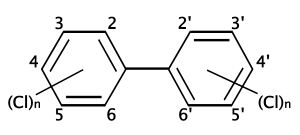
Polychlorinated biphenyl
Polychlorinated biphenyls (PCBs) are a class of organic compounds with 1 to 10 chlorine atoms attached to biphenyl and a general chemical formula of C12H10-xClx. Most PCBs were manufactured as cooling and insulating fluids for industrial transformers and capacitors, and also as stabilizing additives in flexible PVC coatings of electrical wiring and electronic components. However, PCB production was banned in the 1970s, due to the high toxicity of most PCB congeners (PCB congeners are compounds that are derivatives of and related to PCBs in chemical structure) and mixtures. PCBs are classified as persistent organic pollutants.
Physical and chemical properties
Most of the 209 different PCB congeners are colorless, odorless crystals. Commercial PCB mixtures are clear viscous liquids (the more highly chlorinated mixtures are more viscous, for example, Aroclor 1260 is a "sticky resin"). PCBs have low water solubilities and low vapor pressures at room temperature, but they have high solubilities in most organic solvents, oils, and fats. Other physical and chemical properties vary widely across the class.
PCBs readily penetrate skin, PVC (polyvinyl chloride), and latex (natural rubber); organic solvents such as kerosene increase the rate of skin absorption.[1] PCB-resistant materials include Viton, polyethylene, polyvinyl acetate (PVA), polytetrafluoroethylene (PTFE), butyl rubber, nitrile rubber, and Neoprene.[1]
PCBs are very stable compounds and do not degrade readily. They may be destroyed by chemical, thermal, and biochemical processes, though it is extremely difficult to achieve full destruction, and there is the risk of creating extremely toxic dibenzodioxins and dibenzofurans through partial oxidation. Because of the high thermodynamic stability of PCBs, all degradation mechanisms are difficult to sustain. Intentional degradation as a treatment of unwanted PCBs generally requires high heat or catalysis. Environmental and metabolic degradation generally proceeds quite slowly relative to most other compounds.
History
PCBs were commercially produced as complex mixtures containing multiple isomers at different degrees of chlorination. In the United States, commercial production of PCBs was taken over in 1929, by Monsanto from Swann Chemical Company. Manufacturing levels increased in response to the electrical industry's need for a "safer" cooling and insulating fluid for industrial transformers and capacitors. PCBs were also commonly used as stabilizing additives in the manufacture of flexible PVC coatings for electrical wiring and electronic components to enhance the heat and fire resistance of the PVC.[2]
PCBs are persistent organic pollutants and have entered the environment through both use and disposal. The environmental transport of PCBs is complex and nearly global in scale. The public, legal, and scientific concerns about PCBs arose from research indicating they were likely carcinogens having the potential to adversely impact the environment and therefore undesirable as commercial products. Despite active research spanning five decades, extensive regulatory actions, and an effective ban on their production since the 1970s, PCBs still persist in the environment and remain a focus of attention.
The only North American producer, Monsanto, marketed PCBs under the trade name Aroclor, from 1930 to 1977. These were sold under trade names followed by a 4 digit number. The first two digits generally refer to the number of carbon atoms in the biphenyl skeleton (for PCBs this is 12), the second two numbers indicate the percentage of chlorine by mass in the mixture. Thus, Aroclor 1260 has 12 carbon atoms and contains 60 percent chlorine by mass. An exception is Aroclor 1016, which also has 12 carbon atoms, but has 42 percent chlorine by mass. PCB mixtures have been used for a variety of applications, including dielectric fluids for capacitors and transformers, heat transfer fluids, hydraulic fluids, lubricating and cutting oils, and as additives in pesticides, paints, carbonless copy ("NCR") paper, adhesives, sealants, plastics, reactive flame retardants, and as a fixative for microscopy. They were also used in surgical implants.
Manufacture peaked in the 1960s, by which time the electrical industry had lobbied the U.S. Congress to make them mandatory safety equipment, knowing all the while that they were extremely toxic. In 1966, they were determined by Swedish chemist Dr. Soren Jensen to be an environmental contaminant, and it was Dr. Jensen, according to a 1994 article in Sierra, who named them. Previously, they had simply been called "phenols" or referred to by various trade names, such as Aroclor, Kennechlor, Pyrenol, and others.
