Auxin
Auxins are a class of naturally occuring or synthetic organic (carbon-containing) plant growth substances (often called phytohormones or plant hormones) that increase, in low concentrations, the rate of cell elongation in stems, among other influences. All plants have auxins, and the prominent auxin indole-3-acetic acid (IAA) is found in almost all plants examined for auxins, usually in minute amounts.
Auxins play an essential role in coordination of many growth and behavioral processes in the plant life cycle. In addition to the definitive function of shoot elongation under proper conditions, auxins also influence phototropism (growth of stems toward light), inhibit cell elongation in roots, impact cell differentiation, inhibit lateral branching, and can impact development of flowers and fruit.
The special role of human beings relative to nature has led to a desire to investigate auxins and creatively use this knowledge in practical agricultural purposes. One such use is in the production of synthetic auxins that can act as herbicides. These can disrupt the natural auxin balance in plants, causing abnormal growth of mature cells and preventing the formation of new cells (Davy and Buchner 2006). Auxins used in high concentrations likewise can work as herbicides. On the other hand, one such synthetic auxin, 2,4,5-Trichlorophenoxyacetic acid (2,4,5-T), one of the active agents in Agent Orange, used in Vietnam, has an unavoidable dioxin contaminant identified as harmful to human beings.
Overview
Auxins, the first hormones to be discovered, have been demonstrated to be a basic coordinative signal of plant development. Davy and Buchner (2006), who state they can be thought of as a plant's replacement for the nervous system, note that they can regulate other hormones, regulate all stages of plant development, and act as a balancer for the plant's development of organs and other structures.
Auxins' pattern of active transport through the plant is complex, and auxins typically act in concert with (or opposition to) other plant hormones. For example, the ratio of auxin to cytokinin in certain plant tissues determines initiation of root versus shoot buds. As a result, a plant can (as a whole) react on external conditions and adjust to them, without requiring a nervous system.
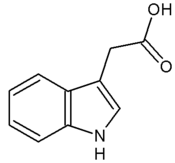
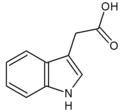
The most important member of the auxin family is indole-3-acetic acid (IAA). It generates the majority of auxin effects in intact plants, and is the most potent native auxin. However, molecules of IAA are chemically labile in aqueous solution, so IAA cannot be applied commercially as a plant growth regulator.
There are two basic groups of auxins.
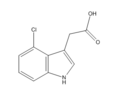
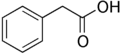
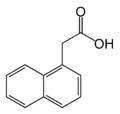
- Naturally-occurring auxins include 4-chloro-indoleacetic acid, phenylacetic acid (PAA), and indole-3-butyric acid (IBA).
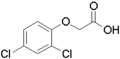
- Synthetic auxin analogs include 1-naphthaleneacetic acid (NAA), 2,4-dichlorophenoxyacetic acid (2,4-D), and others.
Auxins are often used to promote initiation of root growth and are the active ingredient of the commercial preparations used in horticulture to root stem cuttings. They can also be used to promote uniform flowering, to promote fruit set, and to prevent premature fruit drop.
Used in high doses, auxin stimulates the production of ethylene. Excess ethylene can inhibit elongation growth, cause leaves to fall (leaf abscission), and even kill the plant.
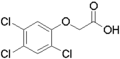
Some synthetic auxins such as 2,4-D and 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) can be used as herbicides. Broad-leaf plants (dicots) such as dandelions are much more susceptible to auxins than narrow-leaf plants (monocots) like grass and cereal crops.
Hormonal activity
Auxins coordinate development at all levels of plants, from the cellular level to organs and ultimately the whole plant.
Molecular mechanisms
Auxins directly stimulate or inhibit the expression of specific genes. Auxin induces transcription by targeting for degradation members of the Aux/IAA family of transcriptional repressor proteins, The degradation of the Aux/IAAs leads to the derepression of ARF-mediated transcription. Aux/IAAs are targeted for degradation by ubiquitination, catalysed by an SCF-type ubiquitin-protein ligase.
