Difference between revisions of "Acetone" - New World Encyclopedia
Rosie Tanabe (talk | contribs) |
|||
| (22 intermediate revisions by 7 users not shown) | |||
| Line 1: | Line 1: | ||
| − | {{ | + | {{Copyedited}}{{Images OK}}{{Submitted}}{{Approved}}{{Paid}} |
| − | {{ | ||
| − | {| align="right" border="1" cellspacing="0" cellpadding="3" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-collapse: collapse; border-color: #C0C090;" | + | {|class="infobox" width="350" style="float:right;"margin:0 0 1em 1em;" align="right" border="1" cellspacing="0" cellpadding="3" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-collapse: collapse; border-color: #C0C090;" |
| − | ! {{chembox header}} | Acetone | + | ! {{chembox header}} | Acetone |
|- | |- | ||
| align="center" colspan="2" | [[Image:Acetone-skeletal.png|100px|Acetone]] [[Image:Acetone-3D-balls.png|100px|Ball-and-stick model of acetone]] [[Image:Acetone-3D-vdW.png|100px|Space-filling model of acetone]] | | align="center" colspan="2" | [[Image:Acetone-skeletal.png|100px|Acetone]] [[Image:Acetone-3D-balls.png|100px|Ball-and-stick model of acetone]] [[Image:Acetone-3D-vdW.png|100px|Space-filling model of acetone]] | ||
| Line 59: | Line 58: | ||
| 2.91 [[Debye|D]] | | 2.91 [[Debye|D]] | ||
|- | |- | ||
| − | ! {{chembox header}} | Hazards <!-- | + | ! {{chembox header}} | Hazards <!-- Summary only- MSDS entry provides more complete information —> |
|- | |- | ||
| [[Material safety data sheet|MSDS]] | | [[Material safety data sheet|MSDS]] | ||
| Line 71: | Line 70: | ||
<!-- {{Chembox/NFPA|1|3|2}} seems to commonly be assigned to less pure grades of acetone —> | <!-- {{Chembox/NFPA|1|3|2}} seems to commonly be assigned to less pure grades of acetone —> | ||
|- | |- | ||
| − | | | + | | R-phrases |
| {{R11}}, {{R36}}, {{R66}}, {{R67}} | | {{R11}}, {{R36}}, {{R66}}, {{R67}} | ||
|- | |- | ||
| − | | | + | | S-phrases |
| {{S2}}, {{S9}}, {{S16}}, {{S26}} | | {{S2}}, {{S9}}, {{S16}}, {{S26}} | ||
|- | |- | ||
| Line 80: | Line 79: | ||
| −20 °C | | −20 °C | ||
|- | |- | ||
| − | | [[Flammable limit]]s<br>in air (by volume) | + | | [[Flammable limit]]s<br/>in air (by volume) |
| 2.55% - 12.80% | | 2.55% - 12.80% | ||
|- | |- | ||
| Line 95: | Line 94: | ||
|- | |- | ||
| [[Acetone (data page)#Thermodynamic properties|Thermodynamic data]] | | [[Acetone (data page)#Thermodynamic properties|Thermodynamic data]] | ||
| − | | Phase behaviour<br>Solid, liquid, gas | + | | Phase behaviour<br/>Solid, liquid, gas |
|- | |- | ||
| [[Acetone (data page)#Spectral data|Spectral data]] | | [[Acetone (data page)#Spectral data|Spectral data]] | ||
| Line 108: | Line 107: | ||
| [[water (molecule)|Water]]<br/>[[Ethanol]]<br/>[[Isopropanol]]<br/>[[Toluene]] | | [[water (molecule)|Water]]<br/>[[Ethanol]]<br/>[[Isopropanol]]<br/>[[Toluene]] | ||
|- | |- | ||
| − | | {{chembox header}} | <small>Except where noted otherwise, data are given for<br> materials in their [[standard state|standard state (at | + | | {{chembox header}} | <small>Except where noted otherwise, data are given for<br/> materials in their [[standard state|standard state (at 25 °C, 100 kPa)]]</small> |
|- | |- | ||
|} | |} | ||
| − | + | '''Acetone''' (also known as '''propanone''', '''dimethyl ketone''', '''2-propanone''', '''propan-2-one''' and '''β-ketopropane''') is the simplest representative of the group of [[chemical compound]]s known as ''[[ketone]]s''. It is a colorless, volatile, flammable liquid. In addition to being manufactured as a chemical, acetone is also found naturally in the environment, including in small amounts in the human body. | |
| + | {{toc}} | ||
| + | Acetone is a highly effective [[solvent]] for many organic compounds and is the active ingredient in [[nail polish]] remover. It is also used to make various [[plastic]]s, [[fiber]]s, [[drug]]s, and other chemicals. | ||
| − | + | == Occurrence in the human body == | |
| − | + | Small amounts of acetone are metabolically produced in the body, mainly from fat. Fasting significantly increases its endogenous production (see [[ketosis]]). Acetone can be elevated in cases of [[diabetes]]. | |
| − | + | == Chemical synthesis == | |
| + | |||
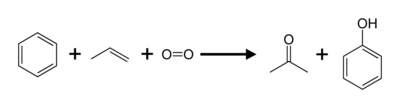
