Naphthalene
| Naphthalene | |
|---|---|
| |
| General | |
| Chemical name | Naphthalene |
| Other names | Tar Camphor, White Tar, Moth Flakes |
| Chemical formula | C10H8 |
| SMILES | c1cccc2c1cccc2 |
| Molar mass | 128.17052 g/mol |
| Appearance | White solid crystals/flakes, strong odor of coal tar |
| CAS number | 91-20-3 |
| Properties | |
| Density | 1.14 g/cm³ |
| Solubility in water | approximately 30mg/L |
| Melting point | 80.2 °C |
| Boiling point | 218 °C |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | Flammable, sensitizer, possible carcinogen. Dust can form explosive mixtures with air |
| NFPA 704 | |
| Flash point | 79-87 °C |
| Autoignition temperature | 525 °C |
| R/S statement | R: 22, 40, 50/53 S: 2, 36/37, 46, 60, 61 |
| RTECS number | QJ0525000 |
| Except where noted otherwise, data are given for materials in their standard state (at 25°C, 100 kPa) | |
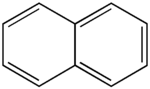
Naphthalene (also known as naphthalin, naphthaline, moth ball, tar camphor, white tar, or albocarbon), is a crystalline, aromatic, white, solid hydrocarbon, best known as the primary ingredient of mothballs. Naphthalene is volatile, forming a flammable vapor. Each molecule of this substance consists of two fused benzene rings. As its most abundant single component, it is obtained from coal tar and converted to phthalic anhydride for the manufacture of plastics, dyes, and solvents. It is also used as an antiseptic and insecticide, especially in mothballs (p-Dichlorobenzene may be used instead of naphthalene as a mothball substitute). Naphthalene easily sublimates at room temperature.
Occurrence in nature
Trace amounts of naphthalene are produced by magnolias and certain types of deer. In addition, this chemical has been found in the Formosan subterranean termite, possibly as a repellent against "ants, poisonous fungi, and nematode worms." [1]
History
In 1819-1820, at least two chemists reported a white solid with a pungent odor derived from the distillation of coal tar. In 1821, John Kidd described many of this substance's properties and the means of its production, and proposed the name naphthaline, as it had been derived from a kind of naphtha (a broad term encompassing any volatile, flammable liquid hydrocarbon mixture, including coal tar). [2] Naphthalene's melting point between 79 to 83 degrees Celsius and has a density of 1.14 g/cm³. Along with being highly flammable, naphthalene is harmful to both humans and nature.
Naphthalene's molecular formula, C10H8, was determined by Michael Faraday in 1826. The structure of two fused benzene rings was proposed by Emil Erlenmeyer in 1866, and confirmed by Carl Graebe three years later.
Notable characteristics
Structure
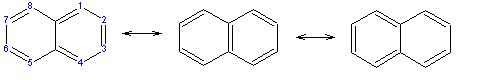
As noted above, a naphthalene molecule is composed of two fused benzene rings. (In organic chemistry, rings are "fused" if they share two or more atoms.) Accordingly, naphthalene is classified as a benzenoid polyaromatic hydrocarbon (PAH). Naphthalene has three resonance structures, which are shown in the drawing below. Naphthalene has two sets of equivalent hydrogens. The alpha positions are positions 1, 4, 5, and 8 on the drawing below. The beta positions are positions 2, 3, 6, and 7.
Unlike benzene, the carbon-carbon bonds in naphthalene are not of the same length. The bonds C1–C2, C3–C4, C5–C6 and C7–C8 are about 1.36 Å (136 pm) in length, whereas all the other carbon-carbon bonds are about 1.42 Å (142 pm) in length. This has been verified by x-ray diffraction and can be expected from the resonance structures, where the bonds C1–C2, C3–C4, C5–C6 and C7–C8 are double in two of the three structures, whereas all the others are double in only one.
Reactivity
Like benzene, naphthalene can undergo electrophilic aromatic substitution. For many electrophilic aromatic substitution reactions, naphthalene is more reactive than benzene, and reacts under milder conditions than does benzene. For example, while both benzene and naphthalene react with chlorine in the presence of a ferric chloride or aluminum chloride catalyst, naphthalene and chlorine can react to form 1-chloronaphthalene even without a catalyst. Similarly, while both benzene and naphthalene can be alkylated using Friedel-Crafts reactions, naphthalene can also be alkylated by reaction with alkenes or alcohols, with sulfuric or phosphoric acid as the catalyst.
Mono-substitution of naphthalene has two possible isomeric products, corresponding to substitution at an alpha or beta position, respectively. Usually, the major product has the electrophile in the alpha position. The selectivity for alpha over beta substitution can be rationalized in terms of the resonance structures of the intermediate: for the alpha substitution intermediate, seven resonance structures can be drawn, of which four preserve an aromatic ring. For beta substitution, the intermediate has only six resonance structures, and only two of these are aromatic. Sulfonation, however, gives a mixture of the "alpha" product 1-naphthalenesulfonic acid and the "beta" product 2-naphthalenesulfonic acid, with the ratio dependent on reaction conditions.
