Difference between revisions of "Filtration" - New World Encyclopedia
(ready tag applied) |
(imported latest version of article from Wikipedia) |
||
| Line 1: | Line 1: | ||
{{Ready}} | {{Ready}} | ||
| − | + | {{otheruses4|operation of solid-fluid separation|the mathematical concept|filtration (mathematics)|the equipment used|filter}} | |
| − | |||
[[Image:Filtration diagram.svg|right|thumb|300px|diagram of a simple filtration]] | [[Image:Filtration diagram.svg|right|thumb|300px|diagram of a simple filtration]] | ||
| − | '''Filtration''' is the | + | '''Filtration''' is a mechanical or physical operation which is used for the separation of solids from fluids (liquids or gases) by interposing a medium to fluid flow through which the fluid can pass, but the solids (or at least part of the solids) in the fluid are retained. It has to be emphasized that the separation is NOT complete, and it will depend on the pore size and the thickness of the medium as well as the mechanisms that occur during filtration. |
| + | |||
| + | * Filtration is used for the purification of fluids: for instance separating dust from the atmosphere to clean ambient air. | ||
| + | |||
| + | * Filtration, as a physical operation is very important in chemistry for the separation of materials of different chemical composition in solution (or solids which can be dissolved) by first using a reagent to precipitate one of the materials and then use a filter to separate the solid from the other material(s). | ||
| + | |||
| + | * Filtration is also important and widely used as one of the unit operations of chemical engineering. | ||
| + | |||
| + | It is important not to confuse '''filtration''' with '''sieving'''. In sieving there is only a single layer of medium where size separation occurs purely by the fact that the fraction of the particulate solid matter which is too large to be able to pass through the holes of the sieve, scientifically called ''oversize'' (See [[particle size distribution]]) are retained. In filtration a multilayer medium is involved, where other mechanisms are included as well, for instance direct interception, diffusion and centrifugal action, where in this latter those particles, which are unable to follow the tortuous channels of the filter will also adhere to the structure of the medium and are retained.<ref>Lecture notes, Postgraduate course on Filtration and Size separation at the Department of Chemical Engineering, University of Lougborough, England</ref> | ||
| + | |||
| + | Depending on the application, either one or both of the components may be isolated. Examples of filtration include A) a [[coffee filter]] to keep the coffee separate from the grounds and B) the use of [[HEPA]] filters in [[air conditioning]] to remove particles from air. | ||
| + | |||
| + | The filtration process separates particles and fluid from a suspension, and the fluid can be either a [[liquid]] or a [[gas]] (or a [[supercritical fluid]]). To separate a mixture of chemical compounds, a [[solvent]] is chosen which dissolves one component, while not dissolving the other. By dissolving the mixture in the chosen solvent, one component will go into the solution and pass through the filter, while the other will be retained. This is one of the most important techniques used by chemists to purify compounds. | ||
| + | |||
| + | Filtration also cleans up water streams or other river streams. Furnaces use filtration to prevent the furnace elements from fouling with particulates. Pneumatic conveying systems often employ filtration to stop or slow the flow of material that is transported, through the use of a [[dust collector|baghouse]]. | ||
| − | + | The remainder of this article focuses primarily on liquid filtration. | |
==Methods== | ==Methods== | ||
| − | There are many different methods of filtration; all aim to attain the [[separation of mixtures|separation]] of | + | There are many different methods of filtration; all aim to attain the [[separation of mixtures|separation]] of 4,000,000 or more substances. This is achieved by some form of interaction between the substance or objects to be removed and the filter. In addition the substance that is to pass through the filter must be a [[fluid]], i.e. a [[liquid]] or [[gas]]. |