| + | Most of the worldwide industrial production of acetone (and [[phenol]]) is currently based on a method called the '''cumene process.''' This process converts two relatively cheap starting materials, [[benzene]] and [[propylene]], into acetone and phenol. Another reactant is [[oxygen]] (from the air).<ref>A small amount of a [[radical initiator]] is added to start the reaction.</ref> The reaction is named after [[cumene]] (isopropyl benzene), the intermediate material formed during the process. | ||
| + | |||
| + | The overall reaction may be written as follows: | ||
| + | |||
| + | [[Image:Cumene-process-overview-2D-skeletal.png|400px|center|The overall cumene process.]] | ||
| + | |||
| + | Before the invention of the [[cumene process]], acetone was produced by the dry [[distillation]] of [[acetate]]s, such as [[calcium acetate]]. | ||
| + | |||
| + | During [[World War I]], a method of producing acetone through [[bacterium|bacterial]] [[Fermentation (biochemistry)|fermentation]] was developed by [[Chaim Weizmann]], (who later became the first president of [[Israel]]), to help the British war effort. | ||
== Characteristics == | == Characteristics == | ||
| − | Acetone has a melting point of −95.4 °C and boiling point of 56.53 °C. It has a [[relative density]] of 0.819 (at 0 °C). It acts as a [[solvent]] and is readily miscible with other solvents, including [[ | + | Acetone has a melting point of −95.4 °C and boiling point of 56.53 °C. It has a [[relative density]] of 0.819 (at 0 °C). It acts as a [[solvent]] and is readily miscible with other solvents, including [[water]], [[ethanol]], and [[ether|diethyl ether]]. |
== Uses == | == Uses == | ||
| − | An important industrial use for acetone involves its reaction with [[phenol]] | + | * An important industrial use for acetone involves its reaction with [[phenol]] to produce [[bisphenol A]], which is a valuable component of many polymers, such as [[polycarbonate]]s, [[polyurethane]]s, and [[epoxy resin]]s. |
| + | |||
| + | * Another industrial application involves its use as a general purpose cleaner in paint and ink manufacturing operations. | ||
| − | Acetone is | + | * Acetone is also used extensively for the safe transport and storage of [[acetylene]]. Vessels containing a porous material are first filled with acetone followed by acetylene, which dissolves into the acetone. One liter of acetone can dissolve around 250 liters of acetylene. |
| − | + | * It is often the primary (or only) component in [[nail polish]] remover.<ref>[[Acetonitrile]], another organic solvent, is sometimes used as well.</ref> It is also used as a [[superglue]] remover. It can be used for thinning and cleaning [[fiberglass]] [[resin]]s and [[epoxy|epoxies]], and it is highly effective in removing stains by permanent markers. | |
| − | |||
| − | + | * It can be used as an agent in art work. When rubbed on the back of any laser print or laser photocopy, it produces a rough ready effect. | |
| − | + | * It has been used in the manufacture of [[cordite]]. | |
| − | + | * It is a strong solvent for most [[plastic]]s (including those used in consumer-targeted water bottles) and synthetic fibers. | |
| − | + | * It is used as a [[desiccant|drying agent]], as it is readily miscible with water and is volatile. | |
| + | |||
| + | * In the laboratory, acetone is used as a [[chemical polarity|polar]] [[aprotic solvent]] (solvent that does not release hydrogen ions) in a variety of [[organic reaction]]s. | ||
==Health effects== | ==Health effects== | ||
| − | + | At relatively low concentrations, acetone is not very toxic. It can, however, irritate and damage [[skin]] and the [[mucosa]]l lining of the [[mouth]]. Its fumes should be avoided, as inhalation may lead to liver damage. In addition, one should always wear goggles when handling the substance, as it can cause permanent eye damage ([[corneal clouding]]). | |
| − | + | Contamination of water, food (such as milk), or air (by acetone vapors) can lead to chronic exposure to acetone. A number of acute poisoning cases have been described. Accidental intake of large amounts of acetone may lead to unconsciousness and death. | |
