Difference between revisions of "Particulate" - New World Encyclopedia
(imported article from Wikipedia) |
(claim, fix, edits) |
||
| Line 1: | Line 1: | ||
| + | {{Claimed}} | ||
{{Pollution}} | {{Pollution}} | ||
| − | '''Particulates''', alternatively referred to as '''particulate matter (PM), [[aerosol]]s''' or '''fine particles''', are tiny particles of solid or liquid suspended in a gas. They range in size from less than 10 [[ | + | |
| + | '''Particulates''', alternatively referred to as '''particulate matter (PM), [[aerosol]]s''', or '''fine particles''', are tiny particles of solid or liquid suspended in a gas. They range in size from less than 10 [[nanometer]]s to more than 100 [[micrometer]]s in diameter. The notation PM<sub>10</sub> is used to describe particles of 10 micrometers or less and PM<sub>2.5</sub> represents particles less than 2.5 micrometers in aerodynamic diameter; other numeric values may also be used. This range of sizes represent scales from a gathering of a few [[molecule]]s to the size where the particles no longer can be carried by the gas. Sources of particulate matter can be anthropogenic or natural. | ||
Some particulates occur naturally, originating from [[volcano]]es, [[dust storm]]s, [[forest fire|forest]] and [[grassland]] fires, living vegetation, and [[sea spray]]. Human activities, such as the burning of [[fossil fuel]]s also generate aerosols. Averaged over the globe, ''anthropogenic'' aerosols—those made by human activities—currently account for about 10 percent of the total amount of aerosols in our atmosphere. | Some particulates occur naturally, originating from [[volcano]]es, [[dust storm]]s, [[forest fire|forest]] and [[grassland]] fires, living vegetation, and [[sea spray]]. Human activities, such as the burning of [[fossil fuel]]s also generate aerosols. Averaged over the globe, ''anthropogenic'' aerosols—those made by human activities—currently account for about 10 percent of the total amount of aerosols in our atmosphere. | ||
| + | |||
| + | The field of [[aerosol science]] and technology has grown in response to the need to understand and control natural and manmade aerosols. | ||
===Sources=== | ===Sources=== | ||
| − | [[Image:Aerosol-India.jpg|thumb|260px|right|Aerosol pollution over Northern India and Bangladesh - Photo: [http://visibleearth.nasa.gov/cgi-bin/viewrecord?10980 NASA]]] | + | [[Image:Aerosol-India.jpg|thumb|260px|right|Aerosol pollution over Northern India and Bangladesh - Photo: [http://visibleearth.nasa.gov/cgi-bin/viewrecord?10980 NASA].]] |
| − | There are both natural and human sources of atmospheric particulates. The biggest natural sources are [[dust]], volcanoes, and forest fires. Sea spray is also a large source of particles though most of these fall back to the ocean close to where they were emitted. The biggest human sources of particles are combustion sources, mainly the burning of fuels in [[internal combustion engine]]s in [[automobile]]s and [[power plant]]s, and wind blown dust from [[construction]] sites and other land areas where the water or vegetation has been removed. Some of these particles are emitted directly to the atmosphere | + | |
| − | as gases and form particles in the atmosphere | + | There are both natural and human sources of atmospheric particulates. The biggest natural sources are [[dust]], volcanoes, and forest fires. Sea spray is also a large source of particles though most of these fall back to the ocean close to where they were emitted. The biggest human sources of particles are combustion sources, mainly the burning of fuels in [[internal combustion engine]]s in [[automobile]]s and [[power plant]]s, and wind blown dust from [[construction]] sites and other land areas where the water or vegetation has been removed. Some of these particles are emitted directly to the atmosphere ''(primary [[Emission standard|emissions]])'' and some are emitted |
| + | as gases and form particles in the atmosphere ''(secondary emissions)''. | ||
:"In Europe and the United States, particulate emissions from vehicles are expected to decline over the next decade. For example, by 2005, the European Union will introduce more stringent standards for particulate emissions from light duty vehicles of 0.025 grams per kilometer [0.04 grams per mile]. | :"In Europe and the United States, particulate emissions from vehicles are expected to decline over the next decade. For example, by 2005, the European Union will introduce more stringent standards for particulate emissions from light duty vehicles of 0.025 grams per kilometer [0.04 grams per mile]. | ||
| − | :The state of California is implementing an even more restrictive standard in 2004, allowing only 0.006 grams per kilometer [0.01 grams per mile] of particulate emissions. Even if the California standard were introduced worldwide, says Jacobson, diesel cars may still warm the climate more than gasoline cars over 13 to 54 years. New particle traps being introduced by some European automobile manufacturers in their diesel cars appear to reduce [[black carbon]] emissions to 0.003 grams per kilometer [0.005 grams per mile], even below the California standard." [http://www.stanford.edu/dept/news/pr/02/jacobsonJGR1023.html] | + | :The state of California is implementing an even more restrictive standard in 2004, allowing only 0.006 grams per kilometer [0.01 grams per mile] of particulate emissions. Even if the California standard were introduced worldwide, says Jacobson, diesel cars may still warm the climate more than gasoline cars over 13 to 54 years. New particle traps being introduced by some European automobile manufacturers in their diesel cars appear to reduce [[black carbon]] emissions to 0.003 grams per kilometer [0.005 grams per mile], even below the California standard."<ref> [http://www.stanford.edu/dept/news/pr/02/jacobsonJGR1023.html] </ref> |