The simplest method of filtration is to pass a solution of a solid and fluid through a porous interface so that the solid is trapped, while the fluid passes through. This principle relies upon the size difference between the particles making up the fluid, and the particles making up the solid. In the laboratory, a [[Büchner funnel]] is often used, with a [[filter paper]] serving as the porous barrier. | The simplest method of filtration is to pass a solution of a solid and fluid through a porous interface so that the solid is trapped, while the fluid passes through. This principle relies upon the size difference between the particles making up the fluid, and the particles making up the solid. In the laboratory, a [[Büchner funnel]] is often used, with a [[filter paper]] serving as the porous barrier. | ||
| − | For example an experiment to prove the existence of [[Microorganism|microscopic organisms]] involves the comparison of water passed through unglazed [[porcelain]] and unfiltered | + | For example an experiment to prove the existence of [[Microorganism|microscopic organisms]] involves the comparison of water passed through unglazed [[porcelain]] and unfiltered water. When left in sealed containers the filtered water takes longer to go foul, showing that very small items (such as [[bacterium|bacteria]]) can be removed from fluids by filtration.{{Fact|date=March 2007}} Alternate methods often take the form of [[electrostatic]] attractions. These form of filters again have the problem of either becoming clogged, or the active sites on the filter all become used by the undesirable. However, most chemical filters are designed so that the filter can be flushed with a chemical that will remove the undesirables and allow the filter to be re-used. |
| − | == | + | ==Flowing== |
| − | Liquids usually flow through the filter by gravity. This is the simplest method, and can be seen in the coffeemaker example. For chemical plants, this is usually the most economical method as well. In the laboratory, pressure in the form of compressed air may be applied to | + | Liquids usually flow through the filter by gravity. This is the simplest method, and can be seen in the coffeemaker example. For chemical plants, this is usually the most economical method as well. In the laboratory, pressure in the form of compressed air may be applied to make the filtration process faster, though this may lead to clogging or the passage of fine particles. Alternatively, the liquid may flow through the filter by the force exerted by a pump. In this case, the filter need not be mounted vertically. |
==Filter media== | ==Filter media== | ||
| − | There are two main types of filter media | + | There are two main types of filter media — a solid sieve which traps the solid particles, with or without the aid of filter paper, and a bed of granular material which retains the solid particles as it passes. The first type allows the solid particles, i.e. the residue, to be collected intact; the second type does not permit this. However, the second type is less prone to clogging due to the greater surface area where the particles can be trapped. Also, when the solid particles are very fine, it is often cheaper and easier to discard the contaminated granules than to clean the solid sieve. |
Filter media can be cleaned by rinsing with solvents or detergents. Alternatively, in engineering applications, such as swimming pool water treatment plants, they may be cleaned by [[backwashing]]. | Filter media can be cleaned by rinsing with solvents or detergents. Alternatively, in engineering applications, such as swimming pool water treatment plants, they may be cleaned by [[backwashing]]. | ||
| − | Examples of the first type include filter paper used with a Buchner, Hirsch, filter funnel or other similar funnel. A sintered-glass funnel is often used in chemistry laboratories because it is able to trap very fine particles, while permitting the particles to be removed by a spatula. | + | Examples of the first type include '''filter paper''' used with a Buchner, Hirsch, filter funnel or other similar funnel. A '''sintered-glass funnel''' is often used in chemistry laboratories because it is able to trap very fine particles, while permitting the particles to be removed by a spatula. |
Examples of the second type include filters at municipal and swimming pool water treatment plants, where the granular material is sand. In the laboratory, [[Celite]] or [[diatomaceous earth]] is packed in a [[Pasteur pipette]] (microscale) or loaded on top of a sintered-glass funnel to serve as the filter bed. | Examples of the second type include filters at municipal and swimming pool water treatment plants, where the granular material is sand. In the laboratory, [[Celite]] or [[diatomaceous earth]] is packed in a [[Pasteur pipette]] (microscale) or loaded on top of a sintered-glass funnel to serve as the filter bed. | ||
| − | |||
