Glycogen
Glycogen, a large, branched polymer of linked glucose (Glc) residues, is the principal storage form of glucose in animal cells, though it is also found in various species of microorganisms, such as bacteria and fungi. A readily mobilized energy store, glycogen increases the amount of glucose immediately available to the organism (1) between meals and (2) during muscular activity. The ability to maintain a steady supply of glucose, which is the major sugar circulating in the blood of higher animals, is crucial to survival since the brain relies on glucose as its preferred fuel.
Glycogen is found in the form of granules in the cytosol, the internal fluid of the cell. About three-fourths of the body’s glycogen supply is stored in muscle cells. However, liver cells (hepatocytes), have the highest concentration of glucose (a maximum of 8% in liver versus 1% of the muscle mass of an adult male human). Small amounts of glycogen are also found in the kidneys, and even smaller amounts in certain glial cells in the brain and in white blood cells.
Glycogen is utilized differently depending on the tissue in which it is stored:
- In liver, the breakdown of glycogen (glycogenolysis) and its synthesis (glycogenesis) help to regulate the blood glucose level. Glucose is not a major fuel for the liver; instead, it primarily exports glucose for the benefit of other tissues.
- In skeletal muscle, glycogen is an energy reserve that can be tapped during exercise. Muscle cells lack the ability to pass glucose into the blood, so the glycogen store is destined for internal use, powering muscle contraction during strenuous activity.
Glycogen-storage disorders are a type of inherited metabolic disease related to deficiencies of enzymes that participate in glycogen metabolism.
Glycogen's branched structure makes it an accessible energy source
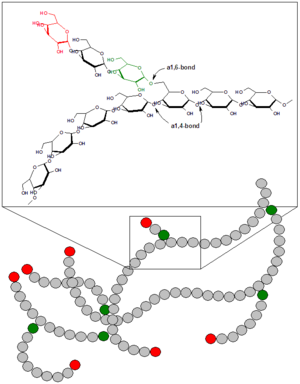
Glycogen is a highly-branched polymer of about 30,000 glucose residues. It has a molecular weight between 106 and 107 daltons. Most of the glucose units are linked by α-1,4 glycosidic bonds, which are formed between the hemiacetal group of a saccharide and the hydroxyl group of an alcohol. Specifically, the carbon-1 of one sugar molecule is linked to the carbon-4 of the other. In the alpha configuration, the oxygen atom is located below the plane of the sugar ring.
However, approximately 1 in 10 glucose residues also forms an α-1,6 glycosidic bond with an adjacent glucose, which results in the creation of a branch. Glycogen has only one reducing end and a large number of non-reducing ends with a free hydroxyl group at carbon-4.
The branches increase the solubility of glycogen and make its sugar units accessible. The enzymes involved in the breakdown and synthesis of glycogen are nested between the outer branches of the glycogen molecules and act on the non-reducing ends. Therefore, the many non-reducing end-branches of glycogen facilitate its rapid synthesis and breakdown, making it a readily mobilized source of energy.
Starch, which plays a similar energy-storage role in plants, can also exist in a branched form called amylopectin, though it has a lower degree of branching (about 1 in 30 glucose residues have α-1,6 bonds). In contrast, cellulose, the other major polysaccharide in plants, is an unbranched polymer of glucose, in which β-1,4 linkages form very long, straight chains. This closed structure is suited to the structural role of cellulose, in contrast to the open helices of glycogen and starch, which are nutritional molecules that provide an accessible store of glucose.
Glycogen in liver functions to maintain blood sugar levels
The liver is a major control site of blood glucose levels; it is able to respond to hormonal signals that indicate reduced or elevated amounts of glucose in the blood. The synthesis and breakdown of glycogen in the liver thus serves as a means for maintaining a steady supply of fuel for organs such as the brain, allowing glucose to be stored or released depending on the energy needs of the organism.
As a carbohydrate meal is eaten and digested, blood glucose levels rise, and the pancreas secretes the hormone insulin. Glucose from the portal vein enters the hepatocytes. Insulin acts on these lever cells to stimulate the action of several enzymes, including glycogen synthase, involved in the synthesis of glycogen. Glucose molecules are added to the chains of glycogen as long as both insulin and glucose remain plentiful. In this postprandial or "fed" state, the liver takes in more glucose from the blood than it releases.
Glucagon is another hormone produced by the pancreas, which in many respects serves as a counter-signal to insulin. When blood glucose levels begin to fall (about four hours after a meal), glucagon signals the breakdown of glycogen. Glycogen phosphorylase is the primary enzyme of glycogen breakdown. The freed glucose is then released from the liver into the blood. For the next 8–12 hours (for example, during an overnight fast), glucose derived from liver glycogen will be the primary source of blood glucose to be used by the rest of the body for fuel.
The liver does not use glucose derived from glycogen to meet most of its energy needs, preferring instead the keto acids derived from the breakdown of amino acids. Thus, one might consider the liver an "altruistic" organ, synthesizing and degrading glycogen for the benefit of the organism as a whole.
Glycogen in muscle is an energy reserve for strenuous exercise
Glycogen in muscle cells functions as an immediate reserve source of available glucose during bursts of activity. In resting muscle, however, fatty acids are the preferred fuel. explain
Muscle cells lack the enzyme glucose-6-phosphatase, which enables liver cells to export glucose into the blood. Therefore, the glycogen they store internally is destined for internal use and is not shared with other cells. Other cells that contain small amounts of glycogen use it locally as well.
epinephrine in muscle
Glycogen and marathon running
Due to the human body's ability to hold no more than around 2,000 kcal of glycogen, marathon runners commonly experience a phenomenon referred to as "hitting the wall" around the 20 mile (32 km) point of a marathon. (Approximately 100 kcal are utilized per mile, depending on the size of the runner and the race course.) When experiencing glycogen debt, runners many times experience fatigue.
Cori cycle: shifts some of the burden to liver
Disorders of glycogen metabolism
The most common disease in which glycogen metabolism becomes abnormal is diabetes, in which, because of abnormal amounts of insulin, liver glycogen can be abnormally accumulated or depleted. Restoration of normal glucose metabolism usually normalizes glycogen metabolism as well.
In hypoglycemia caused by excessive insulin, liver glycogen levels are high, but the high insulin level prevents the glycogenolysis necessary to maintain normal blood sugar levels. Glucagon is a common treatment for this type of hypoglycemia.
Various inborn errors of metabolism are caused by deficiencies of enzymes necessary for glycogen synthesis or breakdown. These are collectively referred to as glycogen storage diseases.
ReferencesISBN links support NWE through referral fees
Stryer, Lubert. 1995. Biochemistry, 4th edition. New York, NY: W.H. Freeman.
External links
Template:ChemicalSources
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.