Chloroethane
| Chloroethane | |
|---|---|
| |
| |
| IUPAC name | Chloroethane |
| Other names | Ethyl chloride Monochloroethane Chlorene Muriatic ether EtCl UN 1037 |
| Identifiers | |
| CAS number | [] |
| PubChem | |
| RTECS number | KH7525000 |
| SMILES | CCCl |
| Properties | |
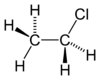
| Molecular formula | C2H5Cl |
| Molar mass | 64.51 g mol−1 |
| Appearance | colourless gas |
| Density | 0.92 g cm−3, liquid |
| Melting point |
−139°C (134 K) |
| Boiling point |
12.3°C (285.4 K) |
| Solubility in water | 0.6 g/100 ml (?°C) |
| Viscosity | ? cP at ?°C |
| Structure | |
| Dipole moment | 2.06 D |
| Hazards | |
| Main hazards | Flammable |
| NFPA 704 |
|
| R-phrases | R12, R40, Template:R52, Template:R53 |
| S-phrases | S9, S16, S33, S36, S37, S61 |
| Flash point | −50°C (closed cup) |
| Related Compounds | |
| Related haloalkanes | 1,1-dichloroethane 1,2-dichloroethane |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Chloroethane or monochloroethane, commonly known by its old name ethyl chloride, is a chemical compound once widely used in producing tetra-ethyl lead, a gasoline additive. It is a colorless, flammable gas or refrigerated liquid with a faintly sweet odor.
Production
Ethyl chloride is produced by reacting ethylene and hydrogen chloride over an aluminium chloride catalyst at temperatures ranging from 130-250°C. Under these conditions, ethyl chloride is produced according to the chemical equation.
At various times in the past, ethyl chloride has also been produced from ethanol and hydrochloric acid, or from ethane and chlorine, but these routes are no longer economical. Some ethyl chloride is generated as a byproduct of polyvinyl chloride production. Should demand for ethyl chloride continue to fall to the point where making it for its own sake is not economical, this may become the leading source of the chemical.
Uses
Beginning in 1922 and continuing through most of the twentieth century, the major use of ethyl chloride was to produce tetraethyl lead (TEL), an anti-knock additive for gasoline. However, due to growing awareness of air pollution, TEL has been or is being phased out in most of the industrialized world, and the demand for ethyl chloride has fallen sharply.
Like other chlorinated hydrocarbons, ethyl chloride has been used as a refrigerant, an aerosol spray propellant, an anesthetic, and a blowing agent for foam packaging. For a time it was used as a promoter chemical in the aluminum chloride catalyzed process to produce ethylbenzene, the precursor for styrene monomer. At present though, it is not widely used in any of these roles.
The only remaining industrially important use of ethyl chloride is in treating cellulose to make ethylcellulose, a thickening agent and binder in paints, cosmetics, and similar products.
Ethyl chloride is a prescription drug in the US, supplied as a liquid in a spray bottle propelled by its own vapor pressure. It acts as a mild topical anesthetic by its chilling effect when sprayed on skin, such as when removing splinters in a clinical setting. The heat absorbed by the boiling liquid on tissues produces a deep and rapid chill, but since the boiling point is well above the freezing point of water, it presents no danger of frostbite. The vapor is flammable and narcotic, which requires care.
Ethyl chloride is a narcotic inhalant drug, sometimes referred to as "Duster." Similar to poppers, ethyl chloride is used as an inhalant (huffed) during sexual activity for an intense several-minute-long high that results in a prolonged orgasm. In Brazil, it is a traditional (though illegal) drug taken during Carnaval parades, known as "lança-perfume."
Safety
Ethyl chloride is the least toxic of the chloroethanes. Like other chlorinated hydrocarbons, it is a central nervous system depressant, albeit a less potent one than many similar compounds. People breathing its vapors at less than 1 percent concentration in air usually experience no symptoms. At higher concentrations, victims usually exhibit symptoms similar to those of alcohol intoxication. Breathing its vapors at 15 percent or higher is often fatal.
Studies on the effects of chronic ethyl chloride exposure in animals have given inconsistent results, and there exist no data for its long-term effects on humans. Some studies have reported that prolonged exposure can produce liver or kidney damage, or uterine cancer in mice, but these data have been difficult to reproduce.
Recent information suggests carcinogenic potential; it has been designated as IARC category A3, Confirmed Animal Carcinogen with Unknown Relevance to Humans. As a result, the State of California has incorporated it into Proposition 65 as a known carcinogen. Nonetheless, it is still used in medicine as a local anesthetic.
See also
ReferencesISBN links support NWE through referral fees
- McMurry, John. 2004. Organic Chemistry, 6th ed. Belmont, CA: Brooks/Cole. ISBN 0534420052.
- Solomons, T.W. Graham, and Craig B. Fryhle. 2004. Organic Chemistry, 8th ed. Hoboken, NJ: John Wiley. ISBN 0471417998.
- Zumdahl, Steven S. 2005. Chemical Principles. New York, NY: Houghton Mifflin. ISBN 0618372067.
External links
All links retrieved December 10, 2023.
- NIOSH Pocket Guide to Chemical Hazards: Ethyl chloride. CDC, National Institute for Occupational Safety and Health.
- Ethyl chloride. NIST Chemistry WebBook.
- Chloroethane (ethyl chloride) fact sheet. National Pollutant Inventory.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