| − | Certain filter aids may be used to aid filtration. These are often incompressible diatomaceous earth or | + | The following points should be considerd while selecting the filter media: |
| + | * ability to build the solid. | ||
| + | * minimum resistance to flow the filtrate. | ||
| + | * resistance to chemical attack. | ||
| + | * minimum cost. | ||
| + | * long life. | ||
| + | |||
| + | ==Filter aid== | ||
| + | |||
| + | Certain filter aids may be used to aid filtration. These are often incompressible diatomaceous earth or kieselguhr, which is composed primarily of [[silica]]. Also used are wood cellulose and other inert porous solids. | ||
| − | These filter aids can be used in | + | These filter aids can be used in two different ways. They can be used as a precoat before the [[slurry]] is filtered. This will prevent gelatinous-type solids from plugging the filter medium and also give a clearer filtrate. They can also be added to the [[slurry]] before filtration. This increases the [[porosity]] of the cake and reduces resistance of the cake during filtration. In a rotary filter, the filter aid may be applied as a precoat; subsequently, thin slices of this layer are sliced off with the cake. |
The use of filter aids is usually limited to cases where the cake is discarded or where the [[precipitate]] can be separated chemically from the filter. | The use of filter aids is usually limited to cases where the cake is discarded or where the [[precipitate]] can be separated chemically from the filter. | ||
| Line 38: | Line 59: | ||
Filtration is a more efficient method for the [[separation of mixtures]] than [[decantation]], but is much more time consuming. If very small amounts of [[solution]] are involved, most of the solution may be soaked up by the filter medium. | Filtration is a more efficient method for the [[separation of mixtures]] than [[decantation]], but is much more time consuming. If very small amounts of [[solution]] are involved, most of the solution may be soaked up by the filter medium. | ||
| − | An alternative to filtration is [[centrifugation]] | + | An alternative to filtration is [[centrifugation]] — instead of filtering the mixture of solid and liquid particles, the mixture is centrifuged to force the (usually) denser solid to the bottom, where it often forms a firm cake. The liquid above can then be decanted. This method is especially useful for separating solids which do not filter well, such as gelatinous or fine particles. These solids can clog or pass through the filter, respectively. |
==Filter types== | ==Filter types== | ||
| Line 45: | Line 66: | ||
*Side stream filter (filter in a closed loop, that filters part of the media per cycle only) | *Side stream filter (filter in a closed loop, that filters part of the media per cycle only) | ||
*Continuous rotary filters | *Continuous rotary filters | ||
| + | |||
| + | ==In kidney== | ||
| + | {{Main|Renal filtration}} | ||
| + | The [[kidney]] works by filtration of [[blood]] in the [[glomerulus]], followed by selectively reabsorbing many substances essential for the body. | ||
==See also== | ==See also== | ||
| − | + | {{wiktionary|filtration}} | |
| + | * [[:Category:Filters]] | ||
* [[Separation of mixtures]] | * [[Separation of mixtures]] | ||
| − | * [[Microfiltration]] | + | * [[Microfiltration]] |
| + | * [[Ultrafiltration]] | ||
| + | * [[Nanofiltration]] | ||
* [[Reverse osmosis]] | * [[Reverse osmosis]] | ||
* [[Filter (water)]] | * [[Filter (water)]] | ||
| + | |||
| + | ==Footnotes== | ||
| + | {{Reflist}} | ||
== References == | == References == | ||
| − | + | [http://www.airfilterengineering.com Air filter Engineering (M) Sdn. Bhd.] | |
| − | |||
| − | |||
| − | [http://www.airfilterengineering.com | ||
| − | |||
| + | [[Category:Analytical chemistry]] | ||
| + | [[Category:Laboratory techniques]] | ||
| + | [[Category:Separation processes]] | ||
| + | [[Category:Unit operations]] | ||
| + | [[Category:Alchemical processes]] | ||
| − | [[ | + | ==Further reading== |
| + | *[http://www.filtsep.com www.filtsep.com] | ||
| + | *[http://www.filtration-and-separation.com filtration equipment expert system and data analysis] | ||
| − | + | [[cs:Filtrace]] | |
| + | [[de:Filtration]] | ||
| + | [[el:Διήθηση]] | ||
| + | [[es:Filtración]] | ||
| + | [[fr:Filtration]] | ||
| + | [[ko:여과]] | ||
| + | [[it:Filtrazione (chimica)]] | ||
| + | [[he:סינון]] | ||
| + | [[nl:Filtratie]] | ||
| + | [[ja:ろ過]] | ||
| + | [[pl:Filtracja]] | ||
| + | [[ru:Фильтрование]] | ||
| + | [[sr:Филтрација]] | ||
| + | [[zh:过滤]] | ||
Revision as of 21:30, 7 May 2008
- This article is about operation of solid-fluid separation. For the mathematical concept, see filtration (mathematics).
