Supercritical fluid
A supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. It has the unique ability to diffuse through solids like a gas, and dissolve materials like a liquid. Additionally, it can readily change in density upon minor changes in temperature or pressure. These properties make it suitable as a substitute for organic solvents in a process called Supercritical Fluid Extraction. Carbon dioxide and water are the most commonly used supercritical fluids.
Introduction
In 1822, Baron Charles Cagniard de la Tour discovered the critical point of a substance in his famous cannon barrel experiments. Listening to discontinuities in the sound of a rolling flint ball in a sealed cannon filled with fluids at various temperatures, he observed the critical temperature. Above this temperature, the densities of the liquid and gas phases become equal and the distinction between them disappears, resulting in a single supercritical fluid phase. In Table 1, the critical properties are shown for some components, which are commonly used as supercritical fluids.
Phase diagram
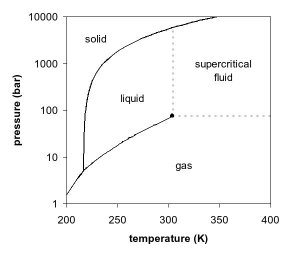
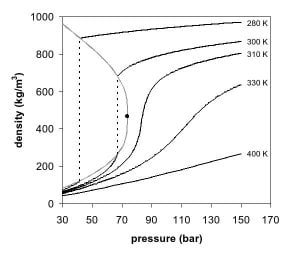
The observations by de la Tour can be explained by looking at the phase diagram of a pure component, for example, carbon dioxide. In Figures 1 and 2, two projections of the phase diagram of carbon dioxide are shown. In the pressure-temperature phase diagram (Fig. 1), the boiling line is observed, which separates the vapor and liquid region and ends in the critical point. At the critical point, the densities of the equilibrium liquid phase and the saturated vapor phases become equal, resulting in the formation of a single supercritical phase. This can be observed in the density-pressure phase diagram for carbon dioxide, as shown in Figure 2, where the critical point is located at 304.1 K and 7.38 MPa (73.8 bar). With increasing temperatures, the liquid-vapor density gap decreases, up to the critical temperature, at which the discontinuity disappears. Thus, above the critical temperature a gas cannot be liquefied by pressure. However, at extremely high pressures the fluid can solidify, as visible at the top of Figure 1. By definition, a supercritical fluid is a substance above both its critical temperature and pressure. In a practical sense, the area of interest in supercritical fluids for processing and separation purposes is limited to temperatures in the vicinity of the critical point, where large gradients in the physical properties are observed. The changes near the critical point are not limited to density. Many other physical properties also show large gradients with pressure near the critical point, for example, viscosity, the relative permitivity and the solvent strength, which are all closely related to the density. At higher temperatures, the fluid starts to behave like a gas, as can be seen in Figure 2. For carbon dioxide at 400 K, the density increases almost linearly with pressure.
Applications
For engineering purposes, supercritical fluids can be regarded as “hybrid solvents” with properties between those of gases and liquids, such as a solvent with a low viscosity, high diffusion rates and no surface tension. In the case of supercritical carbon dioxide, the viscosity is in the range of 20–100 µPa·s (0.02-0.1 cP), where liquids have viscosities of approximately 500–1000 µPa·s (0.5-1.0 cP) and gases approximately 10 µPa·s (0.01 cP), respectively. Diffusivities of solutes in supercritical carbon dioxide are up to a factor 10 higher than in liquid solvents. Additionally, these properties are strongly pressure-dependent in the vicinity of the critical point, making supercritical fluids highly tunable solvents. Of the components shown in Table 1, carbon dioxide and water are the most frequently used in a wide range of applications, including extractions, dry cleaning and chemical waste disposal. In polymer systems, ethylene and propylene are also widely used, where they act both as a solvent and as the reacting monomer.
One of the most important properties of supercritical fluids is that their solvating properties are a complex function of their pressure and temperature, independent of their density. This means that (taking a very simplistic approach) raw materials containing soluble products can be selectively extracted or selectively precipitated to obtain ultra-pure extracts. Although the details are much more complex than this, it remains the dominant chemical-free technology for the production of decaffeinated coffee, nicotine-free tobacco, and many of the world's best spice extracts.