Naphthalene can be hydrogenated under high pressure or with a suitable catalyst to give 1,2,3,4-tetrahydronaphthalene, a solvent sold under the trade name Tetralin. Further hydrogenation yields decahydronaphthalene or Decalin (C10H18, also known as bicyclodecane). Oxidation of naphthalene with chromate or permanganate, or catalytic oxidation with O2 and a vanadium catalyst, gives phthalic acid.
Production
Most naphthalene is derived from coal tar. From the 1960s until the 1990s, significant amounts of naphthalene were also produced from heavy petroleum fractions during petroleum refining, but today, petroleum-derived naphthalene represents only a minor component of naphthalene production.
Although the composition of coal tar varies with the coal from which it is produced, typical coal tar is about 10% naphthalene by weight. In industrial practice, distillation of coal tar yields an oil containing about 50 percent naphthalene, along with a variety of other aromatic compounds. This oil, after being washed with aqueous sodium hydroxide to remove acidic components, chiefly various phenols, and with sulfuric acid to remove basic components, is fractionally distilled to isolate naphthalene. The crude naphthalene resulting from this process is about 95 percent naphthalene by weight. The chief impurity is the sulfur-containing aromatic compound benzothiophene. Petroleum-derived naphthalene is usually purer than that derived from coal tar. Where purer naphthalene is required, crude naphthalene can be further purified by recrystallizing it from any of a variety of solvents.
Uses
Naphthalene's most familiar use is as a household fumigant, such as in mothballs. In a sealed container of naphthalene pellets, naphthalene vapors build up to levels toxic to both the adult and larval forms of many moths that are destructive to textiles. Other fumigant uses of naphthalene include use in soil as a fumigant pesticide, and in attic spaces to repel animals.
In the past, naphthalene was administered orally to kill parasitic worms in livestock.
Larger volumes of naphthalene are used as a chemical intermediate to produce other chemicals. The single largest use of naphthalene is the industrial production of phthalic anhydride, although more phthalic anhydride is made from o-xylene than from naphthalene. Other naphthalene-derived chemicals include alkyl naphthalene sulfonate surfactants, and the insecticide carbaryl. Naphthalenes substituted with combinations of strongly electron-donating functional groups, such as alcohols and amines, and strongly electron-withdrawing groups, especially sulfonic acids, are intermediates in the preparation of many synthetic dyes. The hydrogenated naphthalenes tetrahydronaphthalene (Tetralin) and decahydronaphthalene (Decalin) are used as low-volatility solvents.
Naphthalene vapor can also slow the onset of rust, and, thus, sometimes moth balls are used in places like a tool box.
Health effects
In humans, exposure to large amounts of naphthalene may damage or destroy red blood cells. This could cause the body to have too few red blood cells until it replaces the destroyed cells. Humans, particularly children, have developed this condition after ingesting mothballs or deodorant blocks containing naphthalene. Some of the symptoms of this condition are fatigue, lack of appetite, restlessness, and pale skin. Exposure to large amounts of naphthalene may also cause nausea, vomiting, diarrhea, blood in the urine, and jaundice (yellow coloration of the skin).
Researchers with the U.S. National Toxicology Program exposed male and female rats and mice to naphthalene vapors on weekdays for two years. They found that female mice exhibited some evidence of carcinogenic activity, based on increased incidence of alveolar and bronchiolar adenomas of the lung; but male mice exhibited no evidence of carcinogenic activity. In both male and female mice, naphthalene led to greater incidences and severity of chronic inflammation and abnormalities in the tissues lining the nose and lungs.[3] Regarding rats, they found clear evidence of carcinogenic activity of naphthalene in male and female rats based on increased incidences of respiratory epithelial adenoma and olfactory epithelial neuroblastoma of the nose. Exposure to naphthalene caused significant increases in the incidences of nonneoplastic lesions of the nose in both male and female rats.[4]
Over 400 million people have an inherited condition called glucose-6-phosphate dehydrogenase deficiency (G6PD deficiency). For these people, exposure to naphthalene is harmful and may cause hemolytic anemia, which causes their erythrocytes to break down.
The International Agency for Research on Cancer (IARC) classifies naphthalene as possibly carcinogenic to humans [Group 2B]. It also points out that acute exposure causes cataracts in humans, rats, rabbits, and mice. Furthermore, hemolytic anemia can occur in children and infants after oral or inhalation exposure, or after maternal exposure during pregnancy.
Notes
- ↑ BBC News, Termite 'mothball' keep insects at bay. Retrieved September 27, 2016.
- ↑ John Kidd (1821). Obersvations on Naphthaline, a peculiar substance resembling a concrete essential oil, which is apparently produced during the decomposition of coal tar, by exposure to a red heat. Philosophical Transactions 111: 209-221.
- ↑ National Toxicology Program, NTP Study Report TR-410 Retrieved September 27, 2016.
- ↑ National Toxicology Program, NTP Study Report TR-500 Retrieved September 27, 2016.
ReferencesISBN links support NWE through referral fees
- McMurry, John. Organic Chemistry, 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry, 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692
- Solomons, T.W. Graham and Craig B. Fryhle. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998
External links
All links retrieved June 2, 2025.
- NIOSH Pocket Guide to Chemical Hazards: Naphthalene.
- Naphthalene International Programme on Chemical Safety, IPCS INCHEM.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.