Difference between revisions of "Guanine" - New World Encyclopedia
Rick Swarts (talk | contribs) |
Rick Swarts (talk | contribs) |
||
| Line 30: | Line 30: | ||
! {{chembox header}} | Properties | ! {{chembox header}} | Properties | ||
|- | |- | ||
| − | | [[Density]]* and [[Phase (matter)|phase]] | + | | [[Density]]* and [[Phase (matter)|phase]]* |
| ? g/cm<sup>3</sup>, solid. | | ? g/cm<sup>3</sup>, solid. | ||
|- | |- | ||
| − | | [[Solubility]] in [[Water (molecule)|water]]* | + | | [[Solubility]]* in [[Water (molecule)|water]]* |
| Insoluable. | | Insoluable. | ||
|- | |- | ||
| Line 98: | Line 98: | ||
|- | |- | ||
|} | |} | ||
| − | '''Guanine''' is one of the five main [[nucleotide#Chemical structure and nomenclature|nucleobase]]s found in the [[nucleic acid]]s [[DNA]] and [[RNA]]; the others being [[adenine]], [[cytosine]], [[thymine]], and [[uracil]]. | + | '''Guanine''' is one of the five main [[nucleotide#Chemical structure and nomenclature|nucleobase]]s found in the [[nucleic acid]]s [[DNA]] and [[RNA]]; the others being [[adenine]], [[cytosine]], [[thymine]], and [[uracil]]. Guanine and adenine are [[purine]] derviatives, and cytosine, thymine, and uracil are pyrimidine derivaties. |
| − | + | With the formula C<sub>5</sub>H<sub>5</sub>N<sub>5</sub>O, guanine consists of a fused [[pyrimidine]]-imidazole ring system with conjugated double bonds. Being unsaturated, the bicyclic molecule is planar. The guanine [[nucleotide#Chemical structure and nomenclature|nucleoside]] is called guanosine. | |
| − | + | In DNA, guanine and adenine form hydrogen bonds with their complementary pyrimidine derivatives, cytosine and thymine. In RNA, the complement of adenine is uracil instead of thymine. Thus, guanine, along with adenine and cytosine, is present in both DNA and RNA, whereas thymine is usually seen only in DNA and uracil only in RNA. | |
| − | + | ||
| − | It binds to cytosine through three | + | ==Basic properties== |
| + | Guanine has two tautomeric forms, the keto form and enol form. It binds to cytosine through three hydrogen bonds. In cytosine, the amino group acts as the hydrogen donor and the C-2 carbonyl and the N-3 amine as the hydrogen-bond acceptors. Guanine has a group at C-6 that acts as the hydrogen acceptor, while the group at N-1 and the amino group at C-2 acts as the hydrogen donors. | ||
{| align="center" | {| align="center" | ||
|- | |- | ||
| Line 111: | Line 112: | ||
|} | |} | ||
| − | + | Guanine can be hydrolyzed with strong acid at 180°C to glycine, ammonia, [[carbon dioxide]], and [[carbon monoxide]]. Guanine oxidizes more readily than [[adenine]], the other purine-derivative base in DNA and RNA. Its high melting point of 350°C reflects the strong intermolecular hydrogen bonding between the oxo and amino groups in the molecules in the crystal. Because of this intermolecular bonding, guanine is relatively insoluble in water, although it is soluble in dilute acids and bases. | |
| − | |||
| − | + | ==History== | |
| + | The first isolation of guanine was reported in 1844 from sea bird excreta, known as guano, which was used as a source of fertilizer. About fifty years later, the structure was determined by Fischer and it was shown that uric acid can be converted to guanine. The first complete synthesis was done by Traube and remains among the best large-scale preparations. | ||
==Syntheses== | ==Syntheses== | ||
Revision as of 19:29, 30 December 2006
| Guanine | |
|---|---|
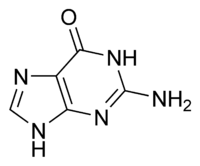
| |
| General | |
| Systematic name | 2-amino-1H-purin-6(9H)-one |
| Other names | 2-amino-6-oxo-purine, 2-aminohypoxanthine, Guanine |
| Molecular formula | C5H5N5O |
| SMILES | NC(NC1=O)=NC2=C1N=CN2 |
| Molar mass | 151.1261 g/mol |
| Appearance | White amorphous solid. |
| CAS number | [73-40-5] [1] |
| Properties | |
| Density and phase | ? g/cm3, solid. |
| Solubility in water | Insoluable. |
| Melting point | 360°C (633.15 K) deco. |
| Boiling point | Sublimes. |
| Structure | |
| Crystal structure | ? |
| Dipole moment | ? D |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | Irritant. |
| NFPA 704 | |
| Flash point | Non-flammable. |
| R/S statement | R: R36, R37, R38. S: R24/25, R26, R36. |
| RTECS number | MF8260000 |
| Supplementary data page | |
| Structure and properties |
n, εr, etc. |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other anions | ? |
| Other cations | ? |
| Related compounds | Cytosine, Adenine, Thymine, Uracil |
| Except where noted otherwise, data are given for materials in their standard state (at 25°C, 100 kPa) | |
Guanine is one of the five main nucleobases found in the nucleic acids DNA and RNA; the others being adenine, cytosine, thymine, and uracil. Guanine and adenine are purine derviatives, and cytosine, thymine, and uracil are pyrimidine derivaties.
With the formula C5H5N5O, guanine consists of a fused pyrimidine-imidazole ring system with conjugated double bonds. Being unsaturated, the bicyclic molecule is planar. The guanine nucleoside is called guanosine.