| − | + | Animal studies have shown that long-term exposure to acetone can damage the [[kidney]]s, [[liver]], and [[nerve]]s, increase [[birth defect]]s, and lower the reproductive capacity of males (only). It is not known if the same effects would occur in humans. Pregnant women should avoid contact with acetone and its fumes, to avoid the possibility of birth defects, including brain damage. | |
| − | Interestingly, acetone has been shown to have [[anticonvulsant]] effects in animal models of [[epilepsy]], in the absence of toxicity, when administered | + | Interestingly, acetone has been shown to have [[anticonvulsant]] effects in animal models of [[epilepsy]], in the absence of toxicity, when administered at low (millimolar) concentrations.<ref name="Likhodii">Likhodii et al., [https://www.ncbi.nlm.nih.gov/pubmed/12891674 Anticonvulsant properties of acetone, a brain ketone elevated by the ketogenic diet] ''Ann Neurol.'', 54(2) (2003): 219–226. Retrieved April 3, 2018.</ref> It has been hypothesized that the high-fat, low-carbohydrate, [[ketogenic diet]] used clinically to control drug-resistant epilepsy in children works by elevating acetone in the brain.<ref name="Likhodii"/> |
| − | == | + | == Chemical safety measures == |
| − | Due to incompatibilities, it is recommended | + | |
| + | Due to chemical incompatibilities, it is recommended that acetone should be kept away from [[bromine]], [[chlorine]], [[nitric acid]], [[sulfuric acid]] and [[trichloromethane]]. | ||
== See also == | == See also == | ||
| − | |||
* [[Ketone]] | * [[Ketone]] | ||
* [[Solvent]] | * [[Solvent]] | ||
| Line 164: | Line 178: | ||
==References== | ==References== | ||
| + | |||
| + | * McMurry, John. ''Organic Chemistry'', 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052. | ||
| + | * Morrison, Robert T., and Robert N. Boyd. ''Organic Chemistry'', 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692. | ||
| + | * Solomons, T. W. Graham, and Craig B. Fryhle. ''Organic Chemistry'', 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998. | ||
==External links== | ==External links== | ||
| − | + | All links retrieved June 14, 2023. | |
| − | *[http:// | + | * [http://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/aldket1.htm#aknom Aldehydes and Ketones] ''Virtual Textbook of Organic Chemistry''. |
| − | * [http://www. | + | * [http://www.cdc.gov/niosh/npg/npgd0004.html Acetone.] ''NIOSH Pocket Guide to Chemical Hazards''. |
| − | *[http:// | + | * [http://webbook.nist.gov/cgi/cbook.cgi?Name=acetone&Units=SI Acetone] ''NIST Chemistry WebBook''. |
| − | |||
[[Category:Physical sciences]] | [[Category:Physical sciences]] | ||
| Line 176: | Line 193: | ||
[[Category:Organic chemistry]] | [[Category:Organic chemistry]] | ||
| − | {{ | + | {{credits|Acetone|131088634|Cumene_process|142543683}} |
Latest revision as of 07:34, 14 June 2023
| Acetone | |
|---|---|
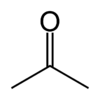  
| |
| General | |
| Systematic name | Propanone |
| Other names | β-ketopropane Dimethyl ketone, |
| Molecular formula | CH3COCH3 |
| SMILES | CC(=O)C |
| Molar mass | 58.09 g/mol |
| Appearance | Colorless liquid |
| CAS number | [67-64-1] |
| Properties | |
| Density and phase | 0.79 g/cm³, liquid |
| Solubility in water | miscible |
| Melting point | −94.9 °C (178.2 K) |
| Boiling point | 56.3 °C (329.4 K) |
| Viscosity | 0.32 cP at 20 °C |
| Structure | |
| Molecular shape | trigonal planar at C=O |
| Dipole moment | 2.91 D |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Flammable (F) Irritant (Xi) |
| NFPA 704 | |
| R-phrases | R11, R36, R66, R67 |
| S-phrases | S2, S9, S16, S26 |
| Flash point | −20 °C |
| Flammable limits in air (by volume) |
2.55% - 12.80% |
| Autoignition temperature | 465 °C |
| RTECS number | AL31500000 |
| Supplementary data page | |
| Structure & properties | n, εr, etc. |
| Thermodynamic data | Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Related ketones | Butanone |
| Related solvents | Water Ethanol Isopropanol Toluene |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Acetone (also known as propanone, dimethyl ketone, 2-propanone, propan-2-one and β-ketopropane) is the simplest representative of the group of chemical compounds known as ketones. It is a colorless, volatile, flammable liquid. In addition to being manufactured as a chemical, acetone is also found naturally in the environment, including in small amounts in the human body.
