Difference between revisions of "Photochemistry" - New World Encyclopedia
(added credit and category tags, deleted foreign language links) |
|||
| Line 1: | Line 1: | ||
| − | [[Image:Spectre.svg|thumb|350px| | + | {{Claimed}} |
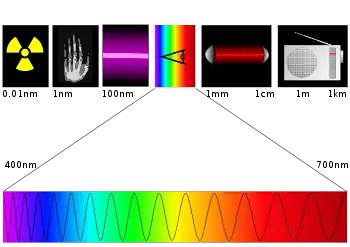
| + | [[Image:Spectre.svg|thumb|350px|Significant regions of the electromagnetic spectrum, with their approximate wavelengths. The bottom part of the illustration shows an expanded version of the visible spectrum.]] | ||
| − | + | '''Photochemistry''', a sub-discipline of [[chemistry]], is the study of the interactions between [[atom]]s, small [[molecules]], and light (or [[electromagnetic radiation]]).<ref>{{GoldBookRef|title=photochemistry|file=P04588}}</ref> The chemical reactions that take place through these interactions are known as '''photochemical reactions'''. The main pillars of photochemistry are photochemical reactions in organic chemistry, UV/VIS (ultraviolet/visible) spectroscopy, and [[photosynthesis]] in biochemistry. | |
| − | + | == Reactions activated by light == | |
| − | + | A photochemical reaction may be thought of as a reaction that proceeds with the absorption of light. Normally, a reaction (not just a photochemical reaction) occurs when the molecules involved gain the [[activation energy]] necessary to undergo change. For example, for the [[combustion]] of [[gasoline]] (a [[hydrocarbon]]) to produce carbon dioxide and water, activation energy is supplied in the form of heat or a spark. In the case of photochemical reactions, light provides the activation energy. | |
| − | + | The absorption of a photon of light by a reactant molecule may permit a reaction to occur not just by bringing the molecule to the necessary activation energy, but also by changing the symmetry of the molecule's electronic configuration, enabling an otherwise inaccessible reaction path. | |
| − | + | == Laws of photochemistry == | |
| − | + | The first law of photochemistry, known as the [[Grotthuss-Draper law]] (for chemists [[Theodor Grotthuss]] and [[John W. Draper]]), states that light must be absorbed by a chemical substance in order for a [[photochemical reaction]] to take place. | |
| − | The | + | |
| + | The second law of photochemistry, the [[Stark-Einstein law]], states that for each photon of light absorbed by a chemical system, only one molecule is activated for a photochemical reaction. This is also known as the [[photoequivalence law]] and was derived by [[Albert Einstein]] at the time when the [[quantum mechanics|quantum (photon) theory]] of light was being developed. | ||
==Regions of the electromagnetic spectrum== | ==Regions of the electromagnetic spectrum== | ||
| − | The [[electromagnetic spectrum]] is broad, | + | |
| − | * Visible Light: 400–700 nm wavelengths | + | The [[electromagnetic spectrum]] is broad, but photochemists find themselves working with several key regions, noted here: |
| + | * Visible Light: 400–700 nanometer (nm) wavelengths | ||
* Ultraviolet: 100–400 nm wavelengths | * Ultraviolet: 100–400 nm wavelengths | ||
* Near Infrared: 700–1000 nm wavelengths | * Near Infrared: 700–1000 nm wavelengths | ||
* Far infrared: 15–1000 µm wavelengths | * Far infrared: 15–1000 µm wavelengths | ||
| + | |||
| + | == Units and constants == | ||
| + | |||
| + | Like most scientific disciplines, photochemistry utilizes the SI or metric measurement system. Important units and constants that show up regularly include the [[meter]] (and variants such as centimeter, millimeter, micrometer, and nanometer), seconds, hertz, joules, moles, the [[gas constant]] ''R'', and the [[Boltzmann constant]]. These units and constants are also integral to the field of [[physical chemistry]]. | ||
==See also== | ==See also== | ||
| − | *[[ | + | |
| − | *[[ | + | * [[Chemical reaction]] |
| + | * [[Electromagnetic spectrum]] | ||
| + | * [[Light]] | ||
==References == | ==References == | ||
| Line 33: | Line 42: | ||
[[Category:Chemistry]] | [[Category:Chemistry]] | ||
| − | {{ | + | {{credits|Photochemistry|146973909}} |
Revision as of 23:51, 20 August 2007
Photochemistry, a sub-discipline of chemistry, is the study of the interactions between atoms, small molecules, and light (or electromagnetic radiation).[1] The chemical reactions that take place through these interactions are known as photochemical reactions. The main pillars of photochemistry are photochemical reactions in organic chemistry, UV/VIS (ultraviolet/visible) spectroscopy, and photosynthesis in biochemistry.
Reactions activated by light
A photochemical reaction may be thought of as a reaction that proceeds with the absorption of light. Normally, a reaction (not just a photochemical reaction) occurs when the molecules involved gain the activation energy necessary to undergo change. For example, for the combustion of gasoline (a hydrocarbon) to produce carbon dioxide and water, activation energy is supplied in the form of heat or a spark. In the case of photochemical reactions, light provides the activation energy.
The absorption of a photon of light by a reactant molecule may permit a reaction to occur not just by bringing the molecule to the necessary activation energy, but also by changing the symmetry of the molecule's electronic configuration, enabling an otherwise inaccessible reaction path.
Laws of photochemistry
The first law of photochemistry, known as the Grotthuss-Draper law (for chemists Theodor Grotthuss and John W. Draper), states that light must be absorbed by a chemical substance in order for a photochemical reaction to take place.
The second law of photochemistry, the Stark-Einstein law, states that for each photon of light absorbed by a chemical system, only one molecule is activated for a photochemical reaction. This is also known as the photoequivalence law and was derived by Albert Einstein at the time when the quantum (photon) theory of light was being developed.
Regions of the electromagnetic spectrum
The electromagnetic spectrum is broad, but photochemists find themselves working with several key regions, noted here:
- Visible Light: 400–700 nanometer (nm) wavelengths
- Ultraviolet: 100–400 nm wavelengths
- Near Infrared: 700–1000 nm wavelengths
- Far infrared: 15–1000 µm wavelengths
Units and constants
Like most scientific disciplines, photochemistry utilizes the SI or metric measurement system. Important units and constants that show up regularly include the meter (and variants such as centimeter, millimeter, micrometer, and nanometer), seconds, hertz, joules, moles, the gas constant R, and the Boltzmann constant. These units and constants are also integral to the field of physical chemistry.
See also
ReferencesISBN links support NWE through referral fees
- ↑ International Union of Pure and Applied Chemistry. "photochemistry". Compendium of Chemical Terminology Internet edition.
| ||||||||
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.