In 2005, it was demonstrated that the F-box protein TIR1, which is part of the ubiquitin ligase complex SCFTIR1, is an auxin receptor. Upon auxin binding, TIR1 recruits specific transcriptional repressors (the Aux/IAA repressors) for ubiquitination by the SCF complex. This marking process leads to the degradation of the repressors by the proteasome, alleviating repression and leading to specific gene expression in response to auxins.
On a cellular level
On the cellular level, auxin is essential for cell growth, affecting both cell division and cellular expansion. Depending on the specific tissue, auxin may promote axial elongation (as in shoots), lateral expansion (as in root swelling), or isodiametric expansion (as in fruit growth). In some cases (coleoptile growth), auxin-promoted cellular expansion occurs in the absence of cell division. In other cases, auxin-promoted cell division and cell expansion may be closely sequenced within the same tissue (root initiation, fruit growth). In a living plant, it appears that that auxins and other plant hormones nearly always interact to determine patterns of plant development.
According to the "acid growth theory," auxins may directly stimulate the early phases of cell elongation by causing responsive cells to actively transport hydrogen ions out of the cell, thus lowering the pH around cells. This acidification of the cell wall region activates enzymes known as expansins, which break bonds in the cell wall structure, making the cell wall less rigid. When the cell wall is partially degraded by the action of auxins, this now-less-rigid wall is expanded by the pressure coming from within the cell, especially by growing vacuoles.
However, the acid growth theory does not by itself account for the increased synthesis and transport of cell wall precursors and secretory activity in the Golgi system that accompany and sustain auxin-promoted cell expansion.
Organ patterns
Growth and division of plant cells together result in growth of tissue, and specific tissue growth contributes to the development of plant organs. Growth of cells contributes to the plant's size, but uneven localized growth produces bending, turning, and directionalization of organs, for example, stems turning toward light sources (phototropism), growth of roots in response to gravity (gravitropism), and other tropisms.
Organization of the plant
As auxins contribute to organ shaping, they are also fundamentally required for proper development of the plant itself. Without hormonal regulation and organization, plants would be merely proliferating heaps of similar cells. Auxin employment begins in the embryo of the plant, where directional distribution of auxin ushers in subsequent growth and development of primary growth poles, then forms buds of future organs. Throughout the plant's life, auxin helps the plant maintain the polarity of growth and recognize where it has its branches (or any organ) connected.
An important principle of plant organization based upon auxin distribution is apical dominance, which means that the auxin produced by the apical bud (or growing tip) diffuses downwards and inhibits the development of ulterior lateral bud growth, which would otherwise compete with the apical tip for light and nutrients. Removing the apical tip and its suppressive hormone allows the lower dormant lateral buds to develop, and the buds between the leaf stalk and stem produce new shoots, which compete to become the lead growth. This behavior is used in pruning by horticulturists.
Uneven distribution of auxin: To cause growth in the required domains, it is necessary that auxins be active preferentially in them. Auxins are not synthesized everywhere, but each cell retains the potential ability to do so, and only under specific conditions will auxin synthesis be activated. For that purpose, not only do auxins have to be translocated toward those sites where they are needed but there has to be an established mechanism to detect those sites. Translocation is driven throughout the plant body primarily from peaks of shoots to peaks of roots. For long distances, relocation occurs via the stream of fluid in phloem vessels, but, for short-distance transport, a unique system of coordinated polar transport directly from cell to cell is exploited. This process of polar auxin transport is directional and very strictly regulated. It is based in uneven distribution of auxin efflux carriers on the plasma membrane, which send auxins in the proper direction.
A 2006 study showed plant-specific pin-formed (PIN) proteins are vital in transporting auxin. PINs also regulate auxin efflux from mammalian and yeast cells (Petraek et al. 2006).
