Carboxylic acid
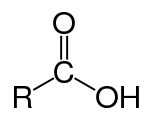
Carboxylic acids are organic acids characterized by the presence of one or more carboxyl groups in their molecules. A carboxyl group consists of a carbon atom attached to an oxygen atom with a double covalent bond and to a hydroxyl group by a single covalent bond. The chemical formula of the carboxyl group may be written as -C(=O)OH, -COOH, or -CO2H.[1] Salts and anions of carboxylic acids are called carboxylates.
Carboxylic acids are widespread in nature. For example, acetic acid is present in vinegar, malic acid is found in apples, lactic acid is present in sour milk, and citric acid is contained in citrus fruits such as lemons, oranges, and grapefruits.
Sources
Lower straight-chain aliphatic carboxylic acids, as well as those of even carbon number up to C18 are commercially available. For example, acetic acid is produced by methanol carbonylation with carbon monoxide, while long chain carboxylic acids are obtained by the hydrolysis of triglycerides obtained from plant or animal oils.
Vinegar, a dilute solution of acetic acid, is biologically produced from the fermentation of ethanol. It is used in food and beverages but is not used industrially.
Physical properties
The simplest series of carboxylic acids are the alkanoic acids, written as R-COOH, where R is a hydrogen atom or an alkyl group. Compounds may also have two or more carboxylic acid groups per molecule.
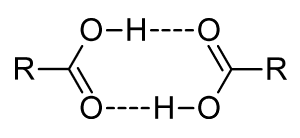
Carboxylic acids are polar and form hydrogen bonds with one another. At high temperatures, in the vapor phase, carboxylic acids usually exist as dimeric pairs, as shown in the diagram.
Lower carboxylic acids (containing one to four carbon atoms per molecule) are miscible with water, but higher carboxylic acids (with large alkyl groups) are much less soluble because of the increasing hydrophobic nature of the alkyl chain. They tend to be soluble in less polar solvents such as ethers and alcohols.[2]
Carboxylic acids are Bronsted acids—that is, they are proton donors. They are typically weak acids, meaning they only partially dissociate into H+ cations and RCOO− anions in aqueous solution. For example, at room temperature, only 0.02 percent of all acetic acid molecules are dissociated in water.
Thus, in an aqueous solution, the undissociated acid exists in a chemical equilibrium with the dissociated acid:
- RCOOH ↔ RCOO- + H+
The acidity of carboxylic acids can be explained either by the stability of the acid, or the stability of the conjugate base using inductive effects or resonance effects.
Stability of the acid
Using inductive effects, the acidity of carboxylic acids can be rationalized by the two electronegative oxygen atoms distorting the electron clouds surrounding the O-H bond, weakening it. The weak O-H bond causes the acid molecule to be less stable, and causing the hydrogen atom to be labile, thus it dissociates easily to give the H+ ion. Since the acid is unstable, the equilibrium will lie on the right.
Additional electronegative atoms or groups such as chlorine or hydroxyl, substituted on the R-group have a similar, though lesser effect. The presence of these groups increases the acidity through inductive effects. For example, trichloroacetic acid (three -Cl groups) is a stronger acid than lactic acid (one -OH group) which in turn is stronger than acetic acid (no electronegative constituent).
Stability of the conjugate base
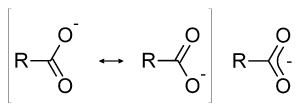
The acidity of a carboxylic acid can also be explained by resonance effects. The result of the dissociation of a carboxylic acid is a resonance stabilized product in which the negative charge is shared (delocalized) between the two oxygen atoms. Each of the carbon-oxygen bonds has what is called a partial double bond characteristic. Since the conjugate base is stabilized, the above equilibrium lies on the right.
Synthesis
Carboxylic acids can be synthesized by any of several methods. Some examples follow.
Oxidation:
- Carboxylic acids can be produced by oxidation of primary alcohols and aldehydes with strong oxidants such as potassium permanganate or sodium chlorite.
