radiation of a sufficiently high energy penetrating inside the atom close to the intense fields immediately surrounding the nucleus can be spontaneously converted into the mass of an electron-positron pair. A positron is the anti-matter equivalent of an electron; it has the same mass as an electron, but it has a positive charge equal in strength to the negative charge of an electron. Energy in excess of the equivalent rest mass of the two particles (1.02 MeV) appears as the kinetic energy of the pair and the recoil nucleus. The positron has a very short lifetime (if immersed in matter) (about 10-8 seconds). At the end of its range, it combines with a free electron through electron-positron annihilation. The entire mass of these two particles is then converted into two gamma photons of 0.51 MeV energy each.
The secondary electrons (or positrons) produced in any of these three processes frequently have enough energy to produce many ionizations up to the end of range.
The exponential absorption described above holds, strictly speaking, only for a narrow beam of gamma rays. If a wide beam of gamma rays passes through a thick slab of concrete, the scattering from the sides reduces the absorption.
Gamma decay
The wavelengths of gamma radiation are on the scale of the atomic nucleus, as shown in the diagram below.
Gamma rays are often produced alongside other forms of radiation such as alpha particles (helium nuclei comprising 2 protons and 2 neutrons) or beta particles (electrons). When a nucleus emits an α or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray in much the same way that an atomic electron can jump to a lower level by emitting visible light or ultraviolet radiation.
Gamma rays, x-rays, visible light, and UV rays are all forms of electromagnetic radiation. The only difference is the frequency and hence the energy of the photons. Gamma rays are the most energetic. An example of gamma ray production follows.
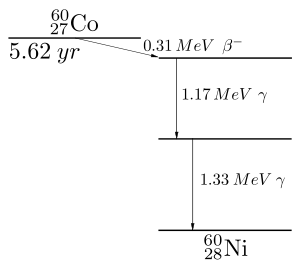
First 60Co decays to excited 60Ni by beta decay:
- Failed to parse (Conversion error. Server ("https://wikimedia.org/api/rest_") reported: "Cannot get mml. Server problem."): {\displaystyle {}^{60}{\hbox{Co}}\;\to \;^{60}{\hbox{Ni*}}\;+\;e^{-}\;+\;{\overline {\nu }}_{e}.}
Then the 60Ni drops down to the ground state (see nuclear shell model) by emitting two gamma rays in succession:
- Failed to parse (Conversion error. Server ("https://wikimedia.org/api/rest_") reported: "Cannot get mml. Server problem."): {\displaystyle {}^{60}{\hbox{Ni*}}\;\to \;^{60}{\hbox{Ni}}\;+\;\gamma .}
Gamma rays of 1.17 MeV and 1.33 MeV are produced.
Another example is the alpha decay of americium-241)(241Am) to form neptunium-237(237Np); this alpha decay is accompanied by gamma emission. In some cases, the gamma emission spectrum for a nucleus (daughter nucleu) is quite simple, (eg 60Co/60Ni) while in other cases, such as with (241Am/237Np and 192Ir/192Pt), the gamma emission spectrum is complex, revealing that a series of nuclear energy levels can exist. The fact that an alpha spectrum can have a series of different peaks with different energies reinforces the idea that several nuclear energy levels are possible.
Because a beta decay (loss of an electron from the nucleus after a neutron fragments into a proton, electron, neutrino) is accompanied by the emission of a neutrino which also carries energy away, the beta spectrum does not have sharp lines, but instead is a broad peak. Hence from beta decay alone it is not possible to probe the different energy levels found in the nucleus.
In optical spectroscopy, it is well known that an entity which emits light can also absorb light at the same wavelength (photon energy). For instance, a sodium flame can emit yellow light as well as absorb the yellow light from a sodium vapor lamp. In the case of gamma rays, this can be seen in Mössbauer spectroscopy. Here, a correction for the energy lost by the recoil of the nucleus is made and the exact conditions for gamma ray absorption through resonance can be attained.
This is similar to effects seen in optical spectroscopy that are explained by the Franck-Condon Principle.
Shielding
When selecting material for shielding from gamma radiation, one must take into account the fact that gamma rays are better absorbed by materials with high atomic number and high density. Also, the higher the energy of the gamma radiation, the thicker the shielding required. Materials for shielding from gamma rays are typically characterized by the thickness required to reduce the intensity of the gamma radiation by one half (the half value layer or HVL). For example, gamma rays that require one cm (0.4 inches) of lead to reduce their intensity by 50 percent will also have their intensity reduced in half by six cm (2.5 inches) of concrete or nine cm (3.5 inches) of packed dirt.
Uses
Because the wavelength of gamma radiation is so short, a single incident photon can impart significant damage to a living cell. This property means that gamma radiation is often used to kill living organisms, in a process called irradiation. Applications of this include sterilizing medical equipment (as an alternative to autoclaves or chemical means), removing decay-causing bacteria from many foodstuffs or preventing fruit and vegetables from sprouting to maintain freshness and flavor.
As a form of ionizing radiation gamma radiation has the ability to effect molecular changes, which means that the radiation can cause cancer when DNA is affected. The molecular changes can also be used to alter the properties of semi-precious stones, and is often used to change white topaz into blue topaz.