In DNA, guanine and adenine form hydrogen bonds with their complementary pyrimidine derivatives, cytosine and thymine. In RNA, the complement of adenine is uracil instead of thymine. Thus, guanine, along with adenine and cytosine, is present in both DNA and RNA, whereas thymine is usually seen only in DNA and uracil only in RNA.
Basic properties
Guanine has two tautomeric forms, the keto form and enol form. It binds to cytosine through three hydrogen bonds. In cytosine, the amino group acts as the hydrogen donor and the C-2 carbonyl and the N-3 amine as the hydrogen-bond acceptors. Guanine has a group at C-6 that acts as the hydrogen acceptor, while the group at N-1 and the amino group at C-2 acts as the hydrogen donors.
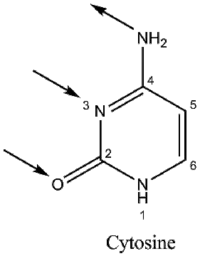
|
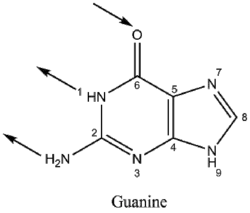
|
Guanine can be hydrolyzed with strong acid at 180°C to glycine, ammonia, carbon dioxide, and carbon monoxide. Guanine oxidizes more readily than adenine, the other purine-derivative base in DNA and RNA. Its high melting point of 350°C reflects the strong intermolecular hydrogen bonding between the oxo and amino groups in the molecules in the crystal. Because of this intermolecular bonding, guanine is relatively insoluble in water, although it is soluble in dilute acids and bases.
History
The first isolation of guanine was reported in 1844 from sea bird excreta, known as guano, which was used as a source of fertilizer. About fifty years later, the structure was determined by Fischer and it was shown that uric acid can be converted to guanine. The first complete synthesis was done by Traube and remains among the best large-scale preparations.
Syntheses
Trace amounts of guanine form by the polymerization of ammonium cyanide (NH4CN). Two experiments conducted by Levy et al., showed that heating 10 M NH4CN at 80°C for 24 hours gave a yield of 0.0007% while using 0.1 M NH4CN frozen at -20°C for 25 years gave a 0.0035% yield. These results indicate guanine could arise in frozen regions of the primitive earth. In 1984, Yuasa reported a 0.00017% yield of guanine after the electrical discharge of NH3, CH4, C2H6, and 50 mL of water, followed by a subsequent acid hydrolysis. However, it is unknown if the presence of guanine was not simply resulted from a contaminant of the reaction.
- 5NH3 + CH4 + 2C2H6 + H2O → C5H8N5O (guanine) + (25/2)H2
A Fischer-Tropsch synthesis can also be used to form guanine, along with adenine, uracil and thymine. Heating an equimolar gas mixture of CO, H2, and NH3 to 700 °C for 0.24 to 0.4 hours, followed by quick cooling and then sustainted reheating to 100-200°C for 16-44 hours with an alumina catalyst yielded guanine and uracil:
- 5CO + (1/2)H2 + 5NH3 → C5H8N5O (guanine) + 4H2O
Traube's synthesis involves heating 2,4,5-triamino-1,6-dihydro-6-oxypyrimidine (as the sulphate) with formic acid for several hours.
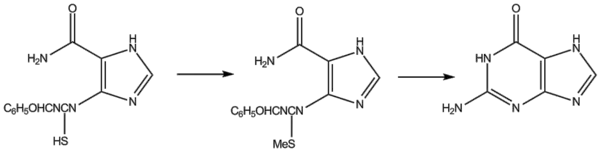
Other uses
In 1656 in Paris, François Jaquin (a rosary maker) extracted from scales of some fishes the so called pearl essence, crystalline guanine forming G-quadruplexes: in cosmetic industry, crystalline guanine is used as an additive to various products (e.g., shampoos), where it provides the pearly iridescent effect. It is also used in metallic paints and simulated pearls and plastics. It provides shimmering lustre to eye shadow and nail polish. Guanine crystals are rhombic platelets composed of multiple, transparent layers but they have a high index of refraction that partially reflects and transmits light from layer to layer thus producing a pearly luster. It can be applied by spray, painting or dipping. It may irritate eyes. Its alternatives are mica, synthetic pearl, and aluminium and bronze particles.
ReferencesISBN links support NWE through referral fees
1. Miyakawa, S., Murasawa, K., Kobayashi, K., Sawaoka, AB. "Abiotic synthesis of guanine with high-temperature plasma." Orig Life Evol Biosph. 30(6): 557-66, Dec. 2000. <br\> 2. Horton, H.R., Moran, L.A., Ochs, R.S., Rawn, J.D., Scrimgeour, K.G. "Principles of Biochemistry." Prentice Hall (New Jersey). 3rd Edition, 2002. <br\> 3. Lister, J.H. "Part II Purines." The Chemistry of Heterocyclic Compounds. Wiley-Interscience (New York). 1971.
External links
| Nucleic acids edit |
|---|
| Nucleobases: Adenine - Thymine - Uracil - Guanine - Cytosine - Purine - Pyrimidine |
| Nucleosides: Adenosine - Uridine - Guanosine - Cytidine - Deoxyadenosine - Thymidine - Deoxyguanosine - Deoxycytidine |
| Nucleotides: AMP - UMP - GMP - CMP - ADP - UDP - GDP - CDP - ATP - UTP - GTP - CTP - cAMP - cGMP |
| Deoxynucleotides: dAMP - dTMP - dUMP - dGMP - dCMP - dADP - dTDP - dUDP - dGDP - dCDP - dATP - dTTP - dUTP - dGTP - dCTP |
| Nucleic acids: DNA - RNA - LNA - PNA - mRNA - ncRNA - miRNA - rRNA - siRNA - tRNA - mtDNA - Oligonucleotide |
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.