- They may also be produced by the oxidative cleavage of olefins by potassium permanganate or potassium dichromate.
- Any alkyl group on a benzene ring will be fully oxidized to a carboxylic acid, regardless of its chain length. This is the basis for the industrial synthesis of benzoic acid from toluene.
Hydrolysis:
- Carboxylic acids can be obtained by the hydrolysis of esters, amides, or nitriles, with the addition of acid or base.
Additional methods:
- Disproportionation of an aldehyde in the Cannizzaro reaction.
- Rearrangement of diketones in the benzilic acid rearrangement.
- Halogenation followed by hydrolysis of methyl ketones in the haloform reaction
Chemical reactions
Acid-base reactions:
A carboxylic acid reacts with a base to form a carboxylate salt in which the hydrogen of the carboxyl group is replaced with a metal cation. For instance, acetic acid reacts with sodium hydroxide (a base) to produce sodium acetate, a water-soluble salt, and water. The reaction may be written as:
- CH3COOH + NaOH → CH3COONa + H2O
When baking soda is added to vinegar, we observe that the solution fizzes. This is because acetic acid in the vinegar reacts with baking soda (sodium bicarbonate) to produce sodium acetate, carbon dioxide (which bubbles up to create the fizz), and water. The reaction may be written as follows:
- CH3COOH + NaHCO3 → CH3COONa + CO2 + H2O
Formation of esters and amides:
Carboxylic acids also react with alcohols to give esters and with amines to generate amides. Like other alcohols and phenols, the hydroxyl group on carboxylic acids may be replaced with a chlorine atom using thionyl chloride to give acyl chlorides.
Reduction to alcohols:
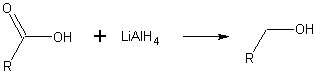 Lithium aluminium hydride reduction of a carboxylic acid to an alcohol.
Lithium aluminium hydride reduction of a carboxylic acid to an alcohol.
Carboxylic acids may be reduced by lithium aluminum hydride or borane to give primary alcohols. It is easier, however, to reduce an ester to an alcohol.[2]
- Like all carbonyl compounds, the protons on the α-carbon are labile due to keto-enol tautomerization. Thus the α-carbon is easily halogenated in the Hell-Volhard-Zelinsky halogenation.
- The Arndt-Eistert synthesis inserts an α-methylene group into a carboxylic acid.
- The Curtius rearrangement converts carboxylic acids to isocyanates.
- The Schmidt reaction converts carboxylic acids to amines.
- Carboxylic acids are decarboxylated in the Hunsdiecker reaction
- The Dakin-West reaction converts an amino acid to the corresponding amino ketone.
- In the Barbier-Wieland degradation (1912) the alpha-methylene group in an aliphatic carboxylic acid is removed in a sequence of reaction steps, effectively a chain-shortening.[3][4]
- The addition of a carboxyl group to a compound is known as carboxylation; the removal of one is decarboxylation. Enzymes that catalyze these reactions are known as carboxylases (EC 6.4.1) and decarboxylases (EC 4.1.1), respectively.
Nomenclature and examples
In IUPAC nomenclature, carboxylic acids have an -oic acid suffix—for example, octadecanoic acid. In common nomenclature, the suffix is usually -ic acid—for example, stearic acid.
The carboxylate anion R-COO– is usually named with the suffix -ate. Thus, for example, the anion of acetic acid is called the acetate ion.
