Sphalerite

Sphalerite is a mineral that consists largely of zinc sulfide in crystalline form, but it almost always contains variable amounts of iron. Its chemical formula may therefore be written as (Zn,Fe)S. Its color is usually yellow, brown, or gray to gray-black, and it may be shiny or dull. This mineral is the chief ore of zinc, the fourth most common metal in use. In addition, appropriate crystals of sphalerite have been fashioned into gemstones.
Occurrence
Sphalerite is usually found in association with other minerals, particularly galena, pyrite, and other sulfides, along with calcite, dolomite, and fluorite. Miners have referred to sphalerite as zinc blende, mock lead, false galena, and black-jack.
Significant quantities of this mineral have been found in many parts of the world, including Australia, Burma, Germany, England, Italy, Spain, and Peru. In the United States, it is found in Elmwood, Tennessee; Joplin, Missouri; and Rosiclare, Illinois.
Properties
As noted above, sphalerite can be found in various colors, particularly yellow, brown, and gray to gray-black. It has a yellow or light brown streak. Its hardness rating is 3.5-4 on the Mohs scale, its specific gravity is 3.9-4.1, and its refractive index is 2.37 (when measured using sodium light at 589.3 nanometers). Its dispersion of light is more than three times that of diamond.
Some specimens, called "ruby sphalerite," have a red iridescence within the gray-black crystals. The pale yellow and red varieties have very little iron and are translucent. The darker, more opaque varieties contain more iron. An opaque, black variety with a high iron content is known as marmatite. Gemmy, pale specimens (such as from Franklin, New Jersey) are highly fluorescent orange or blue under longwave ultraviolet light and are known as cleiophane, an almost pure variety of zinc sulfide.
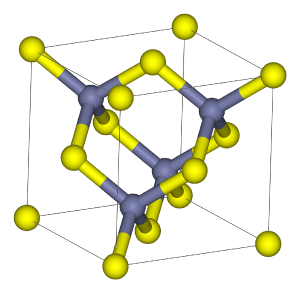
Sphalerite crystallizes in the isometric crystal system and possesses perfect dodecahedral cleavage. In the crystal structure, zinc and sulfur atoms are tetrahedrally coordinated. This structure is closely related to that of diamond.
Two other minerals, wurtzite and matraite, are polymorphs of sphalerite. They share the same chemistry (zinc iron sulfide) but have different crystalline structures.
Uses
Sphalerite is mainly used for the extraction of zinc, the fourth most common metal in use, trailing only iron, aluminum, and copper in annual production. In addition, crystals of suitable size and transparency have been fashioned into gemstones, usually featuring the brilliant cut to best display the stone's high optical dispersion. Freshly cut gems have a lively, adamantine luster and could be mistaken for fancy-colored diamonds, but given sphalerite's softness and fragility, the gems are best left unset as collector's items or museum pieces. Some specimens, however, have been set into pendants. Gem-quality material is usually a yellowish to honey brown, red to orange, or green; the two most important sources are the Chivera mine, Cananea, Sonora, Mexico; and the Picos de Europa, Cordillera Cantabrica, near Santander on Spain's northern coast.
See also
ReferencesISBN links support NWE through referral fees
- Farndon, John. 2006. The Practical Encyclopedia of Rocks & Minerals: How to Find, Identify, Collect and Maintain the World's best Specimens, with over 1000 Photographs and Artworks. London: Lorenz Books. ISBN 0754815412
- Klein, Cornelis, and Barbara Dutrow. 2007. Manual of Mineral Science. 23rd ed. New York: John Wiley. ISBN 978-0471721574
- Mindat.org. 2007. Sphalerite Mindat.org. Retrieved November 7, 2007.
- Mineral Gallery. 2006. The Mineral Sphalerite Amethyst Galleries.
- Pellant, Chris. 2002. Rocks and Minerals. Smithsonian Handbooks. New York: Dorling Kindersley. ISBN 0789491060
- Shaffer, Paul R., Herbert S. Zim, and Raymond Perlman. 2001. Rocks, Gems and Minerals. Rev. ed. New York: St. Martin's Press. ISBN 1582381321
External links
All links retrieved February 7, 2023.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
