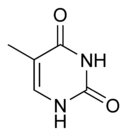
Thymine
| Thymine | |
|---|---|
| Chemical name | 5-Methylpyrimidine-2,4(1H,3H)-dione |
| Chemical formula | C5H6N2O2 |
| Molecular mass | 126.11334 g/mol |
| Melting point | 316 - 317 °C |
| CAS number | 65-71-4 |
| SMILES | CC1=CNC(NC1=O)=O |
| |
Thymine, also known as 5-methyluracil, is one of the five main nucleobases found in the nucleic acids DNA and RNA. The others are adenine, cytosine, guanine, and uracil. Thymine, cytosine, and uracil are pyrimidine derivatives, and guanine and adenine are purine derivatives.
In DNA, thymine and cytosine form hydrogen bonds with their complementary purine derivatives, adenine and cytosine, respectively. In RNA, the complement of adenine usually is uracil instead of thymine. Thus, thymine is usually seen only in DNA and uracil only in RNA.
The fact that thymine is found in all cells, eukaryotic and prokaryotic, and that thymine, as part of DNA, is central to the flow of genetic information (excepting in RNA viruses) shows a connectedness among all life.
Properties
As a pyrimidine nucleobase, thymine is a heterocyclic aromatic organic compound. Heterocyclic compounds are organic compounds (those containing carbon) that contain a ring structure containing atoms in addition to carbon, such as sulfur, oxygen, or nitrogen, as part of the ring. Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone.
As the name implies, thymine may be derived by methylation of uracil at the fifth carbon. In DNA, thymine(T) binds to adenine (A) via two hydrogen bonds to assist in stabilizing the nucleic acid structures.
Thymine combined with deoxyribose creates the nucleoside deoxythymidine, which is synonymous with the term thymidine. Thymidine can be phosphorylated with one, two, or three phosphoric acid groups, creating TMP, TDP or TTP (thymidine mono- di- or triphosphate) respectively.
One of the common mutations of DNA involves two adjacent thymines or cytosine, which in presence of ultraviolet light may form thymine dimers, causing "kinks" in the DNA molecule that inhibit normal function.
Thymine could also be a target for actions of 5-fu in cancer treatment. 5-fu can be a metabolic analog of Thymine (in DNA synthesis) or Uracil (in RNA synthesis). Substitution of this analog inhibit DNA synthesis in actively dividing cells.
ReferencesISBN links support NWE through referral fees
- Kakkar, R., and R. Garg. “Theoretical study of the effect of radiation on thymine.” Journal of Molecular Structure-TheoChem 620(2-3): 139-147, 2003.
- Al Mahroos, M., et al. “Effect of sunscreen application on UV-induced thymine dimers.” Arch Dermatol 138: 1480-5, 2002.
- Reynisson, J., and S. Steenken. “DFT studies on the pairing abilities of the one-electron reduced or oxidized adenine-thymine base pair.” Physical Chemistry Chemical Physics 4(21): 5353-5358, 2002.
| Nucleic acids edit |
|---|
| Nucleobases: Adenine - Thymine - Uracil - Guanine - Cytosine - Purine - Pyrimidine |
| Nucleosides: Adenosine - Uridine - Guanosine - Cytidine - Deoxyadenosine - Thymidine - Deoxyguanosine - Deoxycytidine |
| Nucleotides: AMP - UMP - GMP - CMP - ADP - UDP - GDP - CDP - ATP - UTP - GTP - CTP - cAMP - cGMP |
| Deoxynucleotides: dAMP - dTMP - dUMP - dGMP - dCMP - dADP - dTDP - dUDP - dGDP - dCDP - dATP - dTTP - dUTP - dGTP - dCTP |
| Nucleic acids: DNA - RNA - LNA - PNA - mRNA - ncRNA - miRNA - rRNA - siRNA - tRNA - mtDNA - Oligonucleotide |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.