| Carbon atoms | Common name | IUPAC name | Chemical formula | Common location or use |
|---|---|---|---|---|
| 1 | Formic acid | Methanoic acid | HCOOH | Insect stings |
| 2 | Acetic acid | Ethanoic acid | CH3COOH | Vinegar |
| 3 | Propionic acid | Propanoic acid | CH3CH2COOH | |
| 4 | Butyric acid | Butanoic acid | CH3(CH2)2COOH | Rancid butter |
| 5 | Valeric acid | Pentanoic acid | CH3(CH2)3COOH | |
| 6 | Caproic acid | Hexanoic acid | CH3(CH2)4COOH | |
| 7 | Enanthic acid | Heptanoic acid | CH3(CH2)5COOH | |
| 8 | Caprylic acid | Octanoic acid | CH3(CH2)6COOH | |
| 9 | Pelargonic acid | Nonanoic acid | CH3(CH2)7COOH | |
| 10 | Capric acid | Decanoic acid | CH3(CH2)8COOH | |
| 12 | Lauric acid | Dodecanoic acid | CH3(CH2)10COOH | Coconut oil |
| 18 | Stearic acid | Octadecanoic acid | CH3(CH2)16COOH |
Other carboxylic acids include:
- Short chain unsaturated monocarboxylic acids
- Acrylic acid (2-propenoic acid) – CH2=CHCOOH, used in polymer synthesis
- Fatty acids – medium to long chain saturated and unsaturated monocarboxylic acids, with even number of carbons
- Docosahexaenoic acid – nutritional supplement
- Eicosapentaenoic acid – nutritional supplement
- Amino acids – the building blocks of proteins
- Keto acids – acids of biochemical significance that contain a ketone group
- Pyruvic acid
- Acetoacetic acid
- Aromatic carboxylic acids
- Benzoic acid – C6H5COOH. Sodium benzoate, the sodium salt of benzoic acid is used as a food preservative
- Salicylic acid – found in many skin care products
- Dicarboxylic acids – containing two carboxyl groups
- Aldaric acid – a family of sugar acids
- Oxalic acid – found in many foods
- Malonic acid
- Malic acid – found in apples
- Succinic acid – a component of the citric acid cycle
- Glutaric acid
- Adipic acid – the monomer used to produce nylon
- Tricarboxylic acids – containing three carboxyl groups
- Citric acid – found in citrus fruits
- Alpha hydroxy acids – containing a hydroxy group
- Lactic acid (2-hydroxypropanoic acid) – found in sour milk
See also
- Acid
- Acid anhydride
- Acid chloride
- Amide
- Ester
- Functional group
- Organic chemistry
- Salt
Notes
- ↑ Compendium of Chemical Terminology, carboxylic acids Retrieved December 8, 2007.
- ↑ 2.0 2.1 R.T. Morrison and R.N. Boyd. Organic Chemistry, 6th Ed. 1992. ISBN 0-13-643669-2
- ↑ Organic Syntheses, Coll. Vol. 3, p.234, 1955; Vol. 24, p.38, 1944. Online Link Retrieved December 8, 2007.
- ↑ Organic Syntheses, Coll. Vol. 3, p.237, 1955; Vol. 24, p.41, 1944. Online Link Retrieved December 8, 2007.
ReferencesISBN links support NWE through referral fees
- McMurry, John. 2004. Organic Chemistry. 6th ed. Belmont, CA: Brooks/Cole. ISBN 0534420052
- Morrison, Robert T., and Robert N. Boyd. 1992. Organic Chemistry. 6th ed. Englewood Cliffs, NJ: Prentice Hall. ISBN 0-13-643669-2
- Solomons, T.W. Graham, and Fryhle, Craig B. 2004. Organic Chemistry. 8th ed. Hoboken, NJ: John Wiley. ISBN 0471417998
External links
- IUPAC Compendium of Chemical Terminology - the Gold Book International Union of Pure and Applied Chemistry. Retrieved December 8, 2007.
- CurTiPot (Freeware for pH calculations, data analysis, simulation, and distribution diagram generation.) Retrieved December 8, 2007.
| Functional groups |
|---|
| Chemical class: Alcohol • Aldehyde • Alkane • Alkene • Alkyne • Amide • Amine • Azo compound • Benzene derivative • Carboxylic acid • Cyanate • Ester • Ether • Haloalkane • Imine • Isocyanide • Isocyanate • Ketone • Nitrile • Nitro compound • Nitroso compound • Peroxide • Phosphoric acid • Pyridine derivative • Sulfone • Sulfonic acid • Sulfoxide • Thioether • Thiol • Toluene derivative |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.