Supercritical water reactors (SCWRs) are promising advanced nuclear systems because of their high thermal efficiency (i.e., about 45 percent vs. about 33 percent efficiency for current light water reactors (LWR) and considerable plant simplification.
| Solvent | Molecular weight | Critical temperature | Critical pressure | Density |
|---|---|---|---|---|
| g/mol | K | MPa (atm) | g/cm³ | |
| Carbon dioxide () | 44.01 | 304.1 | 7.38 (72.8) | 0.469 |
| Water () | 18.02 | 647.3 | 22.12 (218.3) | 0.348 |
| Methane () | 16.04 | 190.4 | 4.60 (45.4) | 0.162 |
| Ethane () | 30.07 | 305.3 | 4.87 (48.1) | 0.203 |
| Propane () | 44.09 | 369.8 | 4.25 (41.9) | 0.217 |
| Ethylene () | 28.05 | 282.4 | 5.04 (49.7) | 0.215 |
| Propylene () | 42.08 | 364.9 | 4.60 (45.4) | 0.232 |
| Methanol () | 32.04 | 512.6 | 8.09 (79.8) | 0.272 |
| Ethanol () | 46.07 | 513.9 | 6.14 (60.6) | 0.276 |
| Acetone () | 58.08 | 508.1 | 4.70 (46.4) | 0.278 |
Supercritical carbon dioxide
Supercritical carbon dioxide refers to carbon dioxide with some unique properties. Carbon dioxide usually behaves as a gas in air or as a solid in dry ice. If the temperature and pressure are both increased, it can adopt properties midway between a gas and a liquid. It behaves like a supercritical fluid above its critical temperature (31.1°C) and pressure (73 atm), expanding to fill its container like a gas, but with a density like that of a liquid. Supercritical CO2 is becoming an important commercial and industrial solvent due to its role in compound extraction as well as its low toxicity and environmental impact. The relatively low temperature of the process and the stability of CO2 also allows most compounds to be extracted with little damage or denaturing.
Uses
Supercritical carbon dioxide is gaining popularity amongst coffee manufacturers looking to move away from some of the classic decaffeinating solvents of the past; many of which have led to public outcry because of real or perceived dangers related to their use in food preparation. Supercritical CO2 is forced through the green coffee beans and then sprayed with water at high pressure to remove the caffeine. The caffeine can then be isolated for resale to, for example, the pharmaceutical industry or beverage manufacturers by passing the water through activated charcoal filters or by distillation, crystallization, or reverse osmosis.
Supercritical carbon dioxide is also becoming a more common process for extracting volatile oils and fragrance compounds from various raw materials that are used in perfumery. The relatively low critical temperature and reactivity of CO2 allows the fragrance compounds to be extracted without extensive damage or denaturing, which will alter their odor.
Supercritical carbon dioxide can be used in cleaning clothes, instead of perchloroethylene (PCE or Perc) or water. This new approach of cleaning clothes was developed and commercialized by Dr. Joseph DeSimone, professor of chemical engineering at North Carolina State University, in Raleigh. Supramics, environmentally beneficial, low-cost substitutes for rigid thermoplastic and fired ceramic, are made using supercritical carbon dioxide as a chemical reagent. The supercritical carbon dioxide in these processes is reacted with the alkaline components of fully hardened hydraulic cement or gypsum plaster to form various carbonates. The sole by-product is ultra-pure water. Because supramics consume and sequester carbon as stable compounds in useful products, they may serve to reduce carbon that would otherwise be released into the environment.
