Disaccharide
A disaccharide is a type of carbohydrate consisting of two monosaccharides (simple sugars) linked together by covalent bonds. Common nutritionally important disaccharides are sucrose, lactose, and maltose.
Disaccharide is one of the four basic categories of carbohydrates, the others being monosaccharide, oligosaccharide, and polysaccharide. Monosaccharides, such as glucose, are the monomers out of which disaccharides are constructed.
While the disaccharides sucrose, lactose, maltose, and cellobiose have the same chemical formula, C12H22O11, and all have at least one glucose molecule as part of their structure, they differ substantially in their properties. Even maltose and cellobiose, which are made up of two covalently bonded glucose units, differ due to the nature of the linkage, with maltose easily hydrolyzed to its monosaccharides in the human body whereas cellobiose cannot be digested. This reveals not only a complex coordination in nature, but also reflects on the diversity in creation.
The general chemical formula for carbohydrates, C(H2O), gives the relative proportions of carbon, hydrogen, and oxygen in a monosaccharide (the proportion of these atoms are 1:2:1). In disaccharides (and oligosaccharides and polysaccharides), the molar proportions deviate slightly from the general formula because two hydrogens and one oxygen are lost during each of the condensation reactions that forms them. These carbohydrates have the general formula Cn(H2O)m.
Overview and formation
Carbohydrates are a class of biological molecules that contain primarily carbon (C) atoms flanked by hydrogen (H) atoms and hydroxyl (OH) groups (H-C-OH).
The four categories of carbohydrates are classified by their number of sugar units:
- Monosaccharides (mono- "one," saccharide- "sugar") are the monomers (small molecules that may bond chemically to form a polymer) out of which larger carbohydrates are constructed. Monosaccharides such as glucose, ribose, and fructose are simple sugars.
- Disaccharides (di-"two"), such as sucrose and lactose, are two monosaccharides linked together by covalent bonds.
- Oligosaccharides (oligo- "several") are made up of from three to 20 monosaccharides.
- Polysaccharides (poly- "many") are large polymers composed of hundreds or thousands of monosaccharides. Starch, glycogen, and cellulose are polysaccharides.
In other words, the repeating units of disaccharides (and oligosaccharides and polysaccharides) are simple sugars called monosaccharides.
There are two categories of monosaccharides: aldosugars, with a terminal carbonyl group (a carbon atom double-bonded to an oxygen atom), and ketosugars, with an internal carbonyl group typically on the second carbon atom. Within these two groups, sugars are named according to the number of carbon atoms they contain. Sugars that have between three and seven carbon atoms are termed triose (three carbons), tetrose (four carbons), pentose (five carbons), hexose (six carbons), or heptose (seven carbons). Among the monosaccarides, glucose is an aldohexose, fructose is a ketohexose, and ribose is an aldopentose. Each carbon atom that supports a hydroxyl group (except for the first and last) is optically active, allowing a number of different carbohydrates with the same basic structure. For instance, galactose is an aldohexose but has different properties from glucose because the atoms are arranged differently.
When two monosaccharides are combined, such as glucose with glucose or glucose with fructose, a disaccharide is the result. As the two sugars are joined together, a molecule of water is removed. For example, milk sugar (lactose) is made from glucose and galactose whereas cane sugar (sucrose) is made from glucose and fructose. The two monosaccharides are bonded via a dehydration reaction (also called a condensation reaction) that leads to the loss of the molecule of water.
Structure
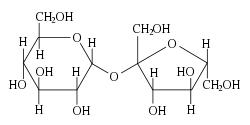
Three common disaccharides, and nutritionally important, are sucrose, lactose, and maltose. Each of these has the same chemical formula (C12H22O11) and each has at least one glucose unit as part of their structure.
That is, the aldohexose D-glucose, the single most common monosaccharide, represented by the formula C6H12O6, occurs not only as the free monosaccharide, but also in disaccharides. The bond that link the two units covalently is called a glycosidic bond—a functional group that joins a carbohydrate (sugar) molecule to an alcohol, which may be another carbohydrate. This is what forms by the condensation reaction in which there is a loss of hydrogen (H) from one molecule and a hydroxyl group (OH) from the other. The resulting glycosidic bond is the characteristic linkage between sugars.
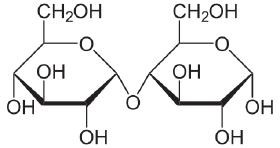
Maltose links two glucose units by an α(1→4) glycosidic linkage, that is, a glycosidic bond forms between carbon one of the first glucose molecule and carbon four of the second glucose molecule. (The carbons of glucose are numbered beginning with the more oxidized end of the molecule, the carbonyl group.) Lactose (milk sugar) involves glucose and galactose bonded through a β1-4 glycosidic linkage. Sucrose (common table sugar) consists of a glucose and a fructose joined by a glycosidic bond between carbon atom one of the glucose unit and carbon atom two of the fructose unit.
Although the disaccharide maltose contains two glucose molecules, it is not the only disaccharide that can be made from two glucoses. When glucose molecules form a glycosidic bond, the linkage will be one of two types, α or β, depending on whether the molecule that bonds its carbon one is an α-glucose or β-glucose. An α-linkage with carbon four of a second glucose molecule results in maltose, whereas a β-linkage results in cellobiose. Although maltose and cellobiose are disaccharide isomers, both having the formula C12H22O11, they are different compounds with different properties. For example, maltose can be hydrolyzed to its monosaccharides in the human body where as cellobiose cannot. Some organisms have the capacity to break down cellobiose.
In other words, the glycosidic bond can be formed between any hydroxyl group on the component monosaccharide. So, even if both component sugars are the same (e.g., glucose), different bond combinations (regiochemistry) and stereochemistry (alpha- or beta-) result in disaccharides that are diastereoisomers with different chemical and physical properties.
Depending on the monosaccharide constituents, disaccharides are sometimes crystalline, sometimes water-soluble, and sometimes sweet-tasting.
Common disaccharides
| Disaccharide | Unit 1 | Unit 2 | Bond | Disaccharidase |
| Sucrose (table sugar, cane sugar, saccharose, or beet sugar) | glucose | fructose | α(1→2) | sucrase |
| Lactose (milk sugar) | galactose | glucose | β(1→4) | lactase |
| Maltose | glucose | glucose | α(1→4) | maltase |
| Trehalose | glucose | glucose | α(1→1)α | trehalase |
| Cellobiose | glucose | glucose | β(1→4) | cellobiase |
Maltose and cellobiose are hydrolysis products of the polysaccharides, starch and cellulose, respectively.
ReferencesISBN links support NWE through referral fees
- Becker, W. M., L. J. Kleinsmith, and J. Hardin. The World of the Cell. San Francisco: Benjamin Cummings, 2006. ISBN 0805346805
- Bender, D. A., and A. E. Bender. A Dictionary of Food and Nutrition. New York: Oxford University Press, 2005. ISBN 0198609612
- King, M. W. Disaccharides Indiana State University, 2006. Retrieved June 30, 2007.
- Purves, W. K., G. H. Orians, and H. C. Heller. Life, The Science of Biology. Sunderland, MA: Sinauer Associates, 2004. OCLC 76768203
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.