Difference between revisions of "Fructose" - New World Encyclopedia
| Line 2: | Line 2: | ||
'''Fructose''' (or '''levulose''') is a simple [[sugar]] ([[monosaccharide]]) with the chemical formula ([[carbon|C]]<sub>6</sub>[[hydrogen|H]]<sub>12</sub>[[oxygen|O]]<sub>6</sub>); it is an [[isomer]] of [[glucose]]. Along with glucose and [[galactose]], fructose is one of the three most important [[blood sugar]]s in [[animal]]s. | '''Fructose''' (or '''levulose''') is a simple [[sugar]] ([[monosaccharide]]) with the chemical formula ([[carbon|C]]<sub>6</sub>[[hydrogen|H]]<sub>12</sub>[[oxygen|O]]<sub>6</sub>); it is an [[isomer]] of [[glucose]]. Along with glucose and [[galactose]], fructose is one of the three most important [[blood sugar]]s in [[animal]]s. | ||
| − | Sources of fructose include [[honey]], [[fruit]]s, and some root [[vegetable]]s. Fructose is often found in combination with glucose as the disaccharide [[sucrose]], a readily transportable and mobilizable sugar that is stored in many plant cells, such as in sugar beets and sugar cane. In animals, fructose may also be utilized as an energy source, and phosphate derivatives of fructose participate in the metabolism of carbohydrates. | + | Sources of fructose include [[honey]], [[fruit]]s, and some root [[vegetable]]s. Fructose is often found in combination with glucose as the disaccharide [[sucrose]], a readily transportable and mobilizable sugar that is stored in many plant cells, such as in sugar beets and sugar cane. In animals, fructose may also be utilized as an energy source, and [[phosphate]] derivatives of fructose participate in the metabolism of [[carbohydrates]]. |
| − | + | Fructose’s [[Glycemic Index]]—i.e., the measure of the speed at which particular carbohydrates are converted into glucose in the body—is relatively low compared to other simple sugars because of its slower absorption into the bloodstream. Thus, fructose may be recommended for patients with [[diabetes mellitus]] or [[hypoglycemia]] (low blood sugar), because intake does not trigger high levels of [[insulin]] secretion. This benefit is tempered by a concern that fructose may have an adverse effect on plasma lipid and uric acid levels and that higher blood levels of fructose can be damaging to [[protein]]s. | |
| − | + | In addition to natural sources, fructose may be found in commercially produced [[high fructose corn syrup]] (HFCS), in which the fructose content of corn syrup ([[glucose]]) is increased through enzymatic processing. While most [[carbohydrates]] have around the same amount of [[calories]], fructose is generally perceived as sweeter than glucose, so [[manufacturers]] may use less fructose to achieve the same perception of sweetness as found in sucrose (“table sugar”). This property makes HFCS useful to manufacturers as a possible substitute for sugar in soft drinks and other processed foods. There are currently suspicions that over-consumption of HFCS may be a contributor to the epidemic of obesity and Type II diabetes in the United States. However, the obesity epidemic has many contributing factors. <ref>[http://www.sfgate.com/cgi-bin/article.cgi?f=/chronicle/archive/2004/02/18/FDGS24VKMH1.DTL Sugar coated: We're drowning in high fructose corn syrup.]</ref> | |
==The chemical structure of fructose== | ==The chemical structure of fructose== | ||
| Line 12: | Line 12: | ||
Fructose is a [[levorotation|levorotatory]] monosaccharide with the same empirical formula as [[glucose]] but with a different structure (i.e., it is an isomer of glucose). Like glucose, fructose is a ''hexose'' (six-carbon) sugar, but it contains a [[keto]] group instead of an [[aldehyde]] group, making it a ''ketohexose''. | Fructose is a [[levorotation|levorotatory]] monosaccharide with the same empirical formula as [[glucose]] but with a different structure (i.e., it is an isomer of glucose). Like glucose, fructose is a ''hexose'' (six-carbon) sugar, but it contains a [[keto]] group instead of an [[aldehyde]] group, making it a ''ketohexose''. | ||
| − | Like glucose, the fructose can also exist in ring form. Its open-chain structure is able to cyclize (form a ring) because a ketone can react with an alcohol to form a [[hemiketal]]. Specifically, the C-2 keto group in the open-chain form of fructose can react with its C-5 hydroxyl group to form an ''intramolecular hemiketal''. Thus, although fructose is a hexose, it forms a five-membered ring called a [[furanose]], a structure that predominates in solution. | + | Like glucose, the fructose can also exist in ring form. Its open-chain structure is able to cyclize (form a ring) because a ketone can react with an alcohol to form a [[hemiketal]]. Specifically, the C-2 keto group in the open-chain form of fructose can react with its C-5 hydroxyl group to form an ''intramolecular hemiketal''. Thus, although fructose is a hexose, it may forms a five-membered ring called a [[furanose]], a structure that predominates in solution. |