There is considerable work being done to develop an enhanced version of a gas-turbine power production cycle to operate at temperatures near 550°C. This is a significant usage, which could have large implications for bulk thermal and nuclear generation of electricity, because the supercritical properties of carbon dioxide at above 500°C and 20 MPa enable very high thermal efficiencies, approaching 45 percent. This could increase the electrical power produced per unit of fuel required by 40 percent or more. Given the huge volume of extremely polluting fuels used in producing electricity, the potential environmental impact of such an efficient cycle could be very large.[1]
Processes which use supercritical carbon dioxide to produce micro and nano scale particles, often for pharmaceutical uses, are currently being developed. The gas antisolvent process, rapid expansion of supercritical solutions, and supercritical antisolvent precipitation (as well as several related methods) have been shown to process a variety of substances into particles. [2]
Supercritical carbon dioxide is also used in the foaming of polymers. Many corporations utilize supercritical carbon dioxide to saturate the polymer with solvent (carbon dioxide). Upon depressurization and heating the carbon dioxide rapidly expands, causing voids within the polymer matrix, such as creating a foam. Research is also ongoing at many universities in the production of microcellular foams using supercritical carbon dioxide.
Supercritical carbon dioxide is beginning to be used to enhance oil recovery in mature oil fields. At the same time, there is the possibility of using the various "clean coal" technologies which are emerging to combine such enhanced recovery methods with carbon sequestation efforts. Using advanced Fischer-Tropsch digesters instead of conventional furnaces, coal and water is reduced to hydrogen gas, carbon dioxide, and ash. This hydrogen gas can be used to produce electrical power in combined-cycle gas turbines, while the CO2 is captured, compressed to the supercritical state, and injected into geological storage, possibly into existing oil fields to improve yields. The unique properties of supercritical CO2 ensure that it will remain out of the atmosphere.[3]
Supercritical carbon dioxide is also an important emerging natural refrigerant, being used in new, low carbon solutions for domestic heat pumps.[4] These systems are undergoing continuous development with the first commercial supercritical carbon dioxide heat pumps being now marketed. The "EcoCute" systems from Japan, developed by consortium of companies including Mitsubishi, develop high temperature domestic water at with small inputs of electric power by moving heat into the system from their surroundings.
Environmental impact
Supercritical carbon dioxide is seen as a promising green solvent because it is non-toxic, and a byproduct of other industrial processes. Furthermore, separation of the reaction components from the starting material is much simpler than with traditional organic solvents.
Notes
- ↑ MIT, V. Dostal, M.J. Driscoll, P. Hejzlar, "A Supercritical Carbon Dioxide Cycle for Next Generation Nuclear Reactors," MIT-ANP-Series, MIT-ANP-TR-100 (2004).
- ↑ DOI, S. Yeo and E. Kiran, "Formation of polymer particles with supercritical fluids: A review," J. Supercrit. Fluids, 34 (2005), 287. Retrieved March 6, 2008.
- ↑ NAP, "The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs," p. 84 (2004). Retrieved March 6, 2008.
- ↑ r744, FAQs—Supercritical CO2 in heat pumps and other applications. Retrieved March 6, 2008.
ReferencesISBN links support NWE through referral fees
- DeSimone, Joseph M. and William Tumas, eds. 2003. Green Chemistry Using Liquid and Supercritical Carbon Dioxide. New York: Oxford University Press. ISBN 0195154835
- Mukhopadhyay, Mamata. 2000. Natural Extracts Using Supercritical Carbon Dioxide. Boca Raton, FL: CRC Press. ISBN 0849308194
- Reid, Robert C., J. M. Prausnitz, and Bruce E. Poling. 1987. The Properties of Gases and Liquids. New York: McGraw-Hill. ISBN 0070517991
- Taylor, Larry T. 1996. Supercritical Fluid Extraction. New York: Wiley. ISBN 0471119903
- Wagner, Wolfgang, and Alfred Kruse. 1998. Properties of Water and Steam: The Industrial Standard IAPWS-IF97 for the Thermodynamic Properties and Supplementary Equations for Other Properties: Tables Based on These Equations. Berlin: Springer-Verlag. ISBN 3540643397
- Yeo, S., and E. Kiran. 2005. Formation of polymer particles with supercritical fluids: a review. J. Supercrit. Fluids 34:287. Retrieved July 10, 2007.
| States of Matter |
|---|
| Solid | Liquid | Gas | Plasma | Colloid | Supercritical fluid | Superfluid | Supersolid | Degenerate matter | Quark-gluon plasma | Fermionic condensate | Bose–Einstein condensate | Strange matter | Melting point | Boiling point | Triple point | Critical point | Equation of state | Cooling curve |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.