[[BlueTec]] is one technology developed to reduce particulate emissions from Diesel engines in order to meet strigent Californian standards. | [[BlueTec]] is one technology developed to reduce particulate emissions from Diesel engines in order to meet strigent Californian standards. | ||
| Line 18: | Line 23: | ||
The composition of aerosol particles depends on their source. | The composition of aerosol particles depends on their source. | ||
| − | Wind-blown [[mineral dust]] [http://www.grida.no/climate/ipcc_tar/wg1/168.htm#5221] tends to be made of | + | Wind-blown [[mineral dust]]<ref> [http://www.grida.no/climate/ipcc_tar/wg1/168.htm#5221] </ref> tends to be made of mineral [[oxide]]s and other material blown from the [[Earth's crust]]. This aerosol is |
| − | mineral [[oxide]]s and other material blown from the [[Earth's crust]] | ||
[[absorption (electromagnetic radiation)|light-absorbing]]. | [[absorption (electromagnetic radiation)|light-absorbing]]. | ||
| − | Sea salt [http://www.grida.no/climate/ipcc_tar/wg1/169.htm#5222] is considered the second largest contributor | + | Sea salt<ref>[http://www.grida.no/climate/ipcc_tar/wg1/169.htm#5222] </ref> is considered the second largest contributor in the global aerosol budget, and consists mainly of [[sodium chloride]] originated from [[sea spray]]. Other constituents of atmospheric sea salt reflect the composition of [[sea water]], and thus include [[magnesium]], [[sulfate]], [[calcium]], [[potassium]], and other ions. In addition, sea spray aerosols may contains organic compounds, which influence their chemistry. Sea salt does not [[absorption (electromagnetic radiation)|absorb]] light. |
| − | in the global aerosol budget, and consists mainly of [[sodium chloride]] originated from [[sea spray]] | ||
| − | constituents of atmospheric sea salt reflect the composition of [[sea water]], and thus include [[magnesium]], | ||
| − | [[sulfate]], [[calcium]], [[potassium]], | ||
| − | In addition, sea spray aerosols may contains organic compounds, which influence their chemistry. | ||
| − | Sea salt does not [[absorption (electromagnetic radiation)|absorb]]. | ||
| − | Secondary particles derive from the [[oxidation]] of primary gases such as [[sulfur oxide|sulfur]] and [[nitrogen oxide]]s into | + | Secondary particles derive from the [[oxidation]] of primary gases such as [[sulfur oxide|sulfur]] and [[nitrogen oxide]]s into [[sulfuric acid]] (liquid) and [[nitric acid]] (gaseous). |
| − | [[sulfuric acid]] (liquid) and [[nitric acid]] (gaseous). | + | The precursors for these aerosols, i.e. the gases from which they originate, may have an anthropogenic origin (from fossil fuel [[combustion]]) and a natural [[biogenic]] origin. In the presence of [[ammonia]], secondary aerosols often take the form of [[ammonium]] salts, such as [[ammonium sulfate]] and [[ammonium nitrate]] (both can be dry or in aqueous [[solution]]). In the |
| − | The precursors for these aerosols, i.e. the gases from which they originate, may have an anthropogenic | + | absence of ammonia, secondary compounds take an [[acid]]ic form, as sulfuric acid (liquid aerosol |
| − | origin (from fossil fuel [[combustion]]) and a natural [[biogenic]] origin. | + | droplets) and nitric acid (atmospheric gas). Secondary sulfate and nitrate aerosols are strong [[scattering|light-scatterers]].<ref> [http://www.grida.no/climate/ipcc_tar/wg1/172.htm#5226] </ref> This is mainly because the presence of sulphate and nitrate causes the aerosols to increase to a size that scatters light effectively. |
| − | In the presence of [[ammonia]], secondary aerosols often take the form of [[ammonium]] salts, | ||
| − | [[ammonium sulfate]] and [[ammonium nitrate]] (both can be dry or in aqueous [[solution]]) | ||
| − | absence of ammonia, secondary compounds take an [[acid]]ic form as sulfuric acid (liquid aerosol | ||
| − | droplets) and nitric acid (atmospheric gas). | ||
| − | Secondary sulfate and nitrate aerosols are strong [[scattering|light-scatterers]]. [http://www.grida.no/climate/ipcc_tar/wg1/172.htm#5226] This is mainly because the presence of sulphate and nitrate causes the aerosols to increase to a size that scatters light effectively. | ||
| − | [[Organic compound|Organic matter]] (OM) can be either primary or secondary, the latter part deriving from | + | [[Organic compound|Organic matter]] (OM) can be either primary or secondary, the latter part deriving from the oxidation of [[VOCs]]; organic material in the atmosphere may either be biogenic or [[anthropogenic]]. Organic matter influences the atmospheric [[electromagnetic radiation|radiation]] field by both [[scattering]] and [[absorption (electromagnetic radiation)|absorption]] of light. |
| − | the oxidation of [[VOCs]]; organic material in the atmosphere may either be biogenic or [[anthropogenic]]. | + | |
| − | Organic matter influences the atmospheric [[electromagnetic radiation|radiation]] field by both [[scattering]] | + | Another important aerosol type is constituted of [[soot|elemental carbon]] (EC, also known as |
| − | and [[absorption (electromagnetic radiation)|absorption]]. | + | ''[[black carbon]]'', BC): this aerosol type includes strongly light-absorbing material and is thought to yield large positive [[radiative forcing]]. Organic matter and elemental carbon together constitute the [[carbonaceous]] fraction of aerosols.<ref>[http://www.grida.no/climate/ipcc_tar/wg1/170.htm] </ref> |