Fructose's specific [[conformation]] (or structure) is responsible for its unique physical and chemical properties relative to glucose. Although the perception of sweetness depends on a variety of factors, such as concentration, pH, temperature, and individual taste buds, fructose is estimated to be approximately 1.2-1.8 times sweeter than glucose. | Fructose's specific [[conformation]] (or structure) is responsible for its unique physical and chemical properties relative to glucose. Although the perception of sweetness depends on a variety of factors, such as concentration, pH, temperature, and individual taste buds, fructose is estimated to be approximately 1.2-1.8 times sweeter than glucose. | ||
== Fructose as an energy source== | == Fructose as an energy source== | ||
| − | + | Most of the dietary fructose passes through the [[liver]], a control point for the circulation of blood sugar. | |
| − | + | In nearly all organisms, energy from [[carbohydrates]] is obtained via [[glycolysis]], a series of [[biochemistry|biochemical]] reactions by which one [[molecule]] of [[Glucose|glucose (Glc)]] is [[oxidized]] to two molecules of pyruvic acid (Pyr), yielding a small net gain of chemical energy. For aerobic organisms such as [[human]]s, glycolysis is only the initial stage of [[carbohydrate]] [[metabolism|catabolism]]; the end-products of glycolysis typically enter into the [[citric acid cycle]] (also known as the TCA or Krebs cycle) and the electron transport chain for further oxidation. These pathways together produce considerably more energy per glucose molecule than anaerobic oxidation. | |
| − | + | Unlike glucose, fructose must undergo a few modifications before it is able to enter the glycolytic pathway. One method occurs in the fructose-1-phosphate pathway, preferred by the liver. There are three steps involved: | |
| + | #The [[phosphorylation]] (addition of a phosphate molecule) of fructose by the enzyme [[fructokinase]]. | ||
| + | #A split of the six-carbon fructose into two three-carbon molecules, ‘’glyceraldehyde’’ and ‘’dihydroxyacetone phosphate’’ (an aldol cleavage). | ||
| + | #Glyceraldehyde is then phosphorylated by another enzyme so that it too can enter glycolysis. | ||
| − | + | Alternatively, fructose can enter via another route: it can be phosphorylated to fructose-6-phosphate (F6P) by the enzyme [[hexokinase]]. Little F6P is formed in the liver given the abundance of glucose relative to fructose. In contrast, [[adipose]] tissue (a specialized [[fat]]-storage tissue) has much more fructose than glucose. Thus, most of the fructose in adipose tissue is mobilized by this latter pathway. | |
==Disorders involving fructose metabolism== | ==Disorders involving fructose metabolism== | ||
| − | Fructose | + | '''Fructose intolerance''' ('''Hereditary Fructose Intolerance''', or '''HFI''') is a [[hereditary condition]] --a deficiency of liver [[enzyme]]s that metabolize [[fructose]]. The deficient enzyme is [[Fructose-1-phosphate aldolase-B]], this means that the fructose cannot be further metabolised beyond fructose-1-phosphate. This traps [[phosphate]]s; which are needed to phosphorolyse [[glycogen phosphorolase]] to carry on to make [[glucose]]. Therefore glucose cannot be made through the breakdown of [[glycogen]] nor from [[gluconeogenesis]], resulting in severe [[hypoglycaemia]]. If fructose is ingested, vomiting, hypoglycaemia and eventually kidney failure will follow. |
| − | + | Hereditary Fructose Intolerance should not be confused with [[fructose malabsorption]] or Dietary Fructose Intolerance (DFI), a deficiency of fructose transporter enzyme in the [[enterocytes]], which leads to abdominal [[bloating]], [[diarrhea]] and/or [[constipation]]. In patients with [[fructose malabsorption]], the small intestine fails to absorb fructose properly. In the large intestine, the unabsorbed fructose osmotically reduces the absorption of water and is metabolized by normal colonic bacteria to short chain fatty acids and the gases hydrogen, carbon dioxide and methane. Foods with a high glucose content actually help sufferers absorb fructose. | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | Hereditary Fructose Intolerance should not be confused with [[fructose malabsorption]] or Dietary Fructose Intolerance (DFI), a deficiency of fructose transporter enzyme in the [[enterocytes]], which leads to abdominal [[bloating]], [[diarrhea]] and/or [[constipation]] | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | Foods with a high glucose content actually help sufferers absorb fructose. | ||