Locations
- In shoot (and root) meristematic tissue
- In young leaves
- In mature leaves in very tiny amounts
- In mature root cells in even smaller amounts
- Transported throughout the plant more prominently downward from the shoot apices
Summary of effects
- Cell elongation. Auxin stimulates cell elongation. It stimulates factors, such as elastins, to loosen the cell walls and allow the elongation. A basic condition to define a substance as an auxin is its ability to increase the rate of stem elongation under proper conditions; however, auxins provide a diversity of functions.
- Cell division. Auxin stimulates cell division if cytokinins are present. When auxin and cytokinin was applied to callus, rooting can be generated if the auxin concentration is higher than cytokinin concentration while xylem tissues can be generated when the auxin concentration is equal to the cytokinins.
- Tropisms. Auxin participates in phototropism, geotropism, hydrotropism, and other developmental changes. The uneven distribution of auxin, due to environmental cues (for example, unidirectional light and gravity force), results in uneven plant tissue growth.
- Sugar and mineral accumulation. Auxin induces sugar and mineral accumulation at the site of application.
- Vascular tissue impacts. Auxin induces formation and organization of phloem and xylem. When the plant is wounded, the auxin can induce the Cell differentiation and regeneration of the vascular tissues.
- Root growth and development. Auxin induces new root formation by breaking root apical dominance induced by cytokinins. However, high concentrations of auxin inhibit root elongation and instead enhance adventitious root formation. Removal of the root tip can lead to inhibition of secondary root formation.
- Apical Dominance. Auxin induces shoot apical dominance; the axillary buds are inhibited by auxin. When the apex of the plant is removed, the inhibitory effect is removed and the growth of lateral buds is enhanced as a high concentration of auxin directly stimulates ethylene synthesis in lateral buds causes inhibition of its growth and potentiation of apical dominance.
- Ethylene biosynthesis. In low concentrations, auxin can inhibit ethylene formation and transport of precursor in plants; however, high concentrations of auxin can induce the synthesis of ethylene.
- Leaf fall. It also inhibits abscission prior to formation of abscission layer and thus inhibits senescence of leaves.
- Fruit growth. Auxin delays fruit senescence. It is required for fruit growth. When seeds are removed from strawberries, fruit growth is stopped; exogenous auxin stimulates the growth in seed removed fruits. For fruit with unfertilized seeds, exogenous auxin results in parthenocarpy ("virgin-fruit" growth).
- Flowering. Auxin plays a minor role in the initiation of flowering. It can delay the senescence of flowers in low concentrations.
Herbicide manufacture
The defoliant Agent Orange was a mix of 2,4-D and 2,4,5-T. 2,4-D is still in use and is thought to be safe, but 2,4,5-T was more or less banned in the United States by the Environmental Protection Agency (EPA) in 1979. The dioxin TCDD is an unavoidable contaminant produced in the manufacture of 2,4,5-T. As a result of the integral dioxin contamination, 2,4,5-T has been implicated in leukemia, miscarriages, birth defects, liver damage, and other complications. Agent Orange was sprayed in Vietnam as a defoliant to deny ground cover to the Vietnamese army.
"Killex" is another herbicide, composed of 2,4-D and Dicamba.
Gallery of auxins
Gallery of native auxins
Gallery of synthetic auxins
ReferencesISBN links support NWE through referral fees
- Davy, J., and R. P. Bucher. 2006. The story behind the new herbicide MilestoneTM. Livestock & Land News April 2006.
- Petrášek, J., J. Mravec, R. Bouchard, J. J. Blakeslee, M. Abas, D. Seifertová, J. Winiewska, Z. Tadele, M. Kubeš, M. Čovanová, P. Dhonukshe, P. Skůpa, E. Benková, L. Perry, P. Křeček, O. Ran Lee, G. R. Fink, M. Geisler, A. S. Murphy, C. Luschnig, E. Zažímalová, and J. Frim. 2006. PIN proteins perform a rate-limiting function in cellular auxin efflux. Science 312(5775): 914-918. Retrieved May 26, 2012.
- Taiz, L., and E. Zeiger. 2006. Chapter 19. Auxin: The growth hormone. Plant Physiology Online. Retrieved May 10, 2007.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.