Acetone is a highly effective solvent for many organic compounds and is the active ingredient in nail polish remover. It is also used to make various plastics, fibers, drugs, and other chemicals.
Occurrence in the human body
Small amounts of acetone are metabolically produced in the body, mainly from fat. Fasting significantly increases its endogenous production (see ketosis). Acetone can be elevated in cases of diabetes.
Chemical synthesis
Most of the worldwide industrial production of acetone (and phenol) is currently based on a method called the cumene process. This process converts two relatively cheap starting materials, benzene and propylene, into acetone and phenol. Another reactant is oxygen (from the air).[1] The reaction is named after cumene (isopropyl benzene), the intermediate material formed during the process.
The overall reaction may be written as follows:
Before the invention of the cumene process, acetone was produced by the dry distillation of acetates, such as calcium acetate.
During World War I, a method of producing acetone through bacterial fermentation was developed by Chaim Weizmann, (who later became the first president of Israel), to help the British war effort.
Characteristics
Acetone has a melting point of −95.4 °C and boiling point of 56.53 °C. It has a relative density of 0.819 (at 0 °C). It acts as a solvent and is readily miscible with other solvents, including water, ethanol, and diethyl ether.
Uses
- An important industrial use for acetone involves its reaction with phenol to produce bisphenol A, which is a valuable component of many polymers, such as polycarbonates, polyurethanes, and epoxy resins.
- Another industrial application involves its use as a general purpose cleaner in paint and ink manufacturing operations.
- Acetone is also used extensively for the safe transport and storage of acetylene. Vessels containing a porous material are first filled with acetone followed by acetylene, which dissolves into the acetone. One liter of acetone can dissolve around 250 liters of acetylene.
- It is often the primary (or only) component in nail polish remover.[2] It is also used as a superglue remover. It can be used for thinning and cleaning fiberglass resins and epoxies, and it is highly effective in removing stains by permanent markers.
- It can be used as an agent in art work. When rubbed on the back of any laser print or laser photocopy, it produces a rough ready effect.
- It has been used in the manufacture of cordite.
- It is a strong solvent for most plastics (including those used in consumer-targeted water bottles) and synthetic fibers.
- It is used as a drying agent, as it is readily miscible with water and is volatile.
- In the laboratory, acetone is used as a polar aprotic solvent (solvent that does not release hydrogen ions) in a variety of organic reactions.
Health effects
At relatively low concentrations, acetone is not very toxic. It can, however, irritate and damage skin and the mucosal lining of the mouth. Its fumes should be avoided, as inhalation may lead to liver damage. In addition, one should always wear goggles when handling the substance, as it can cause permanent eye damage (corneal clouding).
Contamination of water, food (such as milk), or air (by acetone vapors) can lead to chronic exposure to acetone. A number of acute poisoning cases have been described. Accidental intake of large amounts of acetone may lead to unconsciousness and death.
Animal studies have shown that long-term exposure to acetone can damage the kidneys, liver, and nerves, increase birth defects, and lower the reproductive capacity of males (only). It is not known if the same effects would occur in humans. Pregnant women should avoid contact with acetone and its fumes, to avoid the possibility of birth defects, including brain damage.
Interestingly, acetone has been shown to have anticonvulsant effects in animal models of epilepsy, in the absence of toxicity, when administered at low (millimolar) concentrations.[3] It has been hypothesized that the high-fat, low-carbohydrate, ketogenic diet used clinically to control drug-resistant epilepsy in children works by elevating acetone in the brain.[3]
Chemical safety measures
Due to chemical incompatibilities, it is recommended that acetone should be kept away from bromine, chlorine, nitric acid, sulfuric acid and trichloromethane.
See also
Notes
- ↑ A small amount of a radical initiator is added to start the reaction.
- ↑ Acetonitrile, another organic solvent, is sometimes used as well.
- ↑ 3.0 3.1 Likhodii et al., Anticonvulsant properties of acetone, a brain ketone elevated by the ketogenic diet Ann Neurol., 54(2) (2003): 219–226. Retrieved April 3, 2018.
ReferencesISBN links support NWE through referral fees
- McMurry, John. Organic Chemistry, 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052.
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry, 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692.
- Solomons, T. W. Graham, and Craig B. Fryhle. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998.
External links
All links retrieved June 14, 2023.
- Aldehydes and Ketones Virtual Textbook of Organic Chemistry.
- Acetone. NIOSH Pocket Guide to Chemical Hazards.
- Acetone NIST Chemistry WebBook.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.