==The commercial use of high fructose corn syrup== | ==The commercial use of high fructose corn syrup== | ||
| Line 52: | Line 39: | ||
By increasing the [[fructose]] content of corn syrup ([[glucose]]) through enzymatic processing, the syrup is more comparable to table sugar ([[sucrose]]). This makes it useful to manufacturers as a possible substitute for sugar in soft drinks and other processed foods. Common commercial grades of high fructose corn syrup include fructose contents of 42%, 55%, or 90%. The 55% grade is most commonly used in soft drinks and equivalent to [[caster sugar]]. | By increasing the [[fructose]] content of corn syrup ([[glucose]]) through enzymatic processing, the syrup is more comparable to table sugar ([[sucrose]]). This makes it useful to manufacturers as a possible substitute for sugar in soft drinks and other processed foods. Common commercial grades of high fructose corn syrup include fructose contents of 42%, 55%, or 90%. The 55% grade is most commonly used in soft drinks and equivalent to [[caster sugar]]. | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
===Sweetener consumption patterns=== | ===Sweetener consumption patterns=== | ||
Revision as of 11:47, 19 October 2006
Fructose (or levulose) is a simple sugar (monosaccharide) with the chemical formula (C6H12O6); it is an isomer of glucose. Along with glucose and galactose, fructose is one of the three most important blood sugars in animals.
Sources of fructose include honey, fruits, and some root vegetables. Fructose is often found in combination with glucose as the disaccharide sucrose, a readily transportable and mobilizable sugar that is stored in many plant cells, such as in sugar beets and sugar cane. In animals, fructose may also be utilized as an energy source, and phosphate derivatives of fructose participate in the metabolism of carbohydrates.
Fructose’s Glycemic Index—i.e., the measure of the speed at which particular carbohydrates are converted into glucose in the body—is relatively low compared to other simple sugars because of its slower absorption into the bloodstream. Thus, fructose may be recommended for patients with diabetes mellitus or hypoglycemia (low blood sugar), because intake does not trigger high levels of insulin secretion. This benefit is tempered by a concern that fructose may have an adverse effect on plasma lipid and uric acid levels and that higher blood levels of fructose can be damaging to proteins.
In addition to natural sources, fructose may be found in commercially produced high fructose corn syrup (HFCS), in which the fructose content of corn syrup (glucose) is increased through enzymatic processing. While most carbohydrates have around the same amount of calories, fructose is generally perceived as sweeter than glucose, so manufacturers may use less fructose to achieve the same perception of sweetness as found in sucrose (“table sugar”). This property makes HFCS useful to manufacturers as a possible substitute for sugar in soft drinks and other processed foods. There are currently suspicions that over-consumption of HFCS may be a contributor to the epidemic of obesity and Type II diabetes in the United States. However, the obesity epidemic has many contributing factors. [1]
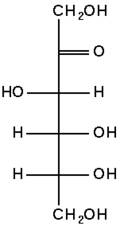
The chemical structure of fructose
Fructose is a levorotatory monosaccharide with the same empirical formula as glucose but with a different structure (i.e., it is an isomer of glucose). Like glucose, fructose is a hexose (six-carbon) sugar, but it contains a keto group instead of an aldehyde group, making it a ketohexose.
Like glucose, the fructose can also exist in ring form. Its open-chain structure is able to cyclize (form a ring) because a ketone can react with an alcohol to form a hemiketal. Specifically, the C-2 keto group in the open-chain form of fructose can react with its C-5 hydroxyl group to form an intramolecular hemiketal. Thus, although fructose is a hexose, it may forms a five-membered ring called a furanose, a structure that predominates in solution.
Fructose's specific conformation (or structure) is responsible for its unique physical and chemical properties relative to glucose. Although the perception of sweetness depends on a variety of factors, such as concentration, pH, temperature, and individual taste buds, fructose is estimated to be approximately 1.2-1.8 times sweeter than glucose.