| − | Another important aerosol type is | ||
| − | ''[[black carbon]]'', BC): this aerosol type includes strongly light-absorbing material and is thought to | ||
| − | yield large positive [[radiative forcing]]. | ||
| − | Organic matter and elemental carbon together constitute the [[carbonaceous]] fraction of aerosols. | ||
| − | [http://www.grida.no/climate/ipcc_tar/wg1/170.htm] | ||
The chemical composition of the aerosol directly affects how it interacts with solar radiation. The chemical constituents within the aerosol change the overall [[refractive index]]. The refractive index will determine how much light is scattered and absorbed. | The chemical composition of the aerosol directly affects how it interacts with solar radiation. The chemical constituents within the aerosol change the overall [[refractive index]]. The refractive index will determine how much light is scattered and absorbed. | ||
| Line 56: | Line 45: | ||
== Radiative forcing from aerosols == | == Radiative forcing from aerosols == | ||
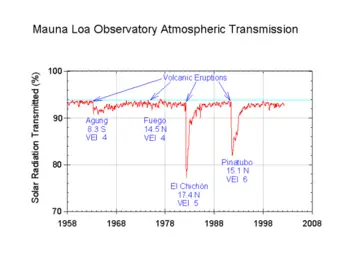
[[Image:Mauna Loa atmospheric transmission.png|thumb|350px|Solar radiation reduction due to volcanic eruptions]] | [[Image:Mauna Loa atmospheric transmission.png|thumb|350px|Solar radiation reduction due to volcanic eruptions]] | ||
| − | |||
| − | A graphic showing the contributions (at 2000, relative to pre-industrial) and uncertainties of various forcings is available [http://www.grida.no/climate/ipcc_tar/vol4/english/wg1figspm-3.htm here]. | + | Aerosols, natural and [[anthropogenic]], can affect the climate by changing the way [[Electromagnetic radiation|radiation]] is transmitted through the atmosphere. Direct observations of the effects of aerosols are quite limited so any attempt to estimate their global effect necessarily involves the use of computer models. The [[Intergovernmental Panel on Climate Change]], IPCC, says: ''While the radiative forcing due to [[greenhouse gas]]es may be determined to a reasonably high degree of accuracy... the uncertainties relating to aerosol radiative forcings remain large, and rely to a large extent on the estimates from global modelling studies that are difficult to verify at the present time.''<ref>[http://www.grida.no/climate/ipcc_tar/wg1/237.htm#678]. </ref> |
| + | |||
| + | A graphic showing the contributions (at 2000, relative to pre-industrial) and uncertainties of various forcings is available.<ref> [http://www.grida.no/climate/ipcc_tar/vol4/english/wg1figspm-3.htm here]. </ref> | ||
=== Sulphate aerosol === | === Sulphate aerosol === | ||
| − | Sulphate aerosol has two main effects, direct and indirect. The direct effect, via [[albedo]], is to cool the planet: the [[IPCC]]'s best estimate of the [[radiative forcing]] is -0.4 [[watt]]s per square meter with a range of -0.2 to -0.8 W/m² [http://www.grida.no/climate/ipcc_tar/wg1/232.htm] but there are substantial uncertainties. The effect varies strongly geographically, with most cooling believed to be at and downwind of major industrial centres. Modern [[climate model]]s attempting to deal with the [[attribution of recent climate change]] need to include sulfate forcing, which appears to account (at least partly) for the slight drop in global temperature in the middle of the 20th century. The indirect effect (via the aerosol acting as cloud condensation nuclei, [[Cloud condensation nuclei|CCN]], and thereby modifying the cloud properties) is more uncertain but is believed to be a cooling. | + | |
| + | Sulphate aerosol has two main effects, direct and indirect. The direct effect, via [[albedo]], is to cool the planet: the [[IPCC]]'s best estimate of the [[radiative forcing]] is -0.4 [[watt]]s per square meter with a range of -0.2 to -0.8 W/m²,<ref>[http://www.grida.no/climate/ipcc_tar/wg1/232.htm] </ref> but there are substantial uncertainties. The effect varies strongly geographically, with most cooling believed to be at and downwind of major industrial centres. Modern [[climate model]]s attempting to deal with the [[attribution of recent climate change]] need to include sulfate forcing, which appears to account (at least partly) for the slight drop in global temperature in the middle of the 20th century. The indirect effect (via the aerosol acting as cloud condensation nuclei, [[Cloud condensation nuclei|CCN]], and thereby modifying the cloud properties) is more uncertain but is believed to be a cooling. | ||
=== Black carbon === | === Black carbon === | ||
[[Black carbon]] (BC), or Carbon Black, or Elemental Carbon (EC), often called soot, is composed of pure carbon clusters, skeleton balls and [[Fullerene|buckyballs]], and is one of the most important absorbing aerosol species in the atmosphere. It should be distinguished from Organic Carbon (OC): clustered or aggregated organic molecules on their own or permeating an EC buckyball. BC from fossil fuels is estimated by the IPCC in the Fourth Assessment Report of the IPCC, TAR, to contribute a global mean radiative forcing of +0.2 W/m² (was +0.1 W/m² in the Second Assessment Report of the IPCC, SAR), with a range +0.1 to +0.4 W/m². | [[Black carbon]] (BC), or Carbon Black, or Elemental Carbon (EC), often called soot, is composed of pure carbon clusters, skeleton balls and [[Fullerene|buckyballs]], and is one of the most important absorbing aerosol species in the atmosphere. It should be distinguished from Organic Carbon (OC): clustered or aggregated organic molecules on their own or permeating an EC buckyball. BC from fossil fuels is estimated by the IPCC in the Fourth Assessment Report of the IPCC, TAR, to contribute a global mean radiative forcing of +0.2 W/m² (was +0.1 W/m² in the Second Assessment Report of the IPCC, SAR), with a range +0.1 to +0.4 W/m². | ||