Their commercial utility was based largely on their chemical stability, including low flammability, and desirable physical properties, including electrical insulating properties. Their chemical and physical stability has also been responsible for their continuing persistence in the environment, and the lingering interest decades after regulations were imposed to control environmental contamination.
From 1973, their use was banned in "open" or "dissipative" sources, such as:
- Plasticisers in paints and cements
- Casting agents
- Fire retardant fabric treatments and heat stabilizing additives for PVC electrical insulation
- Adhesives
- Paints and water-proofing
- Railway sleepers
However, they continued to be allowed in "totally enclosed uses" such as transformers and capacitors, which, however, tended to explode on a fairly regular basis. It was Ward B. Stone of the New York State Department of Environmental Conservation who first published his findings, in the early 1970s, that PCBs were leaking from transformers and had contaminated the soil at the bottom of utility poles. Concern over the toxicity and persistence (chemical stability) of PCBs in the environment led the United States Congress to ban their domestic production in 1977, although some use continues in closed systems, such as capacitors and transformers.
The use of PCBs in "closed" uses include:
- Capacitors
- Insulating fluids in transformers
- Vacuum pump fluids
- Hydraulic fluids
Closed uses were also banned in the UK from 1981, when nearly all UK PCB synthesis ceased. Globally, probably 1 million tons of PCBs were manufactured in total.
Large scale contamination
The General Electric Co. discharged between 209,000 and 1.3 million pounds (94,800 and 590,000 kg) of PCBs into the Hudson River from two capacitor manufacturing plants located in Hudson Falls, New York, and Fort Edward, New York. Since that time, the spread of PCBs throughout the river and its food chain has created an extensive toxic waste problem. About 200 miles of the river is designated as a Superfund site. In 1976, because of the concern over the bioaccumulation of PCBs in fish and other aquatic organisms and their subsequent consumption by people, the state of New York banned fishing in the Upper Hudson River and commercial fishing of striped bass, and several other species, in the Lower Hudson. In August 1995, the Upper Hudson was re-opened to fishing, but only on a catch and release basis.
From the late 1950s through 1977, Westinghouse Electric used PCBs in the manufacture of capacitors in its Bloomington, Indiana, plant. Reject capacitors were hauled and dumped in area salvage yards and landfills. Workers also dumped PCB oil down factory drains which contaminated the city sewage treatment plant. The City of Bloomington gave away the sludge to area farmers and gardeners, creating anywhere from 200 to 2000 sites which remain unaddressed. Over 2 million pounds of PCBs were estimated to have been dumped in Monroe and Owen Counties, making it the biggest concentration of PCBs in the world. Although federal and state authorities have been working on the site remediations, many areas remain contaminated. Concerns have been raised regarding the removal of PCBs from the karst limestone topography, and regarding the possible disposal options. To date, the Westinghouse Bloomington PCB Superfund site case does not have a RI/FS (Remedial Investigation/Feasibility Study) and ROD (Record of Decision), although Westinghouse signed a U.S. Department of Justice Consent Decree in 1985.
PCBs have been detected globally, from the most urbanized areas that are the centers for PCB pollution, to regions north of the Arctic Circle. Typical urban atmospheric concentrations are in the picogram per cubic meter range. The atmosphere serves as the primary route for global transport of PCBs, particularly for those congeners with 1 to 4 chlorine atoms.
Health effects
The toxicity of PCBs to animals was first noticed in the 1970s, when emaciated seabird corpses with very high PCB body burdens were washed up on beaches. The source(s) of the PCBs was (were) unknown though, because seabirds may die at sea and be washed ashore from a very wide area. Where they were found was no reliable indicator of where they had died.