Despite their cancer-causing properties, gamma rays are also used to treat some types of cancer. In the procedure called gamma-knife surgery, multiple concentrated beams of gamma rays are directed on the growth in order to kill the cancerous cells. The beams are aimed from different angles to focus the radiation on the growth while minimizing damage to the surrounding tissues.
Gamma rays are also used for diagnostic purposes in nuclear medicine. Several gamma-emitting radioisotopes are used, one of which is technetium-99m. When administered to a patient, a gamma camera can be used to form an image of the radioisotope's distribution by detecting the gamma radiation emitted. Such a technique can be employed to diagnose a wide range of conditions (such as the spread of cancer to the bones).
Gamma ray detectors are also starting to be used in Pakistan as part of the Container Security Initiative (CSI). These US$5 million machines are advertised to scan 30 containers per hour. The objective of this technique is to pre-screen merchant ship containers before they enter U.S. ports.
Health effects
The gamma radiation is the most dangerous form of radiation emitted by a nuclear explosion because of the difficulty in stopping it. Gamma radiation is not stopped by the skin.
It can induce DNA alteration by interfering with the genetic material of the cell. DNA double-strand breaks are generally accepted to be the most biologically significant lesion by which ionizing radiation causes cancer and hereditary disease.[1].
A study done on Russian nuclear workers exposed to external whole-body gamma radiation at high cumulative doses shows the link between radiation exposure and death from leukemia, lung, liver, skeletal and other solid cancers.[2].
In combination with a thermal burn, gamma rays can reinforce the damage of thermal burn injuries and induce an immunosuppressive effect.[3][4]
Body response
After gamma irradiation, and the breaking of the DNA double-strands, the cells can repair the damaged genetic material in the limit of its capability.
However, a study of Rothkamm and Lobrich has shown that the repairing works relatively better after short high-dose exposure than during chronic low-dose exposure.[5]
It could mean that a chronic low-dose exposure could not be fought by the body.
Risk assessment
The natural outdoor exposure in Great Britain is in the range 20-40 nano-Sieverts/hour.[6] Natural cumulative exposure to gamma rays is about one to two milli-Sievert (mSv) per year, and the average total amount of radiation received in one year per inhabitant in the USA is 3.6 mSv.[7]
By comparison, the radiation dose from chest radiography is a fraction of the annual naturally occurring background radiation dose,[8] and the dose from fluoroscopy of the stomach is, at most, 0.05 Sv on the skin of the back.
For acute full-body equivalent doses, one Sv causes slight blood changes, two - five Sv causes nausea, hair loss, hemorrhaging and will cause death in many cases. More than 3 Sv will lead to death in less than two months in more than 80 percent of cases, and much over 4 Sv is more likely than not to cause death (see Sievert).
For low dose exposure, for example among nuclear workers, who receive an average radiation dose of 19mSv, the risk of dying from cancer (excluding leukemia) increases by two percent. For a dose of 100mSv, that risk increase is at 10 percent. By comparison, it was 32 percent for the Atom Bomb survivors.[9].
See also
- Alpha decay
- Beta decay
- Radiation therapy
- Radioactive decay
- Food irradiation
- X-ray
Notes
- ↑ K. Rothkamm 2003. "Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses." Proceedings of the National Academy of Science of the USA 100(9): 5057-5062.
- ↑ D. L. Shilnikova et al. 2003. "Cancer mortality risk among workers at the Mayak nuclear complex." Radiation Research 159(6): 787-798.
- ↑ X. Z. Ran et al. 1998. "Effects of combined radiation and thermal burn injury on the survival of skin allograft and immune function in." Chinese Medical Journal 111(7): 634-637.
- ↑ K. Randall et al. 1992. "The effect of whole-body gamma-irradiation on localized beta-irradiation-induced skin reactions in mice." International Journal of Radiation Biology 62(6): 729-733.
- ↑ Rothkamm 2003.
- ↑ Keys facts about radioactivity. Department for Environment, Food and Rural Affairs (Defra) UK.
- ↑ 1993. "United Nations Scientific Committee on the Effects of Atomic Radiation Annex E: Medical radiation exposures." Sources and Effects of Ionizing. 249.
- ↑ 1987. U.S. National Council on Radiation Protection and Measurements. NCRP Report 93: 53-55.
- ↑ Cancer risk following low doses of ionizing radiation - a 15 country study. IARC. Retrieved October 14, 2007.
ReferencesISBN links support NWE through referral fees
- Cheng, K. S. and G. E. Romero. 2004. Cosmic Gamma-Ray Sources. New York, NY: Springer. ISBN 1402022557.
- Gilmore, Gordon and John D. Hemingway. Practical Gamma-Ray Spectrometry. Hoboken, NJ: Wiley, 1995. ISBN 0471951501.
- Kelly, K. Radiation may have positive effects on health: study—Low, chronic doses of gamma radiation had beneficial effects on meadow voles. University of Toronto, 2005.
- van Putten, Maurice H.P.M. Gravitational Radiation, Luminous Black Holes and Gamma-Ray Burst Supernovae. Cambridge, NY: Cambridge University Press, 2006. ISBN 0521849608.
External links
All links retrieved April 17, 2024.
| |||||||||||||||||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.