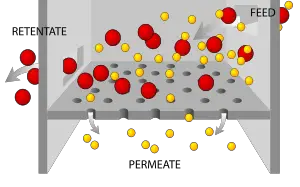
Filtration is a mechanical or physical operation which is used for the separation of solids from fluids (liquids or gases) by interposing a medium to fluid flow through which the fluid can pass, but the solids (or at least part of the solids) in the fluid are retained. It has to be emphasized that the separation is NOT complete, and it will depend on the pore size and the thickness of the medium as well as the mechanisms that occur during filtration.
- Filtration is used for the purification of fluids: for instance separating dust from the atmosphere to clean ambient air.
- Filtration, as a physical operation is very important in chemistry for the separation of materials of different chemical composition in solution (or solids which can be dissolved) by first using a reagent to precipitate one of the materials and then use a filter to separate the solid from the other material(s).
- Filtration is also important and widely used as one of the unit operations of chemical engineering.
It is important not to confuse filtration with sieving. In sieving there is only a single layer of medium where size separation occurs purely by the fact that the fraction of the particulate solid matter which is too large to be able to pass through the holes of the sieve, scientifically called oversize (See particle size distribution) are retained. In filtration a multilayer medium is involved, where other mechanisms are included as well, for instance direct interception, diffusion and centrifugal action, where in this latter those particles, which are unable to follow the tortuous channels of the filter will also adhere to the structure of the medium and are retained.[1]
Depending on the application, either one or both of the components may be isolated. Examples of filtration include A) a coffee filter to keep the coffee separate from the grounds and B) the use of HEPA filters in air conditioning to remove particles from air.
The filtration process separates particles and fluid from a suspension, and the fluid can be either a liquid or a gas (or a supercritical fluid). To separate a mixture of chemical compounds, a solvent is chosen which dissolves one component, while not dissolving the other. By dissolving the mixture in the chosen solvent, one component will go into the solution and pass through the filter, while the other will be retained. This is one of the most important techniques used by chemists to purify compounds.
Filtration also cleans up water streams or other river streams. Furnaces use filtration to prevent the furnace elements from fouling with particulates. Pneumatic conveying systems often employ filtration to stop or slow the flow of material that is transported, through the use of a baghouse.
The remainder of this article focuses primarily on liquid filtration.
Methods
There are many different methods of filtration; all aim to attain the separation of 4,000,000 or more substances. This is achieved by some form of interaction between the substance or objects to be removed and the filter. In addition the substance that is to pass through the filter must be a fluid, i.e. a liquid or gas.
The simplest method of filtration is to pass a solution of a solid and fluid through a porous interface so that the solid is trapped, while the fluid passes through. This principle relies upon the size difference between the particles making up the fluid, and the particles making up the solid. In the laboratory, a Büchner funnel is often used, with a filter paper serving as the porous barrier.
For example an experiment to prove the existence of microscopic organisms involves the comparison of water passed through unglazed porcelain and unfiltered water. When left in sealed containers the filtered water takes longer to go foul, showing that very small items (such as bacteria) can be removed from fluids by filtration.[citation needed] Alternate methods often take the form of electrostatic attractions. These form of filters again have the problem of either becoming clogged, or the active sites on the filter all become used by the undesirable. However, most chemical filters are designed so that the filter can be flushed with a chemical that will remove the undesirables and allow the filter to be re-used.