The toxicity of PCBs varies considerably among congeners. The coplanar PCBs, known as non-ortho PCBs because they are not substituted at the ring positions ortho to (next to) the other ring, (that is, PCBs 77, 126, 169, and so on), tend to have dioxin-like properties, and generally are among the most toxic congeners. Because PCBs are almost invariably found in complex mixtures, the concept of toxic equivalency factors (TEFs) has been developed to facilitate risk assessment and regulatory control, where more toxic PCB congeners are assigned higher TEF values. One of the most toxic compounds known, 2,3,7,8-tetrachlorodibenzo[p]dioxin, is assigned a TEF of 1.[3]
Symptoms
- Humans
The most commonly observed health effects in people exposed to large amounts of PCBs are skin conditions such as chloracne and rashes, but these were known to be symptoms of systemic poisoning dating back to the 1920s. Studies in exposed workers have shown changes in blood and urine that may indicate liver damage. PCB exposures in the general population are not likely to result in skin and liver effects. Most of the studies of health effects of PCBs in the general population examined children of mothers who were exposed to PCBs.
- Animals
Animals that ate food containing large amounts of PCBs for short periods of time had mild liver damage and some died. Animals that ate smaller amounts of PCBs in food over several weeks or months developed various kinds of health effects, including anemia; acne-like skin conditions (chloracne); and liver, stomach, and thyroid gland injuries (including hepatocarcinoma). Other effects of PCBs in animals include changes in the immune system, behavioral alterations, and impaired reproduction. PCBs are not known to cause birth defects in humans, although those that have dioxin-like activity are known to cause a variety of teratogenic effects in animals.
- Effects during pregnancy/breastfeeding
Women who were exposed to relatively high levels of PCBs in the workplace or ate large amounts of fish contaminated with PCBs had babies that weighed slightly less than babies from women who did not have these exposures. Babies born to women who ate PCB-contaminated fish also showed abnormal responses in tests of infant behavior. Some of these behaviors, such as problems with motor skills and a decrease in short-term memory, lasted for several years. Other studies suggest that the immune system was affected in children born to and nursed by mothers exposed to increased levels of PCBs. The most likely way infants will be exposed to PCBs is from breast milk. Transplacental transfers of PCBs were also reported. Because an infant will receive more than ten times the amount of PCBs from breast milk than it will for the rest of its life, it is being debated whether the benefits of breast-feeding are greater than the risks from exposure to PCBs.
Studies have shown that PCBs alter estrogen levels in the body and contribute to reproduction problems. In the womb, males can be feminized or the baby may be intersex, neither a male nor a female. Also, both sets of reproductive organs may develop. More instances of this are being reported. Biological magnification of PCBs has also led to polar bears and whales that have both male and female sex organs and males that cannot reproduce. This effect is also known as endocrine disruption. Endocrine Disrupting Chemicals (EDC's) pose a serious threat to reproduction in top-level predators.
Cancer link
A few studies of workers indicate that PCBs were associated with specific kinds of cancer in humans, such as cancer of the liver and biliary tract. Rats that ate food containing high levels of PCBs for two years developed liver cancer. The Department of Health and Human Services (DHHS) has concluded that PCBs may reasonably be anticipated to be carcinogens. The U.S. Environmental Protection Agency (EPA) and the International Agency for Research on Cancer (IARC) have determined that PCBs are probably carcinogenic to humans. PCBs are also classified as probable human carcinogens by the National Cancer Institute, World Health Organization, and the Agency for Toxic Substances and Disease Registry. Recent research by the National Toxicology Program has confirmed that PCB126 (Technical Report 520) and a binary mixture of PCB126 and PCB153 (Technicial Report 531) are carcinogens.
Mechanism of action
As discussed, PCBs exhibit a wide range of toxic effects. These effects may vary depending on the specific PCB. Toxicity of coplanar PCBs (such as dioxin) and mono-ortho-PCBs are thought to be primarily mediated via binding to aryl hydrocarbon receptor (AhR).[4] Because AhR is a transcription factor, abnormal activation may disrupt cell function by altering the transcription of genes. The concept of toxic equivalency factors (TEF) is based on the ability of a PCB to activate AhR.
However, not all effects may be mediated by the AhR receptor. For example, di-ortho-substituted non-coplanar PCBs interfere with intracellular signal transduction dependent on calcium; this may lead to neurotoxicity.[5] Ortho-PCBs may disrupt thyroid hormone transport by binding to transthyretin.[6]
Containment
Landfill—Large quantities of PCBs have been placed in landfill sites, mainly in the form of transformers and capacitors. Many municipal sites are not designed to contain these pollutants and PCBs are able to escape into the atmosphere or ground water. No emissions above background are seen if the landfill is designed correctly.