Fructose as an energy source
Most of the dietary fructose passes through the liver, a control point for the circulation of blood sugar.
In nearly all organisms, energy from carbohydrates is obtained via glycolysis, a series of biochemical reactions by which one molecule of glucose (Glc) is oxidized to two molecules of pyruvic acid (Pyr), yielding a small net gain of chemical energy. For aerobic organisms such as humans, glycolysis is only the initial stage of carbohydrate catabolism; the end-products of glycolysis typically enter into the citric acid cycle (also known as the TCA or Krebs cycle) and the electron transport chain for further oxidation. These pathways together produce considerably more energy per glucose molecule than anaerobic oxidation.
Unlike glucose, fructose must undergo a few modifications before it is able to enter the glycolytic pathway. One method occurs in the fructose-1-phosphate pathway, preferred by the liver. There are three steps involved:
- The phosphorylation (addition of a phosphate molecule) of fructose by the enzyme fructokinase.
- A split of the six-carbon fructose into two three-carbon molecules, ‘’glyceraldehyde’’ and ‘’dihydroxyacetone phosphate’’ (an aldol cleavage).
- Glyceraldehyde is then phosphorylated by another enzyme so that it too can enter glycolysis.
Alternatively, fructose can enter via another route: it can be phosphorylated to fructose-6-phosphate (F6P) by the enzyme hexokinase. Little F6P is formed in the liver given the abundance of glucose relative to fructose. In contrast, adipose tissue (a specialized fat-storage tissue) has much more fructose than glucose. Thus, most of the fructose in adipose tissue is mobilized by this latter pathway.
Disorders involving fructose metabolism
Fructose intolerance (Hereditary Fructose Intolerance, or HFI) is a hereditary condition —a deficiency of liver enzymes that metabolize fructose. The deficient enzyme is Fructose-1-phosphate aldolase-B, this means that the fructose cannot be further metabolised beyond fructose-1-phosphate. This traps phosphates; which are needed to phosphorolyse glycogen phosphorolase to carry on to make glucose. Therefore glucose cannot be made through the breakdown of glycogen nor from gluconeogenesis, resulting in severe hypoglycaemia. If fructose is ingested, vomiting, hypoglycaemia and eventually kidney failure will follow.
Hereditary Fructose Intolerance should not be confused with fructose malabsorption or Dietary Fructose Intolerance (DFI), a deficiency of fructose transporter enzyme in the enterocytes, which leads to abdominal bloating, diarrhea and/or constipation. In patients with fructose malabsorption, the small intestine fails to absorb fructose properly. In the large intestine, the unabsorbed fructose osmotically reduces the absorption of water and is metabolized by normal colonic bacteria to short chain fatty acids and the gases hydrogen, carbon dioxide and methane. Foods with a high glucose content actually help sufferers absorb fructose.
The commercial use of high fructose corn syrup
Production
High fructose corn syrup (HFCS) is a newer and sweeter form of corn syrup. Like ordinary corn syrup, the high fructose variety is made from corn starch using enzymes. The production process of HFCS was developed by Japanese researchers in the 1970s. HFCS was rapidly introduced in many processed foods and soda drinks in the US over the period of about 1975–1985, and usage continues to increase as sugar use decreases at a nearly one to one level (Bray, 2004 & U.S. Department of Agriculture, Economic Research Service, Sugar and Sweetener Yearbook series, Tables 50–52.). There are three main reasons for this switch; first is cost, as HFCS is a bit cheaper due to corn subsidies and import sugar tariffs. The second reason is that it is a liquid which is easier to blend and transport. The third is that a product made with HFCS has a much longer shelf life. (White JS. 1992. Fructose syrup: production, properties and applications, in FW Schenck & RE Hebeda, eds, Starch Hydrolysis Products – Worldwide Technology, Production, and Applications. VCH Publishers, Inc. 177-200)
By increasing the fructose content of corn syrup (glucose) through enzymatic processing, the syrup is more comparable to table sugar (sucrose). This makes it useful to manufacturers as a possible substitute for sugar in soft drinks and other processed foods. Common commercial grades of high fructose corn syrup include fructose contents of 42%, 55%, or 90%. The 55% grade is most commonly used in soft drinks and equivalent to caster sugar.