| − | All aerosols both [[Absorption (electromagnetic radiation)|absorb]] and [[scattering|scatter]] [[solar]] and terrestrial [[radiation]]. If a substance absorbs a significant amount of radiation, as well as scattering, we call it absorbing. This is quantified in the ''Single Scattering Albedo'' (SSA), the ratio of scattering alone to scattering plus absorption | + | All aerosols both [[Absorption (electromagnetic radiation)|absorb]] and [[scattering|scatter]] [[solar]] and terrestrial [[radiation]]. If a substance absorbs a significant amount of radiation, as well as scattering, we call it absorbing. This is quantified in the ''Single Scattering Albedo'' (SSA), the ratio of scattering alone to scattering plus absorption ''(extinction)'' of radiation by a particle. The SSA tends to unity if scattering dominates, with relatively little absorption, and decreases as absorption increases, becoming zero for infinite absorption. For example, sea-salt aerosol has an SSA of 1, as a sea-salt particle only scatters, whereas soot has an SSA of 0.23, showing that it is a major atmospheric aerosol absorber. |
| − | ==Health effects== | + | ==Health effects== |
| + | [[Image:Luftguete messstation.jpg|thumb|Air pollution measurement station in [[Emden]], [[Germany]].]] | ||
| − | + | The effects of inhaling particulate matter has been widely studied in humans and animals and include [[asthma]], [[lung cancer]], cardiovascular issues, and premature [[death]]. The size of the particle is a main determinant of where in the respiratory tract the particle will come to rest when inhaled. Larger particles are generally filtered in the nose and throat and do not cause problems, but particulate matter smaller than about 10 micrometers, referred to as ''PM<sub>10</sub>'', can settle in the bronchi and [[lung]]s and cause health problems. The 10 micrometer size does not represent a strict boundary between respirable and non-respirable particles, but has been agreed upon for monitoring of airborne particulate matter by most regulatory agencies. Similarly, particles smaller than 2.5 micrometers, ''PM<sub>2.5</sub>'', tend to penetrate into the gas-exchange regions of the lung, and very small particles (< 100 nanometers) may pass through the lungs to affect other organs. In particular, a study published in the ''Journal of the American Medical Association'' indicates that PM<sub>2.5</sub> leads to high plaque deposits in arteries, causing vascular inflammation and atherosclerosis — a hardening of the arteries that reduces elasticity, which can lead to heart attacks and other cardiovascular problems <ref name="pope"> | |
| − | The effects of inhaling particulate matter has been widely studied in humans and animals and include [[asthma]], [[lung cancer]], cardiovascular issues, and premature [[death]]. The size of the particle is a main determinant of where in the respiratory tract the particle will come to rest when inhaled. Larger particles are generally filtered in the nose and throat and do not cause problems, but particulate matter smaller than about 10 | ||
{{cite journal | {{cite journal | ||
| last = Pope | | last = Pope | ||
| Line 88: | Line 79: | ||
| accessdate = }} </ref>. Researchers suggest that even short-term exposure at elevated concentrations could significantly contribute to heart disease. | | accessdate = }} </ref>. Researchers suggest that even short-term exposure at elevated concentrations could significantly contribute to heart disease. | ||
| − | There is also evidence that particles smaller than 100 nanometres can pass through cell membranes. For example, particles may migrate into the brain. It has been suggested that particulate matter can cause similar brain damage as that found in [[Alzheimer's disease|Alzheimer]] patients. Particles emitted from modern [[diesel]] engines (commonly referred to as [[Diesel Particulate Matter]], or DPM) are typically in the size range of 100 nanometres | + | There is also evidence that particles smaller than 100 nanometres can pass through cell membranes. For example, particles may migrate into the brain. It has been suggested that particulate matter can cause similar brain damage as that found in [[Alzheimer's disease|Alzheimer]] patients. Particles emitted from modern [[diesel]] engines (commonly referred to as [[Diesel Particulate Matter]], or DPM) are typically in the size range of 100 nanometres ''(0.1 micrometres)''. In addition, these [[soot]] particles also carry [[carcinogen]]ic components like [[benzopyrene]]s adsorbed on their surface. It is becoming increasingly clear that the legislative limits for engines, which are in terms of emitted mass, are not a proper measure of the health hazard. One particle of 10 µm diameter has approximately the same mass as 1 million particles of 100 nm diameter, but it is clearly much less hazardous, as it probably never enters the human body - and if it does, it is quickly removed. Proposals for new regulations exist in some countries, with suggestions to limit the particle surface area or the particle number. |
The large number of deaths and other health problems associated with particulate pollution was first demonstrated in the early 1970s <ref name="lave"> | The large number of deaths and other health problems associated with particulate pollution was first demonstrated in the early 1970s <ref name="lave"> | ||
| Line 119: | Line 110: | ||
==Regulation== | ==Regulation== | ||
| − | + | ||