Flowing
Liquids usually flow through the filter by gravity. This is the simplest method, and can be seen in the coffeemaker example. For chemical plants, this is usually the most economical method as well. In the laboratory, pressure in the form of compressed air may be applied to make the filtration process faster, though this may lead to clogging or the passage of fine particles. Alternatively, the liquid may flow through the filter by the force exerted by a pump. In this case, the filter need not be mounted vertically.
Filter media
There are two main types of filter media — a solid sieve which traps the solid particles, with or without the aid of filter paper, and a bed of granular material which retains the solid particles as it passes. The first type allows the solid particles, i.e. the residue, to be collected intact; the second type does not permit this. However, the second type is less prone to clogging due to the greater surface area where the particles can be trapped. Also, when the solid particles are very fine, it is often cheaper and easier to discard the contaminated granules than to clean the solid sieve.
Filter media can be cleaned by rinsing with solvents or detergents. Alternatively, in engineering applications, such as swimming pool water treatment plants, they may be cleaned by backwashing.
Examples of the first type include filter paper used with a Buchner, Hirsch, filter funnel or other similar funnel. A sintered-glass funnel is often used in chemistry laboratories because it is able to trap very fine particles, while permitting the particles to be removed by a spatula.
Examples of the second type include filters at municipal and swimming pool water treatment plants, where the granular material is sand. In the laboratory, Celite or diatomaceous earth is packed in a Pasteur pipette (microscale) or loaded on top of a sintered-glass funnel to serve as the filter bed.
The following points should be considerd while selecting the filter media:
- ability to build the solid.
- minimum resistance to flow the filtrate.
- resistance to chemical attack.
- minimum cost.
- long life.
Filter aid
Certain filter aids may be used to aid filtration. These are often incompressible diatomaceous earth or kieselguhr, which is composed primarily of silica. Also used are wood cellulose and other inert porous solids.
These filter aids can be used in two different ways. They can be used as a precoat before the slurry is filtered. This will prevent gelatinous-type solids from plugging the filter medium and also give a clearer filtrate. They can also be added to the slurry before filtration. This increases the porosity of the cake and reduces resistance of the cake during filtration. In a rotary filter, the filter aid may be applied as a precoat; subsequently, thin slices of this layer are sliced off with the cake.
The use of filter aids is usually limited to cases where the cake is discarded or where the precipitate can be separated chemically from the filter.
Alternatives
| |||||||||||||
Filtration is a more efficient method for the separation of mixtures than decantation, but is much more time consuming. If very small amounts of solution are involved, most of the solution may be soaked up by the filter medium.
An alternative to filtration is centrifugation — instead of filtering the mixture of solid and liquid particles, the mixture is centrifuged to force the (usually) denser solid to the bottom, where it often forms a firm cake. The liquid above can then be decanted. This method is especially useful for separating solids which do not filter well, such as gelatinous or fine particles. These solids can clog or pass through the filter, respectively.
Filter types
- Gravity filter (open system that operates with water column pressure only)
- Pressure filter (closed system that operates under pressure from a pump)
- Side stream filter (filter in a closed loop, that filters part of the media per cycle only)
- Continuous rotary filters
In kidney
The kidney works by filtration of blood in the glomerulus, followed by selectively reabsorbing many substances essential for the body.
See also
- Category:Filters
- Separation of mixtures
- Microfiltration
- Ultrafiltration
- Nanofiltration
- Reverse osmosis
- Filter (water)
Footnotes
- ↑ Lecture notes, Postgraduate course on Filtration and Size separation at the Department of Chemical Engineering, University of Lougborough, England
ReferencesISBN links support NWE through referral fees
Air filter Engineering (M) Sdn. Bhd.
Further reading
cs:Filtrace de:Filtration el:Διήθηση es:Filtración fr:Filtration ko:여과 it:Filtrazione (chimica) he:סינון nl:Filtratie ja:ろ過 pl:Filtracja ru:Фильтрование sr:Филтрација zh:过滤