Methods of destruction
These can be separated into three distinct categories: physical, microbial, and chemical destruction.
Physical
Incineration—Although PCBs do not ignite themselves, they can be combusted under extreme and carefully controlled conditions. The current regulations require that PCBs are burnt at a temperature of 1200 °C for at least two seconds, in the presence of fuel oil and excess oxygen. A lack of oxygen can result in the formation of PCDDs, PCDFs, and dioxins, or the incomplete destruction of the PCBs. Such specific conditions mean that it is extremely expensive to destroy PCBs on a tonnage scale, and it can only be used on PCB containing equipment and contaminated liquid. This method is not suitable for the decontamination of affected soils.
Ultrasound—In a similar process to combustion, high power ultrasonic waves are applied to water, generating cavitation bubbles. These then implode or fragment, creating microregions of extreme pressures and temperatures where the PCBs are destroyed. Water is thought to undergo thermolysis, oxidising the PCBs to CO, CO2 and hydrocarbons such as biphenyl, with chlorine present as the inorganic ion 16. The scope of this method is limited to those congeners which are the most water soluble; those isomers with the least chlorine substitution.
Irradiation—If a deoxygenated mixture of PCBs in isopropanol or mineral oil is subject to irradiation with gamma rays then the PCBs will be dechlorinated to form inorganic chloride and biphenyl. The reaction works best in isopropanol if potassium hydroxide (caustic potash) is added. Solvated electrons are thought to be responsible for the reaction. If oxygen, nitrous oxide, sulfur hexafluoride, or nitrobenzene is present in the mixture then the reaction rate is reduced. This work has been done recently in the U.S. often with used nuclear fuel as the radiation source.
Microbial
Much recent work has centered on the study of micro-organisms that are able to decompose PCBs. Generally, these organisms work in one of two ways: Either they use the PCB as a carbon source, or destruction takes place through reductive dechlorination, with the replacement of chlorine with hydrogen on the biphenyl skeleton. However, there are significant problems with this approach. Firstly, these microbes tend to be highly selective in their dechlorination, with lower chlorinated biphenyls being readily transformed, and with preference to dechlorination in the para and meta positions. Secondly, microbial dechlorination tends to be rather slow acting on PCB as a soil contaminant in comparison to other methods. Finally, while microbes work well in laboratory conditions, there is often a problem in transferring a successful laboratory strain to a natural system. This is because the microbes can access other sources of carbon, which they decompose in preference to PCBs. Further recent developments have focused on testing enzymes and vitamins extracted from microbes which show PCB activity. Especially promising seems to be the use of vitamin B12, in which a cobalt ion is in oxidation state (III) under normal redox conditions. Using titanium (III) citrate as a strong reductant converts the cobalt from Co(III) to Co(I), giving a new vitamin known as B12s, which is a powerful nucleophile and reducing catalyst. This can then be used on PCBs, which it dechlorinates in a rapid and selective manner.
Chemical
Many chemical methods are available to destroy or reduce the toxicity of PCBs.
Aromatic nucleophilic substitution is a method of destroying low concentration PCB mixtures in oils, such as transformer oil. Substitution of chlorine by poly (ethylene glycols) occurs in under two hours under a blanket of nitrogen, to prevent oxidation of the oil, to produce aryl polyglycols, which are insoluble in the oil and precipitate out.
Between 700 and 925 °C, H2 cleaves the carbon-chlorine bond, and cleaves the biphenyl nucleus into benzene yielding HCl without a catalyst. This can be performed at lower temperatures with a copper catalyst, and to yield biphenyl. However, since both of these routes require an atmosphere of hydrogen gas and relatively high temperatures they are prohibitively expensive.
Reaction with highly electropositive metals, or strong reducing agents such as sodium naphthalide, in aprotic solvents results in a transfer of electrons to the PCB, the expulsion of a chloride ion, and a coupling of the PCBs. This is analogous to the Wurtz Reaction for coupling halogenoalkanes. The effect is to polymerize many molecules, therefore reducing the volatility, solubility, and toxicity of the mixture. This methodology is most successful on low strength PCB mixtures and can also be performed electrochemically in a partly aqueous bicontinuous microemulsion.