| − | Due to the health effects of particulate matter, maximum standards have been set by various governments. | + | Due to the health effects of particulate matter, maximum standards have been set by various governments. Many [[urban area]]s in the U.S. and Europe still exceed the particulate standards, though urban air on these continents has become cleaner, on average, with respect to particulates over the last quarter of the twentieth century.{{Fact|date=July 2007}} |
===United States=== | ===United States=== | ||
| + | |||
The [[United States Environmental Protection Agency]] (EPA) sets standards for PM<sub>10</sub> and PM<sub>2.5</sub> concentrations in urban air. (See [[National Ambient Air Quality Standards]].) EPA regulates primary particulate emissions and precursors to secondary emissions ([[NOx]], [[sulfur]], and [[ammonia]]). | The [[United States Environmental Protection Agency]] (EPA) sets standards for PM<sub>10</sub> and PM<sub>2.5</sub> concentrations in urban air. (See [[National Ambient Air Quality Standards]].) EPA regulates primary particulate emissions and precursors to secondary emissions ([[NOx]], [[sulfur]], and [[ammonia]]). | ||
| Line 131: | Line 123: | ||
|- | |- | ||
! | ! | ||
| − | ! Phase 1<br> | + | ! Phase 1<br/> |
| − | from | + | from 1 January 2005 |
| − | ! Phase 2¹<br> | + | ! Phase 2¹<br/> |
| − | from | + | from 1 January 2010 |
|- | |- | ||
||Yearly average | ||Yearly average | ||
| Line 140: | Line 132: | ||
||20 µg/m³ | ||20 µg/m³ | ||
|- | |- | ||
| − | ||Daily average (24-hour)<br> | + | ||Daily average (24-hour)<br/> |
allowed number of exceedences per year. | allowed number of exceedences per year. | ||
| − | ||50 µg/m³<br> | + | ||50 µg/m³<br/> |
35 | 35 | ||
| − | ||50 µg/m³<br> | + | ||50 µg/m³<br/> |
7 | 7 | ||
|} | |} | ||
| Line 153: | Line 145: | ||
{|class="wikitable" align="right" | {|class="wikitable" align="right" | ||
|- | |- | ||
| − | !colspan=2|Most Polluted World Cities by PM<ref>http://siteresources.worldbank.org/DATASTATISTICS/Resources/table3_13.pdf</ref> | + | !colspan=2|Most Polluted World Cities by PM<ref>[http://siteresources.worldbank.org/DATASTATISTICS/Resources/table3_13.pdf] </ref> |
|- | |- | ||
| − | !Particulate matter,<br> μg/m<sup>3</sup> (2004) | + | !Particulate matter,<br/> μg/m<sup>3</sup> (2004) |
!City | !City | ||
|- | |- | ||
| Line 189: | Line 181: | ||
|} | |} | ||
| − | The most concentrated particulate matter pollution tends to be in densely populated metropolitan areas in developing countries. | + | The most concentrated particulate matter pollution tends to be in densely populated metropolitan areas in developing countries. The primary cause is the burning of fossil fuels by transportation and industrial sources. |
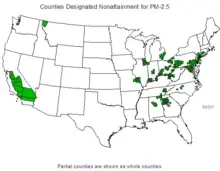
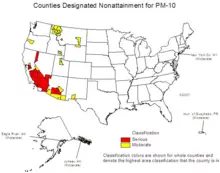
[[Image:US-PM2.5-nonattainment-2007-06.png|thumb|left|220px|U.S. counties violating national PM<sub>2.5</sub> standards, roughly correlated with population density.]] | [[Image:US-PM2.5-nonattainment-2007-06.png|thumb|left|220px|U.S. counties violating national PM<sub>2.5</sub> standards, roughly correlated with population density.]] | ||
| Line 196: | Line 188: | ||
{{clear}} | {{clear}} | ||
| − | == | + | == See also == |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
* [[Aerosol science]] | * [[Aerosol science]] | ||
* [[Air pollution]] | * [[Air pollution]] | ||
| Line 218: | Line 195: | ||
* [[Criteria air contaminants]] | * [[Criteria air contaminants]] | ||
* [[Deposition (Aerosol physics)|Deposition]] | * [[Deposition (Aerosol physics)|Deposition]] | ||
| − | |||
* [[Dust]] | * [[Dust]] | ||
* [[Global dimming]] | * [[Global dimming]] | ||
* [[Global warming]] | * [[Global warming]] | ||
| − | |||
* [[Haze]] | * [[Haze]] | ||
| − | |||
* [[Radiological weapon]] | * [[Radiological weapon]] | ||
| − | * [[ | + | * [[Cloud]] |
* [[Fog]] | * [[Fog]] | ||
* [[pea soup#pea soup fog|"Pea soup" fog]] | * [[pea soup#pea soup fog|"Pea soup" fog]] | ||
| + | |||
| + | == Notes == | ||
| + | <references/> | ||
| + | |||
| + | == References == | ||
| + | |||
| + | * [http://earthobservatory.nasa.gov/Library/Aerosols/ Article at earthobservatory.nasa.gov describing the possible influence of aerosols on the climate] | ||
| + | * [http://www.grida.no/climate/ipcc_tar/wg1/160.htm The Intergovernmental Panel on Climate Change (the principal international scientific body on climate change) chapter on atmospheric aerosols and their radiative effects] | ||
| + | * [http://insideepa.com/secure/insider_display.asp?f=epa_2001.ask&docid=142006_links] InsideEPA.com, Study Links Air Toxics To Heart Disease In Mice Amid EPA Controversy | ||
| + | * Preining, Othmar and E. James Davis (eds.), "History of Aerosol Science," Österreichische Akademie der Wissenschaften, ISBN 3700129157 (pbk.) | ||
| + | * G Invernizzi et al., ''Particulate matter from tobacco versus diesel car exhaust: an educational perspective''. [http://tc.bmjjournals.com/cgi/reprint/13/3/219?ijkey=330b5aea15a8e36fcc2f4208cf99da58d84150f3 Tobacco Control 13, S.219-221] (2004) | ||
| + | * Sheldon K.Friedlander, "Smoke, Dust and Haze." | ||
| + | *JEFF CHARLTON ''Pandemic planning: a review of respirator and mask protection levels.'' | ||
== External links == | == External links == | ||
| − | + | ||
* [http://www.npi.gov.au/database/substance-info/profiles/69.html National Pollutant Inventory - Particulate matter fact sheet] | * [http://www.npi.gov.au/database/substance-info/profiles/69.html National Pollutant Inventory - Particulate matter fact sheet] | ||
