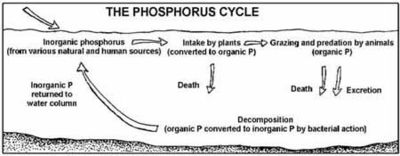
Phosphorus cycle
The phosphorus cycle is the biogeochemical cycle that describes the movement of phosphorus through the geosphere, hydrosphere, and biosphere. Unlike the other major biogeochemical cycles (oxygen, carbon, nitrogen, and water), the atmosphere does not play a significant role in the movements of phosphorus because phosphorus and phosphorus-based compounds are usually solids at the typical ranges of temperature and pressure found on Earth.
The phosphorus cycle reflects the harmonious interactions between organisms and their biotic and abiotic environments, with phosphorus flowing through each compartment by give and receive actions and allowing life to exist. However, sometimes the harmony that has been built up over many years is disrupted by human beings, who may put excessive amounts of phorphorus into a particular ecosystem through fertilizer, sewage, or other means. This can result in eutrophication and decreased biodiversity.
Overview
A biogeochemical cycle is a circuit or pathway by which a chemical element or molecule moves through both biotic ("bio-") and abiotic ("geo-") compartments of an ecosystem. The most well-known and important biogeochemical cycles include the carbon cycle, the nitrogen cycle, the oxygen cycle, the phosphorus cycle, and the water cycle.
All the chemicals, nutrients, or elements used in ecosystems by living organisms—such as carbon, nitrogen, oxygen, and phosphorus—operate on a closed system, which means that these chemicals are recycled as opposed to an open system in which the chemicals are lost. The energy of an ecosystem occurs on an open system; the sun constantly gives the planet energy in the form of light, which is eventually used and lost in the form of heat.
Although components of the biogeochemical cycle are not lost as in the energy system, they can be held for long periods of time in one place. In effect, the element is recycled, although there may be places (called "sinks") where the element is accumulated or held. When they are held for a long period of time, the place is called a reservoir. When chemicals are held for only short periods of time, they are being held in exchange pools. Generally, reservoirs are abiotic factors while exchange pools are biotic factors.
Phosphorus is an essential element for living organisms, being part of nucleic acids (DNA and RNA), ATP (the "molecular currency" of intracellular energy transfer), and diverse coenzymes, and serving as the building blocks of animal bones and teeth, among numerous other necessary roles. Photosynthesis, metabolism, energy transport, nerve function, and muscle movement are all dependent on phosphates.
Phosphorus in the Environment
Phosphorus normally occurs in nature as part of a phosphate ion, consisting of a phosphorus atom and some number of oxygen atoms, the most abundant form (called orthophosphate) having four oxygens: PO43-. Most phosphates are found as salts in ocean sediments or in rocks.
Over time, geologic processes can bring ocean sediments to land, and weathering will carry terrestrial phosphates back to the ocean. Plants absorb phosphates from the soil. The plants may then be consumed by herbivores who in turn may be consumed by carnivores. After death, the animal or plant decays, and the phosphates are returned to the soil. Runoff may carry them back to the ocean or they may be reincorporated into rock.
The primary biological importance of phosphates is as a component of nucleotides, which serve as energy storage within cells (ATP) or when linked together, form the nucleic acids DNA and RNA. Phosphorus is also found in bones, whose strength is derived from calcium phosphate, and in phospholipids (found in all biological membranes).
Phosphates move quickly through plants and animals; however, the processes that move them through the soil or ocean are very slow, making the phosphorus cycle overall one of the slowest biogeochemical cycles.
Human Influence
Artificial fertilizers and other wastes not absorbed by plants mostly enter the groundwater and collect in streams, lakes, and ponds. The extra phosphates are a major contributor to the process called eutrophication. Eutrophication is the enrichment of an aquatic ecosystem with chemical nutrients, typically compounds containing nitrogen, phosphorus, or both. The increase in available nutrients promotes plant growth, favoring certain species over others, and forcing a change in species composition. In aquatic environments, enhanced growth of choking aquatic vegetation or phytoplankton (that is, an algal bloom) disrupts normal functioning of the ecosystem, causing a variety of problems, including depletion of dissolved oxygen, potentially suffocating fish and other aquatic fauna.
Nutrients from human activities tend to accumulate in soils and remain there for years. It has been shown that the amount of phosphorus lost to surface waters increases linearly with the amount of phosphorus in the soil (Sharpley et al., 1996). Thus, much nutrient loading in soil eventually makes its way to water. Furthermore, phosphorus has the capacity to be released from the soil after a lag time of ten years.
ReferencesISBN links support NWE through referral fees
- Dévai, I., Felföldy, L., Wittner, I., and Plósz, S. “Detection of Phosphine: New Aspects of the Phosphorus Cycle in the Hydrosphere.” Nature. 333: 343-345. 1988.
- Sharpley A.N., Daniel, T.C., Sims, J.T., and Pote, D.H. “Determining Environmentally Sound Soil Phosphorus Levels.” Journal of Soil and Water Conservation. 51:160-166. 1996.
- United States Environmental Protection Agency (EPA). Monitoring and Assessing Water Quality: Section 5.6 Phosphorus. Retrieved August 27, 2007.
| Biogeochemical cycles |
|---|
| Carbon cycle - Hydrogen cycle - Nitrogen cycle |
| Oxygen cycle - Phosphorus cycle - Sulfur cycle - Water cycle |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.