The solution photochemistry of PCBs is based on the transfer of an electron to a photochemically excited PCB from a species such as an amine, to give a radical anion. This either expels a chloride ion and the resulting aryl radical extracts a hydrogen atom from the solvent, or immediately becomes protonated, leading to the loss of a chlorine atom. It is useful only for water soluble PCBs.
The major pathway for atmospheric destruction of PCBs is via attack by OH radicals. Direct photolysis can occur in the upper atmosphere, but the ultraviolet wavelengths necessary to excite PCBs are shielded from the troposphere by the ozone layer. It has, however, been shown that higher wavelengths of light (greater than 300 nm) can degrade PCBs in the presence of a photosensitizer, such as acetone.
The Schwartz reaction is the subject of much study, and has significant benefits over other routes. It is advantageous since it proceeds via a reductive process, and thus yields no dioxins through oxidation. The proposed reaction scheme involves the electron transfer from a titanium (III) organometallic species to form a radical anion on the PCB molecule which expels chlorine to eventually form the relatively non-toxic biphenyl.
Appendix
PCB Homolog Table
For a complete list of PCB congeners, see PCB Congener List. Note that biphenyl, while not technically a PCB congener due to its lack of chlorine substituents, is still typically included in the literature.
| PCB Homolog | CASRN | Cl Substituents | Congeners |
|---|---|---|---|
| Biphenyl | 92-52-4 | 0 | 1 |
| Monochlorobiphenyl | 27323-18-8 | 1 | 3 |
| Dichlorobiphenyl | 25512-42-9 | 2 | 12 |
| Trichlorobiphenyl | 25323-68-6 | 3 | 24 |
| Tetrachlorobiphenyl | 26914-33-0 | 4 | 42 |
| Pentachlorobiphenyl | 25429-29-2 | 5 | 46 |
| Hexachlorobiphenyl | 26601-64-9 | 6 | 42 |
| Heptachlorobiphenyl | 28655-71-2 | 7 | 24 |
| Octachlorobiphenyl | 55722-26-4 | 8 | 12 |
| Nonachlorobiphenyl | 53742-07-7 | 9 | 3 |
| Decachlorobiphenyl | 2051-24-3 | 10 | 1 |
Notes
- ↑ 1.0 1.1 ANZECC, Identifying PCB-Containing Capacitors (Parkes, AU: Australian and New Zealand Environment and Conservation Council, ISBN 0642545073). Retrieved November 4, 2007.
- ↑ Karlyn Black Kaley, Jim Carlisle, David Siegel, and Julio Salinas, Health Concerns and Environmental Issues with PVC-Containing Building Materials in Green Buildings, Integrated Waste Management Board, California Environmental Protection Agency, U.S. Retrieved November 4, 2007.
- ↑ M. Van den Berg, Environ Health Perspect. 106(12): 775-92.
- ↑ S. Safe, Polychlorinated biphenyls (PCBs) and polybrominated biphenyls (PBBs): Biochemistry, toxicology, and mechanism of action. Crit. Rev. Toxicol. 13(4): 319-95.
- ↑ T. Simon, J.K. Britt, and R.C. James, Development of a neurotoxic equivalence scheme of relative potency for assessing the risk of PCB mixtures. Regul Toxicol Pharmacol. 48(2): 148-70.
- ↑ K.R. Chauhan, P.R. Kodavanti, and J.D. McKinney, Assessing the role of ortho-substitution on polychlorinated biphenyl binding to transthyretin, a thyroxine transport protein. Toxicol. Appl. Pharmacol. 162(1): 10-21.
ReferencesISBN links support NWE through referral fees
- Albaiges, Juan. 1993. Environmental Analytical Chemistry of PCBs (Current Topics in Environmental and Toxicological Chemistry). Langhorne, PA: Gordon and Breach Science. ISBN 2881249388.
- Erickson, Mitchell D. 1997. Analytical Chemistry of PCBs. Boca Raton, FL: CRC. ISBN 0873719239.
- Erickson, Mitchell D. 1993. Remediation of PCB Spills. Boca Raton, FL: CRC. ISBN 087371945X.
External links
All links retrieved November 24, 2022.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.