*WHO-Europe reports: [http://www.who.dk/document/e79097.pdf Health Aspects of Air Pollution (2003)] (PDF) and "[http://www.euro.who.int/document/E82790.pdf Answer to follow-up questions from CAFE (2004)] (PDF) | *WHO-Europe reports: [http://www.who.dk/document/e79097.pdf Health Aspects of Air Pollution (2003)] (PDF) and "[http://www.euro.who.int/document/E82790.pdf Answer to follow-up questions from CAFE (2004)] (PDF) | ||
| Line 242: | Line 229: | ||
* [http://siteresources.worldbank.org/INTDATASTA/64199955-1178226923002/21322619/LGDB2007.pdf Little Green Data Book 2007], World Bank. Lists C02 and PM statistics by country. | * [http://siteresources.worldbank.org/INTDATASTA/64199955-1178226923002/21322619/LGDB2007.pdf Little Green Data Book 2007], World Bank. Lists C02 and PM statistics by country. | ||
* [http://econ.worldbank.org/WBSITE/EXTERNAL/EXTDEC/EXTRESEARCH/0,,contentMDK:20785646~pagePK:64214825~piPK:64214943~theSitePK:469382,00.html Air Pollution in World Cities (PM10 Concentrations)] | * [http://econ.worldbank.org/WBSITE/EXTERNAL/EXTDEC/EXTRESEARCH/0,,contentMDK:20785646~pagePK:64214825~piPK:64214943~theSitePK:469382,00.html Air Pollution in World Cities (PM10 Concentrations)] | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | [[Category:Physical sciences]] | |
| − | [[ | + | [[Category:Environmental science]] |
| − | [[ | + | |
| − | + | {{credit|160206296}} | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
Revision as of 23:53, 8 October 2007
| |||
| Air pollution | |||
| Acid rain • Air Pollution Index • Air Quality Index • Atmospheric dispersion modeling • Chlorofluorocarbon • Global dimming • Global warming • Haze • Indoor air quality • Ozone depletion • Particulate • Smog • Roadway air dispersion | |||
| Water pollution | |||
| Eutrophication • Hypoxia • Marine pollution • Ocean acidification • Oil spill • Ship pollution • Surface runoff • Thermal pollution • Wastewater • Waterborne diseases • Water quality • Water stagnation | |||
| Soil contamination | |||
| Bioremediation • Herbicide • Pesticide •Soil Guideline Values (SGVs) | |||
| Radioactive contamination | |||
| Actinides in the environment • Environmental radioactivity • Fission product • Nuclear fallout • Plutonium in the environment • Radiation poisoning • radium in the environment • Uranium in the environment | |||
| Other types of pollution | |||
| Invasive species • Light pollution • Noise pollution • Radio spectrum pollution • Visual pollution | |||
| Government acts | |||
| Clean Air Act • Clean Water Act • Kyoto Protocol • Water Pollution Control Act • Environmental Protection Act 1990 | |||
| Major organizations | |||
| DEFRA • Environmental Protection Agency • Global Atmosphere Watch • Greenpeace • National Ambient Air Quality Standards | |||
| Related topics | |||
| Natural environment |
Particulates, alternatively referred to as particulate matter (PM), aerosols, or fine particles, are tiny particles of solid or liquid suspended in a gas. They range in size from less than 10 nanometers to more than 100 micrometers in diameter. The notation PM10 is used to describe particles of 10 micrometers or less and PM2.5 represents particles less than 2.5 micrometers in aerodynamic diameter; other numeric values may also be used. This range of sizes represent scales from a gathering of a few molecules to the size where the particles no longer can be carried by the gas. Sources of particulate matter can be anthropogenic or natural.
Some particulates occur naturally, originating from volcanoes, dust storms, forest and grassland fires, living vegetation, and sea spray. Human activities, such as the burning of fossil fuels also generate aerosols. Averaged over the globe, anthropogenic aerosols—those made by human activities—currently account for about 10 percent of the total amount of aerosols in our atmosphere.
The field of aerosol science and technology has grown in response to the need to understand and control natural and manmade aerosols.
Sources

There are both natural and human sources of atmospheric particulates. The biggest natural sources are dust, volcanoes, and forest fires. Sea spray is also a large source of particles though most of these fall back to the ocean close to where they were emitted. The biggest human sources of particles are combustion sources, mainly the burning of fuels in internal combustion engines in automobiles and power plants, and wind blown dust from construction sites and other land areas where the water or vegetation has been removed. Some of these particles are emitted directly to the atmosphere (primary emissions) and some are emitted as gases and form particles in the atmosphere (secondary emissions).
- "In Europe and the United States, particulate emissions from vehicles are expected to decline over the next decade. For example, by 2005, the European Union will introduce more stringent standards for particulate emissions from light duty vehicles of 0.025 grams per kilometer [0.04 grams per mile].
- The state of California is implementing an even more restrictive standard in 2004, allowing only 0.006 grams per kilometer [0.01 grams per mile] of particulate emissions. Even if the California standard were introduced worldwide, says Jacobson, diesel cars may still warm the climate more than gasoline cars over 13 to 54 years. New particle traps being introduced by some European automobile manufacturers in their diesel cars appear to reduce black carbon emissions to 0.003 grams per kilometer [0.005 grams per mile], even below the California standard."[1]
BlueTec is one technology developed to reduce particulate emissions from Diesel engines in order to meet strigent Californian standards.