Sweetener consumption patterns
The accompanying graph shows the consumption of sweeteners per capita in the United States since 1966. Since HFCS and sucrose (cane and beet sugars) provide almost identical proportions of fructose and glucose, no metabolic changes would be expected from substituting one for the other. However, it is apparent from this graph that overall sweetener consumption, and in particular glucose-fructose mixtures, has increased since the introduction of HFCS. Thus, the proportion of fructose as a component of overall sweetener intake in the United States has increased since the early 1980s. This would be true whether the added sweetener was HFCS, table sugar, or any other glucose-fructose mixture.
HFCS is produced in the industrialized countries.The production of HFCS is dependent on the agricultural, especially sugar, policy.
In Europe, due to the fact that HFCS (isoglucose) is under the adjustment of production, the greater availability of cane sugar over maize would make its production there uneconomical.
In Japan, HFCS consumption accounts for one quarter of total sweetener consumption.
The potential impact on human health
The average American consumed approximately 19.2 kg of HFCS versus 20 kg of sugar in 2004.[citation needed] Where HFCS is not used or rarely used, the sugar consumption per person can be higher than the USA; for example, the 2002 figures for some countries are: USA 32.4 kg, EU 40.1 kg, Brazil 59.7 kg, and Australia 56.2 kg.[2]
One study concluded that fructose "produced significantly higher fasting plasma triacylglycerol values than did the glucose diet in men" and "if plasma triacylglycerols are a risk factor for cardiovascular disease, then diets high in fructose may be undesirable"[3]. A study in mice suggests that fructose increases adiposity.[4] However, these studies looked at the effects of fructose alone. As noted by the U.S. Food and Drug Administration in 1996, the saccharide composition (glucose to fructose ratio) of HFCS is approximately the same as that of honey, invert sugar and the disaccharide sucrose (or table sugar).
A more recent study found a link exists between obesity and high HFCS consumption, especially from soft drinks.[5]
However, the obesity epidemic has many contributing factors. University of California, Davis nutrition researcher Peter Havel has pointed out that while there are likely differences between sweeteners, "the increased consumption of fat, the increased consumption of all sugars, and inactivity are all to blame for the obesity epidemic."[6]
ReferencesISBN links support NWE through referral fees
- ↑ Sugar coated: We're drowning in high fructose corn syrup.
- ↑ WHO Oral Health Country/Area Profile Programme Global Sugar Consumption
- ↑ Bantle, John P. and Susan K. Raatz, William Thomas and Angeliki Georgopoulos (November 2000). Effects of dietary fructose on plasma lipids in healthy subjects. American Journal of Clinical Nutrition 72 (5): 1128-1134.
- ↑ Jurgens, Hella and et al. (2005). Consuming Fructose-sweetened Beverages Increases Body Adiposity in Mice. Obesity Res 13: 1146-1156.
- ↑ Bray, George A. and Samara Joy Nielsen and Barry M. Popkin (April 2004). Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. American Journal of Clinical Nutrition 79 (4): 537-543.
- ↑ Warner, Melanie, "A Sweetener With a Bad Rap", New York Times, 2006-07-02. Retrieved 2006-10-03.
- Levi B, Werman MJ. Long-term fructose consumption accelerates glycation and several age-related variables in male rats. J Nutr 1998;128:1442-9. Fulltext. PMID 9732303.
- McPherson JD, Shilton BH, Walton DJ. Role of fructose in glycation and cross-linking of proteins. Biochemistry 1988;27:1901-7. PMID 3132203.
- Higdon, J., Linus Pauling Institute, Oregon State U. Chromium 2003
- Wylie-Rosett,Judith, et al, Carbohydrates and Increases in Obesity: Does the Type of Carbohydrate Make a Difference? Obesity Research 12:124S-129S (2004)[1]
- Havel, PJ, Dietary fructose: Implications for dysregulation of energy homeostasis and lipid/carbohydrate metabolism. Nutr Rev. 2005 May;63(5):133-57 [2]
- Bray, George A, Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity American Journal of Clinical Nutrition, Vol. 79, No. 4, 537-543, April 2004 [3]
- Dennison, Barbara Excess Fruit Juice Consumption by Preschool-aged Children Is Associated With Short Stature and Obesity, PEDIATRICS Vol. 99 No.1, January 1997, pp. 15-22 [4]
External links
Template:ChemicalSources
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.