Composition
The composition of aerosol particles depends on their source. Wind-blown mineral dust[2] tends to be made of mineral oxides and other material blown from the Earth's crust. This aerosol is light-absorbing. Sea salt[3] is considered the second largest contributor in the global aerosol budget, and consists mainly of sodium chloride originated from sea spray. Other constituents of atmospheric sea salt reflect the composition of sea water, and thus include magnesium, sulfate, calcium, potassium, and other ions. In addition, sea spray aerosols may contains organic compounds, which influence their chemistry. Sea salt does not absorb light.
Secondary particles derive from the oxidation of primary gases such as sulfur and nitrogen oxides into sulfuric acid (liquid) and nitric acid (gaseous). The precursors for these aerosols, i.e. the gases from which they originate, may have an anthropogenic origin (from fossil fuel combustion) and a natural biogenic origin. In the presence of ammonia, secondary aerosols often take the form of ammonium salts, such as ammonium sulfate and ammonium nitrate (both can be dry or in aqueous solution). In the absence of ammonia, secondary compounds take an acidic form, as sulfuric acid (liquid aerosol droplets) and nitric acid (atmospheric gas). Secondary sulfate and nitrate aerosols are strong light-scatterers.[4] This is mainly because the presence of sulphate and nitrate causes the aerosols to increase to a size that scatters light effectively.
Organic matter (OM) can be either primary or secondary, the latter part deriving from the oxidation of VOCs; organic material in the atmosphere may either be biogenic or anthropogenic. Organic matter influences the atmospheric radiation field by both scattering and absorption of light.
Another important aerosol type is constituted of elemental carbon (EC, also known as black carbon, BC): this aerosol type includes strongly light-absorbing material and is thought to yield large positive radiative forcing. Organic matter and elemental carbon together constitute the carbonaceous fraction of aerosols.[5]
The chemical composition of the aerosol directly affects how it interacts with solar radiation. The chemical constituents within the aerosol change the overall refractive index. The refractive index will determine how much light is scattered and absorbed.
Removal processes
In general, the smaller and lighter a particle is, the longer it will stay in the air. Larger particles (greater than 10 micrometers in diameter) tend to settle to the ground by gravity in a matter of hours whereas the smallest particles (less than 1 micrometer) can stay in the atmosphere for weeks and are mostly removed by precipitation.
Radiative forcing from aerosols
Aerosols, natural and anthropogenic, can affect the climate by changing the way radiation is transmitted through the atmosphere. Direct observations of the effects of aerosols are quite limited so any attempt to estimate their global effect necessarily involves the use of computer models. The Intergovernmental Panel on Climate Change, IPCC, says: While the radiative forcing due to greenhouse gases may be determined to a reasonably high degree of accuracy... the uncertainties relating to aerosol radiative forcings remain large, and rely to a large extent on the estimates from global modelling studies that are difficult to verify at the present time.[6]
A graphic showing the contributions (at 2000, relative to pre-industrial) and uncertainties of various forcings is available.[7]
Sulphate aerosol
Sulphate aerosol has two main effects, direct and indirect. The direct effect, via albedo, is to cool the planet: the IPCC's best estimate of the radiative forcing is -0.4 watts per square meter with a range of -0.2 to -0.8 W/m²,[8] but there are substantial uncertainties. The effect varies strongly geographically, with most cooling believed to be at and downwind of major industrial centres. Modern climate models attempting to deal with the attribution of recent climate change need to include sulfate forcing, which appears to account (at least partly) for the slight drop in global temperature in the middle of the 20th century. The indirect effect (via the aerosol acting as cloud condensation nuclei, CCN, and thereby modifying the cloud properties) is more uncertain but is believed to be a cooling.
Black carbon
Black carbon (BC), or Carbon Black, or Elemental Carbon (EC), often called soot, is composed of pure carbon clusters, skeleton balls and buckyballs, and is one of the most important absorbing aerosol species in the atmosphere. It should be distinguished from Organic Carbon (OC): clustered or aggregated organic molecules on their own or permeating an EC buckyball. BC from fossil fuels is estimated by the IPCC in the Fourth Assessment Report of the IPCC, TAR, to contribute a global mean radiative forcing of +0.2 W/m² (was +0.1 W/m² in the Second Assessment Report of the IPCC, SAR), with a range +0.1 to +0.4 W/m².
All aerosols both absorb and scatter solar and terrestrial radiation. If a substance absorbs a significant amount of radiation, as well as scattering, we call it absorbing. This is quantified in the Single Scattering Albedo (SSA), the ratio of scattering alone to scattering plus absorption (extinction) of radiation by a particle. The SSA tends to unity if scattering dominates, with relatively little absorption, and decreases as absorption increases, becoming zero for infinite absorption. For example, sea-salt aerosol has an SSA of 1, as a sea-salt particle only scatters, whereas soot has an SSA of 0.23, showing that it is a major atmospheric aerosol absorber.
Health effects

The effects of inhaling particulate matter has been widely studied in humans and animals and include asthma, lung cancer, cardiovascular issues, and premature death. The size of the particle is a main determinant of where in the respiratory tract the particle will come to rest when inhaled. Larger particles are generally filtered in the nose and throat and do not cause problems, but particulate matter smaller than about 10 micrometers, referred to as PM10, can settle in the bronchi and lungs and cause health problems. The 10 micrometer size does not represent a strict boundary between respirable and non-respirable particles, but has been agreed upon for monitoring of airborne particulate matter by most regulatory agencies. Similarly, particles smaller than 2.5 micrometers, PM2.5, tend to penetrate into the gas-exchange regions of the lung, and very small particles (< 100 nanometers) may pass through the lungs to affect other organs. In particular, a study published in the Journal of the American Medical Association indicates that PM2.5 leads to high plaque deposits in arteries, causing vascular inflammation and atherosclerosis — a hardening of the arteries that reduces elasticity, which can lead to heart attacks and other cardiovascular problems [9]. Researchers suggest that even short-term exposure at elevated concentrations could significantly contribute to heart disease.
There is also evidence that particles smaller than 100 nanometres can pass through cell membranes. For example, particles may migrate into the brain. It has been suggested that particulate matter can cause similar brain damage as that found in Alzheimer patients. Particles emitted from modern diesel engines (commonly referred to as Diesel Particulate Matter, or DPM) are typically in the size range of 100 nanometres (0.1 micrometres). In addition, these soot particles also carry carcinogenic components like benzopyrenes adsorbed on their surface. It is becoming increasingly clear that the legislative limits for engines, which are in terms of emitted mass, are not a proper measure of the health hazard. One particle of 10 µm diameter has approximately the same mass as 1 million particles of 100 nm diameter, but it is clearly much less hazardous, as it probably never enters the human body - and if it does, it is quickly removed. Proposals for new regulations exist in some countries, with suggestions to limit the particle surface area or the particle number.
The large number of deaths and other health problems associated with particulate pollution was first demonstrated in the early 1970s [10] and has been reproduced many times since. PM pollution is estimated to cause 22,000-52,000 deaths per year in the United States (from 2000) [11] and 200,000 deaths per year in Europe).
Regulation
Due to the health effects of particulate matter, maximum standards have been set by various governments. Many urban areas in the U.S. and Europe still exceed the particulate standards, though urban air on these continents has become cleaner, on average, with respect to particulates over the last quarter of the twentieth century.[citation needed]
United States
The United States Environmental Protection Agency (EPA) sets standards for PM10 and PM2.5 concentrations in urban air. (See National Ambient Air Quality Standards.) EPA regulates primary particulate emissions and precursors to secondary emissions (NOx, sulfur, and ammonia).
EU legislation
In directives 1999/30/EC and 96/62/EC, the European Commission has set limits for PM10 in the air:
| Phase 1 from 1 January 2005 |
Phase 2¹ from 1 January 2010 | |
|---|---|---|
| Yearly average | 40 µg/m³ | 20 µg/m³ |
| Daily average (24-hour) allowed number of exceedences per year. |
50 µg/m³ 35 |
50 µg/m³ 7 |
¹ indicative value.
Affected areas
| Most Polluted World Cities by PM[12] | |
|---|---|
| Particulate matter, μg/m3 (2004) |
City |
| 169 | Cairo, Egypt |
| 161 | Beijing, China |
| 150 | Delhi, India |
| 128 | Kolkata, India (Calcutta) |
| 125 | Taiyuan, China |
| 123 | Chongqing, China |
| 109 | Kanpur, India |
| 109 | Lucknow, India |
| 104 | Jakarta, Indonesia |
| 101 | Shenyang, China |
The most concentrated particulate matter pollution tends to be in densely populated metropolitan areas in developing countries. The primary cause is the burning of fossil fuels by transportation and industrial sources.
See also
- Aerosol science
- Air pollution
- Biological warfare
- Criteria air contaminants
- Deposition
- Dust
- Global dimming
- Global warming
- Haze
- Radiological weapon
- Cloud
- Fog
- "Pea soup" fog
Notes
- ↑ [1]
- ↑ [2]
- ↑ [3]
- ↑ [4]
- ↑ [5]
- ↑ [6].
- ↑ here.
- ↑ [7]
- ↑ Pope, Arden C and et al. (2002). Cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. J. Amer. Med. Assoc. 287: 1132-1141.
- ↑ Lave, Lester B. and Eugene P. Seskin (1973). An Analysis of the Association Between U.S. Mortality and Air Pollution. J. Amer. Statistical Association 68: 342.
- ↑ Mokdad, Ali H. and et al. (2004). Actual Causes of Death in the United States, 2000. J. Amer. Med. Assoc. 291 (10): 1238.
- ↑ [8]
ReferencesISBN links support NWE through referral fees
- Article at earthobservatory.nasa.gov describing the possible influence of aerosols on the climate
- The Intergovernmental Panel on Climate Change (the principal international scientific body on climate change) chapter on atmospheric aerosols and their radiative effects
- [9] InsideEPA.com, Study Links Air Toxics To Heart Disease In Mice Amid EPA Controversy
- Preining, Othmar and E. James Davis (eds.), "History of Aerosol Science," Österreichische Akademie der Wissenschaften, ISBN 3700129157 (pbk.)
- G Invernizzi et al., Particulate matter from tobacco versus diesel car exhaust: an educational perspective. Tobacco Control 13, S.219-221 (2004)
- Sheldon K.Friedlander, "Smoke, Dust and Haze."
- JEFF CHARLTON Pandemic planning: a review of respirator and mask protection levels.
External links
- National Pollutant Inventory - Particulate matter fact sheet
- WHO-Europe reports: Health Aspects of Air Pollution (2003) (PDF) and "Answer to follow-up questions from CAFE (2004) (PDF)
- American Association for Aerosol Research
- Particulate Air Pollution
- Aerosol Society - The Development of Aerosol Science in the United Kingdom
- Watch and read 'Dirty Little Secrets', 2006 Australian science documentary on health effects of fine particle pollution from vehicle exhausts
- Aerosol Science and Technology
- Canada-Wide Standards
- Little Green Data Book 2007, World Bank. Lists C02 and PM statistics by country.
- Air Pollution in World Cities (PM10 Concentrations)
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.